The temperature at state A is 20.0°C, that is 293 K. During the last test, you have found the temperature at state D is 73.0 K and n = 164 moles for this monatomic ideal gas. What is the change in thermal energy for process A to D, in MJ (MegaJoules)? Your answer needs to have 2 significant figures, including the negative sign in your answer if needed. Do not include the positive sign if the answer is positive. No unit is needed in your answer, it is already given in the question statement. P(atm) 3 B V (m) I 2 3 45
The temperature at state A is 20.0°C, that is 293 K. During the last test, you have found the temperature at state D is 73.0 K and n = 164 moles for this monatomic ideal gas. What is the change in thermal energy for process A to D, in MJ (MegaJoules)? Your answer needs to have 2 significant figures, including the negative sign in your answer if needed. Do not include the positive sign if the answer is positive. No unit is needed in your answer, it is already given in the question statement. P(atm) 3 B V (m) I 2 3 45
Chapter3: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 31P: A dilute gas at a pressure of 2.0 atm and a volume of 4.0 L is taken through the following...
Related questions
Question
100%
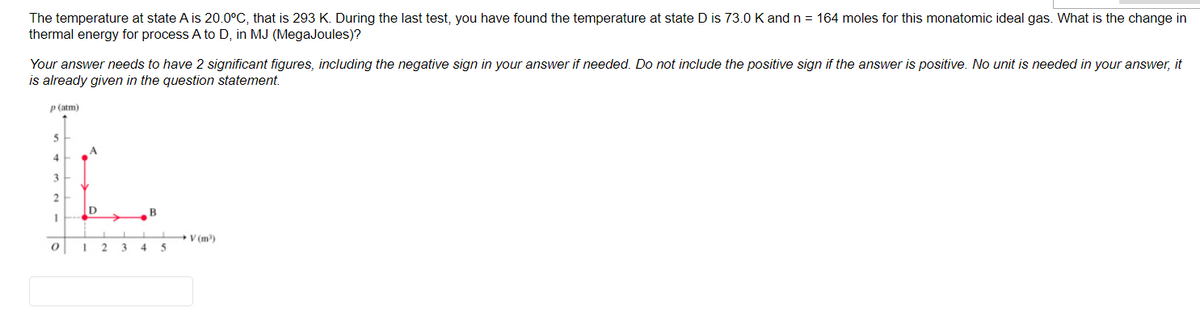
Transcribed Image Text:The temperature at state A is 20.0°C, that is 293 K. During the last test, you have found the temperature at state D is 73.0 K and n = 164 moles for this monatomic ideal gas. What is the change in
thermal energy for process A to D, in MJ (MegaJoules)?
Your answer needs to have 2 significant figures, including the negative sign in your answer if needed. Do not include the positive sign if the answer is positive. No unit is needed in your answer, it
is already given in the question statement.
P (atm)
4
3
ID
B
+V (m)
4 5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

