Q: The molecular weight for His is 155.1546. At what mass would you observe the positive ion peak for…
A: Electrospray ionization (ESI) is a method for producing ions for mass spectrometry by delivering a…
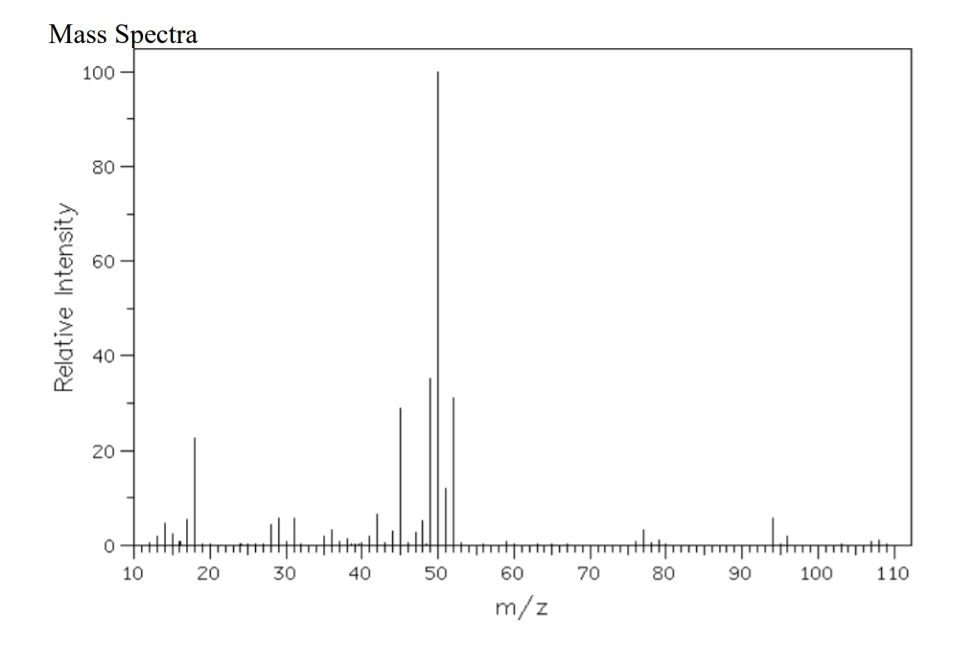
Q: Propose o structure for the compound having the mass spectrum shown below. m/a Rete tty
A: Now after seeing the given graph and the peaks of it we observe that.
Q: c. The following figure shows the mass spectrum of a hydrocarbon. i. Propose a molecular formula for…
A: The mass spectrum given for a hydrocarbon has following fragmentation peaks: m/z: 72, 57, 43, 29
Q: The picture shows the given compound that was analyzed through mass spectra where notable peaks were…
A: The mass spectrum is used for identification of molecular ion.
Q: which significant information can be provide by the mass spectra? I. identity of the compounds II.…
A: What significant information can be provided by mass spectra?
Q: The mass spectrum of 2-methyl-2-pentanol is shown below. Identify the fragments.
A: We have to identify the fragments of 2-methyl- 2- pentanol in mass spectrum.
Q: Which compound correlates to the following mass spectrum? 100 20 50 75 100 125 150 m/z HO Br Br…
A: A mass spectrum is an important tool for the determination of the mass of a compound. A mass…
Q: A mass spectrum shows significant peaks at m>z = 87, 115, 140, and 143. Which of the following…
A: The given compounds are,
Q: Elemental Analysis Results: C(74.09%) H(7.11%) O(18.80%). The following mas spectra belong to this…
A: The mass to charge ratio of ion is determine by mass spectroscopy.
Q: 1. What is the molecular ion peak in a mass spec represent? 2. What is the base peak in the mass…
A: In IR spectroscopy, two type of vibration exhibited by the molecules.
Q: The M+ peak for an alcohol in the mass spectrum is 60. Find the structure of the alcohol.
A: The answer is either Propan-1-ol (or) Propan-2-ol.
Q: Identify the main peaks for a mass spectrum of the compound in the attachment
A: The given mass spectrum of 2-chloropropane is :
Q: How could you distinguish the mass spectrum of 2,2-dimethylpropane from those of pentane and…
A: Given distinguish the mass spectrum of 2,2-dimethylpropane Vs pentane and 2-methylbutane
Q: Assign each mass spectrum to the molecule on the list. Write down the molecule that corresponds to…
A: To determine the mass spectrum, the base or molecular ion peak needs to be considered. Check the…
Q: The following figure shows the mass spectrum of a hydrocarbon. i. Propose a molecular formula for…
A:
Q: H3 C-O H.
A: An m/z ratio represents the mass to charge ratio. It can be calculated by the summation of the mass…
Q: The mass spectrum of an alcohol will usually contain the following peak: O M+18 O M-16 O M-18 O M-15
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: The mass spectrum of an unknown compound has a molecular ion peak with a relative intensity of…
A:
Q: Assign each mass spectrum to the molecule on the list. Write down the molecule that corresponds to…
A: A typical fragmentation of the molecular ion of a carbonyl compound having at least one gamma…
Q: Answer the following questions based on the mass spectrum shown below: a. What is the molar mass of…
A: a. The mass spectrum of the compound shows two peaks M+ and M+2, of almost equal intensities. The…
Q: Mass spectrometry is often used to both identify and quantify compounds. What is different between…
A: Mass spectrum is the technique used in analytical chemistry for the separation of ions based on the…
Q: Analyze the spectrum and include identification of the different fragments available based on peaks…
A: To analyze the given spectrum:
Q: In mass spectrometry, the molecular ion peak occurs at O A. the largest m/z value. O B. the smallest…
A: Here, we have to find where the molecular ion peak occurs in mass spectrometry.
Q: In the space below sketch the Mass Spectrum of the compound circled above. Show up to 3 possible…
A:
Q: Identify the base peak, the molecular ion, and C13 isotope in the mass spectrum shown below
A: Following are the base peak and molecular ion peak and C13 isotope in the given mass spectra plot.
Q: Question 4 Which significant information given below CANNOT be derived from mass spectra? molecular…
A: Mass spectra determine the masses of particles and of molecules, and to elucidate the chemical…
Q: draw the structure of ıons responsible for the peaks in mass spectrum in BENZOİN
A: Given Compound Benzoin Peaks in mass Spectrum = To be determined
Q: Q.1. Explain why, even though the average atomic mass of silver is 107.9amu, no peak at 108 amu is…
A: Mass spectroscopy is an analytical technique used to determine the mass of compounds. This technique…
Q: A mass spectrum shows significant peaks at m/z = 87, 115, 140, and 143. Which of the following…
A: The IUPAC name of the given compound is
Q: 42) Determine the mass of the likely fragment ion that will be observed in the mass spectrum for…
A: The given compound is a secondary amine and it undergoes alpha cleavage in mass spectrometer. Mass…
Q: Define process of Identifying fragments in the mass spectrum of hexane ?
A: The mass spectrum is obtained as m/z versus the relative intensity. The m/z value is the mass to…
Q: Which of the following is less likely to observe in the mass spectrum of the following molecule? OH
A: The given molecule is : 2-Methyl-butan-2-ol. Mass peak vaule which is not shown in the Mass…
Q: Please refer to the table below Name Chemical Formula Molecular Weight (CRC Handbook of Chemistry…
A: An unknown sample is run on a mass spectrometer and produced a mass spectrum with molecular ion, M+…
Q: Describe, using exact masses, the molecular ion region in the mass spectrum of the following…
A:
Q: What molecular ions will be present in a mass spectrum of 2-chloropropane,(CH3)2CHCl ?
A: Mass spectrometry is one of the important analytical technique which is helpful in structure…
Q: A compound gives a mass spectrum with essentially only three peaks at m/z = 77 (40%), 112 (100%),…
A: The given information is a compound gives a mass spectrum with peaks at m/z = 77 (40%), 112 (100%),…
Q: A B Which compound, if any, can be distinguished from the others by the molecular ions in their mass…
A:
Q: The compound in the picture was determined through mass spectra. Notable peaks were observed. What…
A: Mass spectroscopy measures the mass to charge ratio of ions in a compound. The m/z ratio of the two…
Q: Label the major peaks in each spectrum and propose a structure for the compound.
A:
Q: Using the rule of 13, what is a possible formula for M=120 (100%) on a mass spectrum? C9H12 C5H1NCI…
A:
Q: Which of the labeled ions in the mass spectrum is least stable? (a) Peak II (b) Рeak I (c) Peak IV…
A:
Q: Answer the following questions based on the mass spectrum shown below. a. What is the molar mass of…
A: In mass spectrum the peak at very right side is generally represents mass of the molecule ion (M+),…
Q: Which of the following is most likely to produce a significant El fragment at m/z = 57 in its mass…
A:
Q: Explain what can be determined from the following mass spectrum. M* = 123
A: From the mass spectrum we can determine the structure of unknown compound via molecular weight. It…
Q: Butane, C4H10, would have which of the following fragments in its mass spectrum? m/z | 15 II 29 III…
A: Mass spectrometry In this fragmentation is used . The fragmentation is the dissociation of the…
Q: a) Calculate the rule of 13. b) Determine the molecular formula of the compound using the mass…
A: a). The rule of 13 means a base formula is made and the molecular mass is divided by 13. The number…
Q: Is the compound giving rise to the mass spectrum shown here more likely to contain sulfur, bromine,…
A:
Q: e compound that gave this mass spectrum probably contains: itrogen b) bromine c) chlorine d) sulfur…
A: As M+ and (M+)+2 ratio is 3:1 it must be chlorine.
Step by step
Solved in 2 steps with 1 images

- Sample IdentificationCodeConcentration of M%TA=2-log(%T)Q50004.00 x 10-417.90.75Q50013.20 x 10-425.00.6Q50022.40 x 10-435.70.46Q50031.60 x 10-450.20.3Q50048.000 x 10-570.80.15SampleIdentificationCode%TA=2-log(%T)AMQ0210150143.70.359518560.360.000192Q0210150244.10.355561410.360.00018Q0210150343.80.358525890.360.00017Q0210150444.10.355561410.360.00018Q0210150543.80.358525890.360.00017What was their percent error?43%Does Batch 021015 meet legal requirements?No, because it is not between 2.85 * 10(4) and 3.15 * 10(4)Well #DropsBluedye1234567891012345678910Drops 9Distilled water876543210Concne 0.26tration0.52Test Tube #0.781.041.3Solutions3Concentration (M)2.082.32.6Concentration (ppm)1:1 dilution11.82Starting Dilution21.562:1 dilution0A.Zero standard0Was your calibration curve as linear as you expected?B.Did you experience any “drift†of the resistance readings?C.What is the equation of your best-fit line?D.What commercial drink did you analyze?E.Assuming…What is the difference between determinate and indeterminate errors? Cite at least two examples of each that can be found in your medical field. Can we avoid these errors? Why? *What are the 5 important characteristics of spectrophotometric methods
- Calculate the following exercise with correct sig figs: ((2.9365mL - 3.00mL)/3.00mL)x100%Please create a caption for this table. Solution NaCl Conc. (%) Osmolality (mOsm) % transmittance Absorbance % hemolysis % crenation C distilled 0 0 0.001029 4.987584625 100 0.03354 1 0.177179111 54.61 0.001551 4.809388202 96.42720001 0.05837 2 0.297126222 91.58 0.01012 3.994819487 80.09527231 0.08444 3 0.442542222 136.4 3.849 1.414652089 28.3634704 0.134 4 0.590164444 181.9 64.8 0.188424994 3.777880643 0.2125 5 0.74752 230.4 95.64 0.019360433 0.388172513 0.3368 6 0.89644 276.3 99.56 0.001915112 0.038397585 0.5336 7 1.095648889 337.7 99.98 8.68676E-05 0.001741676 0.9834 8 1.336711111 412 100 0 0 2.1 9 1.755568889 541.1 100 0 0 7.9 10 2.674395556 824.3 100 0 0 57.83 11 4.490211111 1384 100 0 0 99.72Describe how to use a spectrophotometer
- At 36.0 °C the density of water is 0.9936870 g/cm3.At 37.0 °C the density of water is 0.9933390 g/cm3.Use linear interpolation to calculate the density (D) of water at 36.2 °C, to the nearest 10-7g/cm3. ANSWER IS: D = 0.9936174 g/cm^3As used in the definition of a sampling distribution, "all possible random samples refers toDescribe the origin of shot noise in a spectrophotometer. How does the relativeuncertainty vary with concentration if shot noise is the major noise source?
- What is the relative error in the determination of a density for which theaccepted value is 0.945 g*cm3 and the experimental value is 0.948 g*cm3 ?Apply the Q test to the following data sets to determine whether the outlying result should be retained or rejected at the 95% confidence level. a. 85.10, 84.62, 84.70b. 85.10, 84.62, 84.65, 84.70Which part of the instrument perform the given function introduction of a micro amount of sample converts sample components into gaseous ions orts the ions according to m/z converts m/z values to electrical signals converts signal/results into a mass spectrum A. mass analyzer B. Vacuum system C. Ion source D. Processor E. inlet system F. detectors



