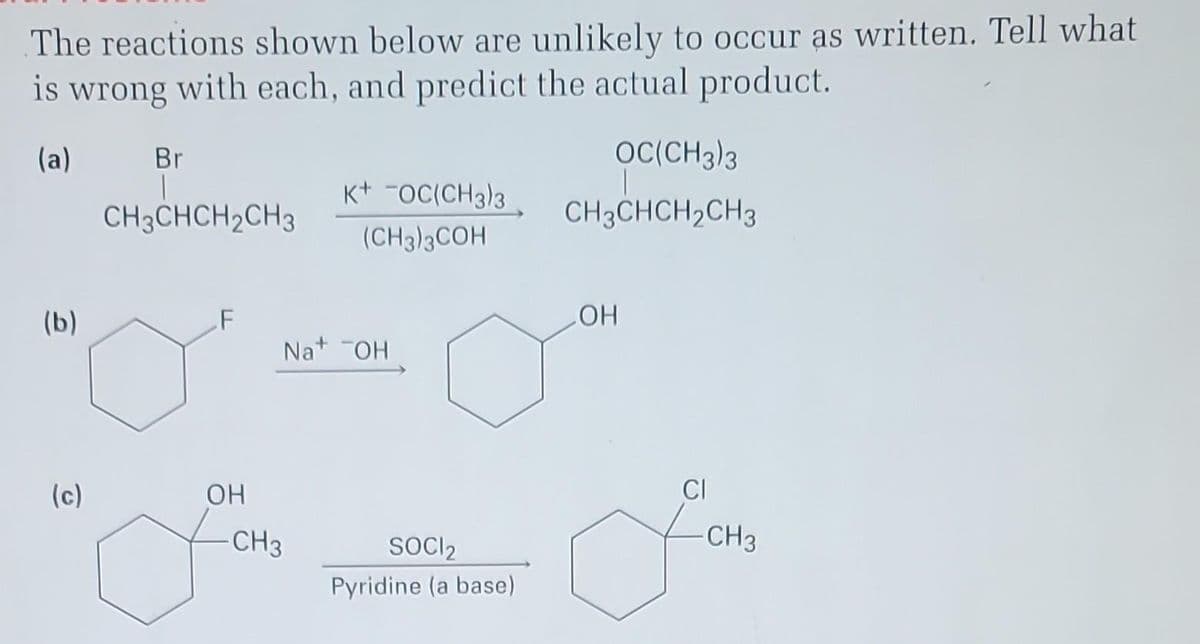
The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actual product. (a) (b) (c) Br CH3CHCH2CH3 OH K+ OC(CH3)3 (CH3)3COH Na+ OH - CH3 SOCI2 Pyridine (a base) OC(CH3)3 CH3CHCH₂CH3 OH -CH3
Q: Which of the following compounds exhibit hydrogen bonding in the pure liquid state? (The lone pair…
A:
Q: This is the chemical formula for nickel tetracarbonyl (a powerfully poisonous liquid used in nickel…
A: we have to calculate the number of moles of O
Q: explain that has atom has 7 prtons, 8 neutrons, 7 electronsatomic number is 7, atomic mass is 15…
A: An atom consists of positively charged nucleus in the center that is surrounded by negatively…
Q: In each row of the table, select the stronger base. N O S O O O N_ O
A: According to Lewis base is a substance which can donate a pair of electrons. Basicity of a compound…
Q: A 1,365.8 mL aqueous solution of 407.9 mM Na2S is combined with 100.0 mL of an aqueous HCl solution…
A: Given , Na2S reacts with HCI
Q: A solution of organic solvents has been prepared by mixing 45.0 mL of ethylbenzene, 300.0 mL of…
A: The weight percent, molality, and molarity are the different concentration terms. The weight…
Q: An insulin drip is mixed as 500.0 units in 250.0 mL Normal Saline (NS). Calculate the drip rate in…
A:
Q: A chemist measures the amount of nitrogen gas produced during an experiment. She finds that 51.1 g…
A: Given, mass of Nitrogen (N2) gas produced = 51.1 g moles of Nitrogen (N2) gas produced = ? Note:…
Q: What is the hybridization of each labeled atom below? A: sp²; B: sp; C: sp²; D: sp3 A: sp²; B: sp³;…
A: We are given a structure of a large organic molecule and asked to write the hybridization of the…
Q: Q8. Calculate the second ionization energy of He. A) 8.71 x 10-21 J B) 8.71 x 10-15 J C) 8.71 x…
A: Ionization energy is the amount of energy required to remove an electron from a neutral atom or ion.…
Q: 30. A single displacement reaction occurs between copper and silver nitrate. When 63.5 g of copper…
A:
Q: Draw the structure of the molecule and upload it here. Molecular Formula: C,H₁002 -1 FTIR: Strong…
A:
Q: Identify the structure that is not correct. O
A: This question is related to reaction intermediates. Reaction intermediates are of many types such as…
Q: What is the freezing point (in degrees Celcius) of 3.97 kg of water if it contains 151.6 g of…
A:
Q: A patient is to receive: Nitroglycerin 10.0 mcg/min continuous IV infusion via non-absorptive…
A:
Q: Combustion analysis of an unknown compound containing only carbon and hydrogen produced 2.845 g of…
A: Since, Empirical formula is the smallest whole number of the ratio of atoms in a molecule. Thus,
Q: A 25 mL sample is pipetted from a AB, stock solution. Which answer correctly describes the…
A: 25 ml sample is pipetted from a AB3 stock solution
Q: How would you expect the second ionization energies for Na and Mg to compare? Explain using both a…
A: Solution - The first ionisation energy of magnesium is higher than sodium and second ionisation…
Q: 0.0519 kg of biphenyl (C₁₂H₁₀) is dissolve in benzene (C₆H₆) to create a solution with a total…
A: Given , Biphenyl is mixed with benzene.
Q: Q4. Which of the following functions is NOT an eigenfunction of the operator d/dr²: A cos(kx) B. sin…
A: Since, Function is known as an eigen function if after operating it is not as multiple of part of…
Q: Balance the chemical equation given below, and determine the number of grams of Fe needed to produce…
A:
Q: Below is a solubility table for an unknown organic soli. The solid is white with a melting point of…
A: Given the solubility data, methanol would be the best recrystallization solvent.In a…
Q: Determine the mass of CO2CO2 produced by burning enough carbon (in the form of charcoal) to produce…
A: The balanced chemical reaction is C(s)+O2 (g)→CO2(g); ΔrH∘=−393.5 kJ-mol-1 The mass of CO2…
Q: The rate of a certain reaction was studied at various temperatures. The table shows temperature (T)…
A:
Q: You are planning to run a gel using a Tris-HCl buffer. You have 0.75 M Tris-HCl, pH 7.30 buffer, but…
A:
Q: 2. Draw all products HC- (8 Substitution, 9 elimination) H N CH3 CH3 CH₂OH Δ
A: Substitution will have through the leaving of iodine atom, as iodine (I-) is a good leaving group.…
Q: Search this book 37. What is the half-life for the first-order decay of carbon-14? (14C 14N+e) The…
A:
Q: Decimal Notation Scientific Notation…
A:
Q: Complete the table below by writing the symbols for the cation and anion that make up each ionic…
A: In an ionic compound, total positive charge carried by cations is equal to the total charge carried…
Q: The reactants molecules should collide with the reacting functionalities away from each other and…
A: We have been asked about the requirements for formation of product from colliding reactant…
Q: 1. What is the molar mass of sodium phosphate, Na3PO4(aq)? a. 69.95 g/mol b. 99.04 g/mol c. 117.96…
A: AS PER THE ANSWERING GUIDELINES I CAN ANSWER ONLY THE FIRST ONE, IF YOU WANT THE REST POST THEM…
Q: Name the three alkenes using systematic names. A. H₂C B. H3C CIC The name of compound A is: H3C CH3…
A: IUPAC rule-The ene suffix (ending) indicates an alkene use it by replacing e from alkane 2. The…
Q: (Show your complete solution) For each of the following redox reactions, (i) Write the…
A: Redox reaction is a reaction in which oxidation and reduction take place simultaneously. In this…
Q: Q12. Which of the following functions is antisymmetric? A. v=0.5[1s(1)2s(2)+1s(2)2s(1)][a(1) a (2)]…
A:
Q: A molecule with the molecular formula of C3H8O produces the IR spectrum shown below. Draw a…
A: Infra red spectrum gives information about what about type of functional group present
Q: The solubility of phthalic acid in water is 0.54 g/100ml at 14°C and 18g/100ml at 99°C. What is the…
A: This question involves a phthalic acid covalent compound being dissolved as a solute in a liquid…
Q: How many Pounds of film are there in 120,333 feet of 0.480 mil PET/3.00 mil (LDPE/EVOH/LLDPE) at…
A: The first step is to calculate the area of the film in square feet. To do this, we'll use the…
Q: A 'H NMR signal is observed at 106.0 Hz (downfield of TMS) on a 115-MHz instrument. a) What is the…
A: Chemical shift of protons in 1H-NMR measured in delta (δ) unit in ppm. Because this unit is…
Q: An aqueous solution of a nonionic (that is, nonelectrolyte) solute has a freezing point of -0.238…
A: Given, mass of the solute = 1.34 g mass of H2O = 78.2 g = 0.0782 kg Freezing point of this solution…
Q: 22. If you perform an acid base titration and place 50 mL of 0.25 mol/L acetic acid into an…
A:
Q: A chemist adds 480.0 mL of a 0.00368 mmol/L copper(II) fluoride (CuF₂) solution to a reaction flask.…
A:
Q: Draw the dipeptide Ala-Ala.
A: A peptide bond is an amide-type bond between two amino acids. An amino acid contains amino and…
Q: discharge tube containing an unknown gas emits light with a wavelength of 485 nm. Given the line…
A: You have to identify the gas that emits light with a wavelength of 485nm. From the line spectra of…
Q: The next drug is an a1 blocker. Which statement is considered true concerning this drug? * O H₂N…
A: Given a drug , We have to select the correct option. Now,
Q: Place the following solutions in order of increasing osmotic pressure. 0.15 M C2H6O 0.15M…
A:
Q: Read each description in the first column of the table below. If any chemical element with atomic…
A: Solution- an element is period 3 and group 2A As magnesium is places in group 2A and period 3 so…
Q: The number of double bonds in this molecule is/are: The number of sigma bonds in this molecule…
A: Expanded octet occurs when an atom is able to have more than 8 valence electrons. Example :…
Q: Which of the following statement is correct about moles? A. 1 mole of Argon gas has the same number…
A: We know that, 1 mole of any substance contains = 6.022 x1023 atoms Therefore, 1 mole of Argon…
Q: 150.00 grams of Iron (III) sulfate is dissolved in enough water to prepare 200.0 ml of solution.…
A:
Q: The half-life of 27Mg is 9.50 min. (a) Initially there were 3.60 ×1012 27Mg nuclei present. How…
A:
Give detailed solution and explanation!!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- The sex attractant of the housefly has the formula C23H46. When treated with warm potassium permanganate, this pheromone gives two products: CH3(CH2)12COOH and CH3(CH2)7COOH. Suggest a structure for this sex attractant. Explainwhich part of the structure is uncertainA solution of acetone [(CH3)2C=O] in ethanol (CH3CH2OH) in the presence of a trace of acid was allowed to stand for several days, and a new compound of molecular formula C7H16O2 was formed. The IR spectrum showed only one major peak in the functional group region around 3000 cm−1, and the 1H NMR spectrum is given here. What is the structure of the product?An allylic halide with the chemical formula C5H9Cl undergoes an SN1 reaction with water to produce B and C, two isomeric products. What would be the structure of the allylic halide and the products B and C? What peaks correspond with what protons in B and C?
- Triphenylmethanol is insoluble in water, but when it is treated with concentrated sulfuric acid, a bright yellow solutionresults. As this yellow solution is diluted with water, its color disappears and a precipitate of triphenylmethanol reappears.Suggest a structure for the bright yellow species, and explain this unusual behavior.1Amines are converted into alkenes by a two-step process called Hofmann elimination. SN2 reaction of the amine with an excess of CH3I in the first step yields an intermediate that undergoes E2 reaction when treated with silver oxide as base. Pentylamine, for example, yields 1-pentene. Propose a structure for the intermediate, and explain why it readily undergoes elimination.The base-promoted rearrangement of an -haloketone to a carboxylic acid, known as the Favorskii rearrangement, is illustrated by the conversion of 2-chlorocyclohexanone to cyclopentanecarboxylic acid. It is proposed that NaOH first converts the a-haloketone to the substituted cyclopropanone shown in brackets and then to the sodium salt of cyclopentanecarboxylic acid. (a) Propose a mechanism for base-promoted conversion of 2-chlorocyclohexanone to the proposed intermediate. (b) Propose a mechanism for base-promoted conversion of the proposed intermediate to sodium cyclopentanecarboxylate.
- (a) Arrange the following compounds in an increasing order of their indicated property :(i) Benzoic acid, 4-Nitrobenzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (acid strength)(ii) CH3CH2CH (Br) COOH, CH3CH (Br) CH2COOH,(CH3)2CHCOOH, CH3CH2CH2COOH (acid strength)(b) How would you bring about the following conversions :(i) Propanone to Propene (ii) Benzoic acid to Benzaldehyde(iii) Bromobenzene to 1-phenylethanol2. Predict the product of the reaction and propose an arrow-pushing mechanism.1. Compound A, C9H12, absorbs 3 equivalents of H2 on catalytic hydrogenation over palladium catalyst to give B, C9H18. On the treatment with acidic KMnO4, compound A gives among other things, a ketone that was identified as cyclohexanone. On reaction with NaNH2 in NH3, followed by addition of iodomethane, compound A gives a new hydrocarbon C, C10H14. What are the structures of A , B and C?
- Wolff-Kishner reduction of compound W gave compound A. Treatment of A with m-chloroperbenzoic acid gave B which on reduction with LiAH4 gave C. Oxidation of compound C with chromic acid gave D (C9H14O). Suggest the structures for A, B, C, and D.When 2,2-dibromo-1-phenylpropane is heated overnight with sodium amide at 150 °C, the major product (after addition of water) is a different foul-smelling compound of formula C9H8. Propose a structure for this product, and give a mechanism to account for its formation.The sex pheromone of the common house fly has the molecular formula C23H46. It can be synthesized in the lab using the following synthetic sequence. Give the structures of the pheromone and the intermediates A and B. n-C13H27C≡CH + n-butyl lithium → A (C15H27Li) A + n-C8H17Br → B (C23H44) B + H2 with Lindlar catalyst → the pheromone



