The solubility of alcohols in water O decreases as the carbon chain length increases increases with increasing molecular mass decreases as the number of -OH groups present increases more than one correct response
The solubility of alcohols in water O decreases as the carbon chain length increases increases with increasing molecular mass decreases as the number of -OH groups present increases more than one correct response
General, Organic, and Biological Chemistry
7th Edition
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:H. Stephen Stoker
Chapter19: Lipids
Section19.6: Chemical Reactions Of Triacylglycerols
Problem 1QQ
Related questions
Question
Which option is correct in both question and explain?
Provide clear handwriting and clear image

Transcribed Image Text:The solubility of alcohols in water
decreases as the carbon chain length increases
increases with increasing molecular mass
O decreases as the number of -OH groups present increases
O more than one correct response
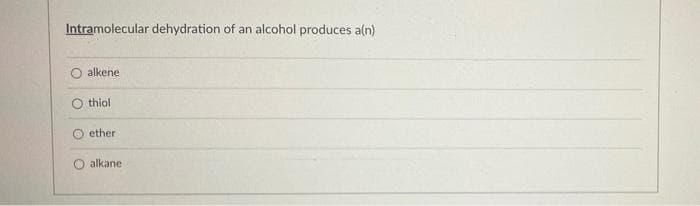
Transcribed Image Text:Intramolecular dehydration of an alcohol produces a(n)
alkene
thiol
ether
alkane
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:
9781305081079
Author:
STOKER, H. Stephen (howard Stephen)
Publisher:
Cengage Learning,

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:
9781305081079
Author:
STOKER, H. Stephen (howard Stephen)
Publisher:
Cengage Learning,

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning