Q: 25. What is E° for the following balance reaction? Zn(s) + Fe* (aq) → Zn*(aq) + Fe(s) K= 4.38 x 100…
A: The value of standard potential and equilibrium constant has a relationship as follows:…
Q: In initial steps of the sol-gel process, a reactive metal is treated with an alcohol to form a metal…
A:
Q: 4 Illustrate how complex formation between Fe3+ and SCN Occurs by presenting electrons by arrows (1)…
A: This question is related to coordination chemistry. Coordination cplexes are the compounds…
Q: MnO aq) + 2+ + C20laq) → COri) + Mn) А. 2 В. 5 С. 10 D. 16 Е. 15
A: The coefficient of a compound can be determined from the balanced chemical reaction. A reaction will…
Q: The number of electrons in a neutral atom spread the element its unique identity. True False
A:
Q: Which set of reagents can be used to achieve this transformation?
A:
Q: Please how did you calculate 1.4007
A:
Q: The mechanism for the reaction described by rst A common NO,(g) + CO(g) → CO,(g) + NO(g) stronger…
A:
Q: What is the IUPAC name?
A: Let us find out its R/S nomenclature and then name it normally according to IUPAC.
Q: The reaction of a Grignard reagent with a carboxylic acid does not give a secondary alcohol because…
A: The reaction of grignard reagent with a carboxylic acid does not give secondary alcohol because of…
Q: write an argument that incorporates 3 pieces of evidence that explains how acids and bases are…
A: There are mainly three theories that identify the particular characteristics of acid and base. They…
Q: A)
A: Here we have to prepare the following given compounds from alkyl halide containing four carbon atom…
Q: The titration reaction: 2Ce4+ + 3I- ⇌ 2Ce3+ + I3- is an example of: Precipitation titration…
A: The titration reaction: 2Ce4+ + 3I- ⇌ 2Ce3+ + I3- is an example of - Iodometry. In the above…
Q: In the amount of YOI consumed, my answer seems to be different from the solution. My answer is 0.319…
A:
Q: How does lime reduce sulfur dioxide emissions from the burning of coal?
A: Coal is primarily made up of carbon and it is readily combustible. The colour of coal is…
Q: calculation of AG° in Kj/mol
A:
Q: ror the following (oordinatien 1omplea (Ma(c.0.)(H,O), C \;} 344 ) De termine the geometry Tuotsong…
A: Here we have write the name , geometry , co-ordination number and geometrical isomers of the…
Q: Identify the functional group and name the following 15. 13. NH2 14.
A: In this question, we will see the functional group in each of the given compound and then given the…
Q: Calcium in a 5.00 mL urine sample was precipitated, redissolved and required 16.15 mL of 0.1157 N…
A:
Q: The following data are for a liquid chromatographic column: Length of Packing 24.7 cm Flow rate…
A: We will calculate the retention factor for A, B, C and D provided by following data.
Q: 11. Consider the following reaction and its AG-AGo at 25.00 °C. Wate eb. Calculate the equilibrium…
A:
Q: The salt bridge is essential in maintaining the electrical neutrality in a Daniel cell. In the absen…
A:
Q: did you get the ppm Cu in the first table?
A: The relation between ppm and molarity M is as follows : ppm = M × MM ×…
Q: What is the expected product for the following reaction? H3C Me. Me Product Me Me
A:
Q: Which one of the following salts, when dissolved in water, produces the solution with pH less then…
A: When a salt is made up of strong acid and weak base, then acid remains excess after…
Q: 28. For the following reaction, all of the reactants and products are in their standard…
A:
Q: classify each chemical compound listed in the table below. type of compound (check all that apply)…
A:
Q: helium-filled weather balloon has a volume of 506 L at 19.9°C and 758 mmHg. It is released and rises…
A: Given : Initial volume = 506 L Initial temperature = 19.9 °C = 292.9 K Initial pressure = 758 mm…
Q: The number of 13C NMR signals for this compound is 7. 8. 10 11 12 13 14 15 16 a. 1 17 18 19 b. 2 20…
A:
Q: 20. How many grams of Ca metal are produced by the electrolysis of the molten CaBr2 using a current…
A: 20.
Q: 1. What are the chemical hazards of both aniline and acetic anhydride?
A: there are various chemical hazards with different chemicals. Some acute disease also happens with…
Q: Label the top face. top H3C bottom
A:
Q: Queston 15 Casder the following reaction at asc if ais=0.0029 atm, Pers=027atm, What is the the mhe…
A:
Q: Question 17 For the following coordinmtion complu{Fe (C.0.XH,0),3' name Determine the geometry Gire…
A:
Q: Aldehydes and ketones that have a hydrogen atom at the alpha position can be converted into a…
A: To tell the source of stability of the carbanion formed.
Q: 6. What is the coefficient of the species that will be in the greatest amount if there are 12.5…
A: Given, 12.5 mol of sodium oxalate reacted with 12.5 moles of potassium permanganate.
Q: b) Complex II links the citric acid cycle and oxidative phosphorylation. Table 1: Biochemical…
A: In the redox reaction, succinate is undergoing oxidation and ubiquinone is undergoing reduction.
Q: 3. Consider the elementary or single step reaction, A → B, which we will depict using the following…
A: In the given reaction, A is the only reactant. Since this is an elementary single-step reaction…
Q: Can you please indicate the ion flow for the image I posted below
A: Here we are required to find the direction of flow of electron in the electrochemical cell
Q: lentify the peptide bond in the following
A: A peptide bond is an amide-type bond between two amino acids. An amino acid contains amino and…
Q: (f) H С —F: (c) 0-Sn-O :O: (d) F F (g) F F : : .. B :F: :F : (e) H-O=F:
A: Correct Lewis structure of given species are shown below
Q: The IUPAC name of tert-butyl group is: 1,1-dimethylethyl 1,1-dimethylethane 1,2-dimethylethyl…
A: The IUPAC name of tert-butyl group is:
Q: A metal cube having a mass of a112 kg is dropped into a graduated cylinder containing 0030 Laf…
A:
Q: Consider a collection of N, rubidium-87 (rubidium is atomic number 37) atoms confined inside a cube…
A: Bose-Einstein condensation is a matter of state in which most of the particles (mainly Bosons)…
Q: 12 12. What occurs during the electrolysis of a molten salt? A. Electricity is produced by a…
A:
Q: The heat capacity of a diatomic gas fits the expression Cp = a + BT + yT² + ST³ . The values of the…
A:
Q: 22. Consider an electrochemical cell based on the reaction: 2H+ (aq) + Sn (s) → Sn2+ (aq) + H2 (g)…
A: Given: 2H+ (aq) + Sn (s) → Sn2+ (aq) + H2 (g) Which of the following actions…
Q: Consider the salt bridge formed E at physiological pH. Draw the m-
A: This question belongs to biomolecules chapter.
Q: For the following reaction at a5C:N, Hyl(l) + 302(3) → „HV Cakalate AG (1) oHe+ (6)ON
A: Here we are required to find the Enthalpy change and entropy change and gibbs free energy of the…
Q: 28. For the following reaction, all of the reactants and products are in their standard…
A: Concentration of given species = 1.0 M E° = +0.60 V
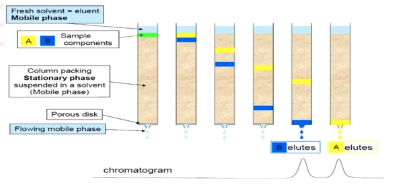
If the stationary phase used in the given setup is silica, which is more polar between A or B?
Step by step
Solved in 2 steps

- An Ostwald viscometer was used to determine the time of flow of syrup at 20 °C. Data for Water:Time of flow: 0.23 secondsDensity at 20 °C: 0.998 g/cm³Viscosity: 0.0101 poiseData for Syrup:Time of flow: 1.42 secondsDensity at 20 °C: 1.153 g/cm³ 1. What is the fluidity of water? 2. From the computed viscosity of syrup, what will be its kinematic viscosity? 3. From the computed viscosity of syrup, what will be its fluidity? 4. What is the viscosity of syrup? 5. What is the kinematic viscosity of water?Separation of the single phase solid from single phase liquid line region is done byWhat effect will the following have on plate height (H). Make sure to explain your full and complete reasoning. a. Increasing stationary phase thickness
- In a 1:1 molar ratio of SDS (sodium dodecyl sulfate) - betaine (cocoamidopropyl betaine) system, the ratio of the surfactants at the air - liquid interface near the CMC will be: A - Richer in betaine and leaner in SDS B - same as in the bulk solution C-Both will show negative surface excess because of synergy in micelle formation D -Richer in SDS and leaner in betaine Please answer very soon will give rating surelyA researcher wants to calculate the flux of palm oil (PO) in a simulated body fluid (SBF). To perform the experiment, he designed a 30 cm capillary tube. At the very beginning of the tube, he designed a partition with a separator as shown in the figure below. He filled the partition with a stock solution prepared by solving 10 g of PO in a liter of SBF and the capillary tube with pure SBF. Then he removed the separator. The viscosity of the BSF is 3.6x10-3 Pa.s and molecular weight of PO is 850.92.a. Calculate the flux of PO in kg mol/s.m2 at steady state. (Use Polson equation for diffusivity.) (Assume the temperature is maintained at 37oC during whole experiment.)b. Concentration of PO at the half of the tube.An Ostwald viscometer was used to determine the time of flow of syrup at 20 °C. Data for Water:Time of flow: 0.23 secondsDensity at 20 °C: 0.998 g/cm³Viscosity: 0.0101 poiseData for Syrup:Time of flow: 1.42 secondsDensity at 20 °C: 1.153 g/cm³ Input all numerical answers to the 3rd decimal place 1. What is the viscosity of syrup? * 2. From the computed viscosity of syrup, what will be its fluidity? 3. What is the kinematic viscosity of water? 4. From the computed viscosity of syrup, what will be its kinematic viscosity? 5. What is the fluidity of water?
- In a 1:1 molar ratio of SDS (sodium dodecyl sulfate) - betaine (cocoamidopropyl betaine) system, the ratio of the surfactants at the air - liquid interface near the CMC will be: A - Richer in betaine and leaner in SDS B - same as in the bulk solution C-Both will show negative surface excess because of synergy in micelle formation D -Richer in SDS and leaner in betaineDiluted skim milk with a protein concentration of 4.3 g/L is to undergoDEF microfiltration. Experiments have been performed using a cellulose-acetate membrane with an average pore diameter of 0.45 µm and AM = 17.3 cm2. For Stage 1 operation at a constant-permeate rate of 15 mL/minute, pressure drop across the cake and membrane increases from 0.3 psi to an upper limit of 20 psi in 400 seconds. The permeate viscosity is 1 cP. If continued in a second stage at constant ∆P at the upper limit until the permeate rate drops to 5 mL/minute, estimate the additional time of operation.Prepare plots of permeate volume in mL and permeate flux in mL/cm2-minute as functions of time.An ostwald viscometer was used to determine the time of flow of syrup at 20°C Data of water: Time of flow: 0.23 sec Density at 20°C: 0.998 g/cm3 Viscosity: 0.0101 poise Data of Syrup: Time of flow: 1.42 sec Density at 20°C: 1.153 g/cm3 1. What is the fluidity of water? 2. What is the Viscosity of syrup? 3. From the computed Viscosity of syrup, what will be its kinematic Viscosity? Show complete solution.
- What are the physical states of dispersed phase and dispersion medium in foam rubber.Ascetic acid with a specific gravity of 3.25 has a kinematic viscosity equal to1.35 × 10-6 m2/s , what is its dynamic viscosity?PLEASE ANSWER THEM ALL IN ASAP FOR AN UPVOTE. I PROMISE THAT. THANKYOU. An Ostwald viscometer was used to determine the time of flow of honey at 20 °C. Data for Water:Time of flow: 0.23 secondsDensity at 20 °C: 0.998 g/cm³Viscosity: 0.0101 poise Data for Honey:Time of flow: 20.79 secondsDensity at 20 °C: 1.426 g/cm³ a.) What is the viscosity of honey? b.)From the computed viscosity of honey, what will be its kinematic viscosity? c.) From the computed viscosity of honey, what will be its fluidity?



