Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.12QAP
Related questions
Question
100%
Transcribed Image Text:ALEKS - Kaîtlin Wo`x
C Solved: Safari Filex
Bb WS 2.3 - KEY.pdf x
G how many kg are
E
A www-awn.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IBMBkpcnaFu0F7UjsroJM
pps
Apple
Focus2 Dashboard
O GRLContent A ALEKS -- Asses...
I Real-Time Lab Us...
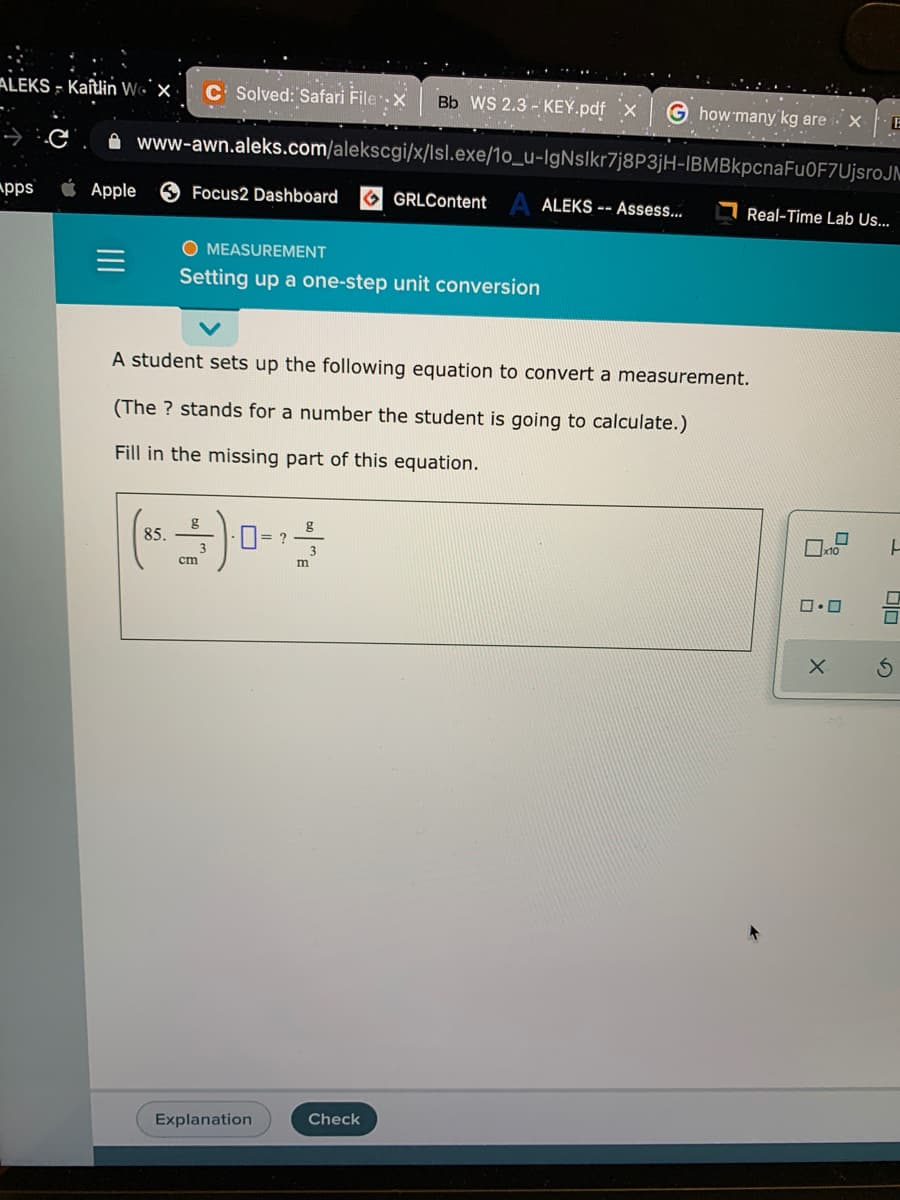
O MEASUREMENT
Setting up a one-step unit conversion
A student sets up the following equation to convert a measurement.
(The ? stands for a number the student is going to calculate.)
Fill in the missing part of this equation.
g
85.
g
cm
m
Check
Explanation
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

