Titration of the I2 produced from 0.1187 g of primary- standard KIO3 (Molar Mass= 214.0o g/mol) required 26.78 mL of Na2S203. 103 + 51 + 6H 312 + 3H2O : 12 + 2S2032- 21 - + S,062- What is the molarity of Na2S203?
Q: Write the formula of the conjugate acid of the Brønsted-Lowry base caffeine, CSH10N4O2
A: Given that, Bronsted -Lowry base = C8H10N4O2 Conjugate acid = ?
Q: A factory has a solid copper sphere that ne s to be drawn into a wire. The mass of the copper s, nee...
A: According to the mass conservation law when the copper sphere is converted into copper wire the mass...
Q: What is the pH of a solution that is 0.071 M LiCN at 25°C?
A: A solution is made up of 0.071 M LiCN at 25 ◦ C. Here, we have to calculate the pH of the solution. ...
Q: predict the products, blanace the chemical equation and indicate if the reaction is a precipitation ...
A: Predict the products, balance the chemical equation and indicate if the reaction is a precipitation ...
Q: TQhB66q?1oB O CHEMICAL REACTIONS Percent yield of chemical reactions OOOC 15 quid octane (CH,(CH,),C...
A: Given : We have to calculate the percentage yield.
Q: Which mechanism is consistent with the reaction below? (CH3)3CBr+OH-→(CH3)3COH+Br- 1. (CH3)3CBr+(...
A: Carbocation can be defined as the species in which carbon atom bear positive charge. It can be teria...
Q: Why is it necessary to acidify the mixture obtained after reaction of Grignard reagent with both the...
A: Grignard reaction is an organometallic chemical reaction alkyl, aryl, vinyl or aryl magnesium halide...
Q: Give an IUPAC name for the compound below. Accepted names for branched alkyl groups are isopropyl, i...
A: The following steps are required for IUPAC naming 1->first of all identify the longest carbon cha...
Q: C For the mechanism step below, draw curved arrows to show electron reorganization. Arrow-pushing In...
A:
Q: CIO2 + A13+. CIO3" + AI Water appears in the balanced equation as a (reactant, product, neither) wit...
A: ClO2 + Al+3 -----> ClO3- + Al
Q: CI AICI3
A: generation of electrophile from alkyl halide by lewis acid carbocation rearrangement nucleophilic-...
Q: What is the change in internal energy if the heat absorbed by the system is 97 J and the work being ...
A: Using first law of thermodynamics internal energy = heat + work
Q: When the following equation is balanced properly under basic conditions, what are the coefficients o...
A: Answer: Given reaction is a redox reaction, therefore we will use ion-electron method to balance thi...
Q: Give a clear handwritten answer...
A: Acidic strength of molecules can be compared by resonance stabilization of their conjugate bases. M...
Q: Classify each of the following compounds as an alkane, alkene, alkyne, alcohol, aldehyde, amine, and...
A:
Q: An atom gained an electron, thus, becoming negatively charged. If it was then attracted to a polar m...
A: The attraction forces between the different molecules are known as intermolecular forces.
Q: An acetic acid buffer contains 0.6 M CH3COOH and 0.6 M CH3COONA. (CH3COOH has a K, 1.76 x 105.) What...
A: Given- Concentration of CH3COOH= 0.6M Concentration of CH3COONa= 0.6M Ka of CH3COOH=1.76 ×10 -5 Mole...
Q: of the following is expected to be the most reactive with Br2 in thne presence of Fe? „CF3 OH b. с. ...
A: As it is bromination of aromatic ring. It undergoes Aromatic electrophilic substitution reaction. S...
Q: State why the reaction between acetone and 2-pentanone would not be a good method to prepare 2-methy...
A:
Q: 13. Calculate AH for the reaction NO + Og O NOZ18) Given the following information: As part of your ...
A: ∆H of a reaction depends on Stoichiometry of chemical equation If the equation is reversed then si...
Q: Determine the direction of bond polarity for the O-H bond by identifying the ô+/õ- ends: Wto ō-ends:...
A: Electron cloud in a covalent bond shifts more towards more electronegative element and thereby attai...
Q: When the following skeletal equation is balanced under acidic conditions, what are the coefficients ...
A: First we need to balance the given redox reaction in acidic medium , then we can determine where wat...
Q: B3. Write a detailed mechanism for the following Knorr Synthesis 1) (BUO-NO (0.5 equiv) , HCI A .CO2...
A: Preparation of pyrrole compound by using beta amino keto-ester and beta-keto ester is called Knorr s...
Q: How many monochloro substitution products are produced when the alkanes below are chlorinated? Consi...
A: The total types of C-H bonds give total monochlorinated substitution product.
Q: Starting with 2-methylbutane, and using any organic or inorganic reagents necessary, outline a synth...
A: We have find out organic or inorganic reagents for outline a synthetic strategy.
Q: 3. In a solution containing H3O* ions at a concentration of 0.003 mol.L', what is the concentration ...
A: Given, [H3O+] = 0.003 mol.L-1 [OH-] = ? The formula used here are, pH = -log[H3O+] pH + pOH = 14 pOH...
Q: Requires a light source with a wavelength between 10 picometers and 10 nanometers X-ray crystallogra...
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for yo...
Q: How can we calculate the concentration (molarity) of OH- in a solution that is 0.0859 M NaOH at 25°C...
A: Given-> Molarity of NaOH = 0.0859 M
Q: We prepared a solution containing 1.0 mol.L·' HF and 1.0 mol.L-' C,H;OH. The K, of hydrofluoric acid...
A: Answer: Given data: [HF]=1.0M[[C6H5OH]=1.0M Ka(HF)=7.2×10-4M Ka(C6H5OH)=1.6×10-10M
Q: H3C C-CH3 H.
A:
Q: Show your complete solution for the ff: Calculate AS° for CaCo, (s) CaO(s) + CO, (g) b. 2H,(g) + O, ...
A:
Q: Identify the polyprotic acids. Select all H3PO4 HCI H2CO3 H2SO3 HCIO3 HNO3
A:
Q: hich has the least viscosity? O CH3CH20H HOCH2CHOHCH2OH CH3(CH2)3CH3 O CH3(CH2)4CH3 hich has the gre...
A: Since you have posted multiple questions as per the guidelines we can answer only one per session . ...
Q: Solid silver nitrate is slowly added to 75.0 mL of a 0.333 M potassium hydroxide solution until the ...
A:
Q: KBr
A: Answer: option third is the correct answer . When KBr is dissolved in water then KOH and HBr is form...
Q: How many molecules of ethylene diamine will react with Ni(H,0),**? Explain your answer.
A:
Q: The formation of magnesium bromide from Mg and Br, is represented in the particle diagram. Which of ...
A:
Q: Write a reaction mechanism for the following conversion, including reagents and/or conditions. (Non-...
A:
Q: If tripling the concentration of a single reactant Rin a multi-reactant reaction (while leaving all ...
A: Rate = k [R]x
Q: The base protonation constant K, of 1-H-imidazole (C;H,N,) is 9.0 x 10-8. Calculate the pH of a 0.57...
A: We have to predict the pH of solution.
Q: A mixture of cyclopropane gas and oxygen is used as an anesthetic. Cyclopropane contains 85.7% C and...
A: Molecular formula: In a molecular formula we describe the number of atoms of all the elements presen...
Q: A o.8024 g sample of an iron ore is dissolved in acid. The iron is then reduced to Fe2" and titrated...
A:
Q: Give the IUPAC name of the product that would form when the following cycloalkene undergoes catalyti...
A: Alkene on reduction gives Alkane
Q: O Problem: Determine the blood sugar result for a patient from a Random Blood Sugar Test
A: Ans. Random Blood Sugar Test: This measures your blood sugar at the time you’re tested. You can take...
Q: What is the molecular geometry and bond angle of letters A,B, and C.
A: The compound given is,
Q: Calculate the pH of a solution that contains 0.10 M ammonium hydrochloride and 0.010 M HCl
A: GIVEN:- Calculate the pH of a solution that contains 0.10 M ammonium hydrochloride and 0.010 M HCl [...
Q: 2. Consider the five esters below with formula C.H 20:: (a) (b) (e) (d) (c) O Which is/are mad e fro...
A:
Q: A gas has a pressure of 645 Torr at 128 °C. What is the temperature in Celsius if the pressure incre...
A:
Q: Number of Formal Charge Electron-Group Geometry Molecular Geometry Resonance Molecule valence electr...
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for yo...
Q: CH;OH H,SO4 Salicylic Acid
A:
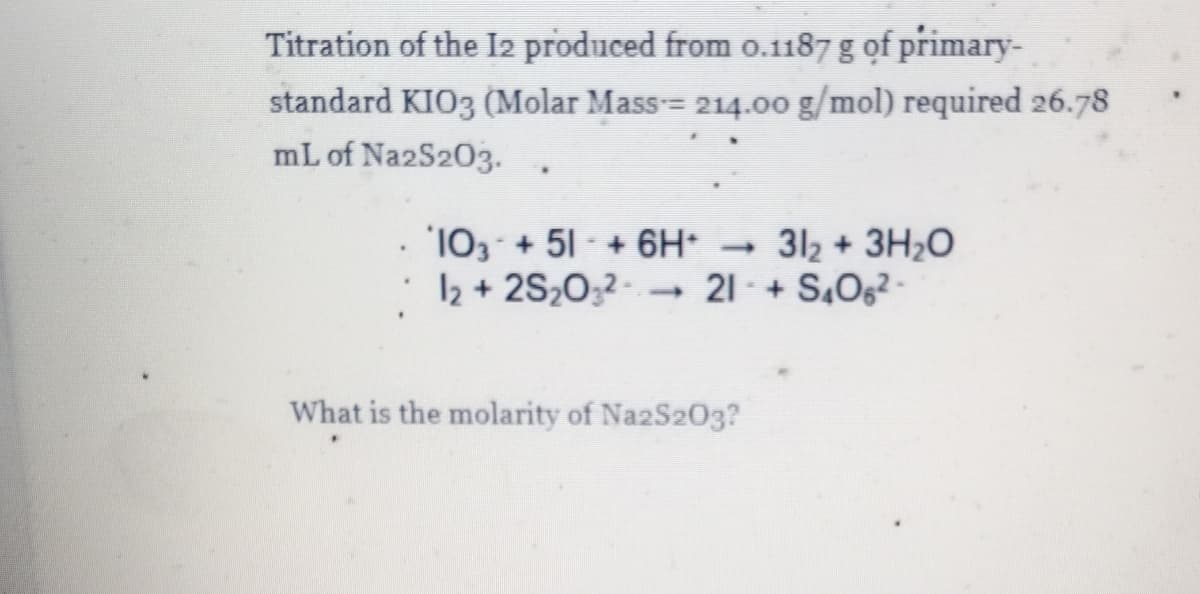
Step by step
Solved in 2 steps with 1 images

- Titration of I2 produced from 0.1238 g of primary-standard-grade KIO3 required 41.27 mL of sodium thiosulfate. Calculate the concentration of the Na2S2O3. IO3- + 5 I- + 6 H+ → 3 I2 + 3 H2O I2 + 2 S2O32- → 2 I- + S4O62-Titration of the I2 produced from 0.1142g of primary standard KIO3 required 27.95 mL of sodium thiosulfate. Calculate the molar concentration of the KMno4 solution.A 25.0025.00 mL solution of 0.088300.08830 M NaINaI is titrated with 0.051500.05150 M AgNO3AgNO3. Calculate pAg+pAg+ following the addition of the given volumes of AgNO3AgNO3. The KspKsp of AgIAgI is 8.3×10−178.3×10−17. 37.40 mLpAg+=37.40 mLpAg+= VepAg+=VepAg+= 47.30 mLpAg+=47.30 mLpAg+=
- A 0.4482 g sample of impure NaCN is titrated with 0.1018 M AgNO3 requiring 39.68 mL to reach the end point. Report the purity of the sample as %w/w NaCN. a. 61.13% b. 63.13% c. 62.31% d. 66.25%Titration of the I2 produced from .1045g of primary standard KIO3 required 30.72ml of sodium thiosulfateCalculate the concentration of the Na2S2O3What mass of Ba(OH)2 is present in a sample if it is titrated to its equivalence point with 44.20 mL of 0.1000 N H2SO4? Note: Present complete solutions for the following problem. Express your final answers up to two (2) decimal places.
- You have performed an iodimetric titration using a commercial vitamin C tablet. Based on the following information below, calculate the %(w/w) of vitamin C(MM=176.16 g/mol) in the tablet: Mass of tablet dissolved in 250.0 mL: 5.422 g Aliquot volume of sample titrated: 25.00 mL Concentration of KIO3: 0.023 M Final burrette volume: 41.31 mL Initial burrette volume: 8.89 mL Blank volume: 0.14 mL2. A 0.5131-g sample containing KBr is dissolved in 50 mL of distilled water. Titrating with 0.04614 M AgNO3 requires 25.13 mL to reach the Mohr end point. A blank titration requires 0.65 mL to reach the same endpoint. Report the %w/w KBr in the sample.The titration of 0.2121 g of pure Na2C2O4 (134.00 g/mol) required 43.31 mL of KMnO4. What is the molar concentration of the KMnO4 solution? Given that the chemical reaction is 2MnO4- + 5C2O42- + 16H+ -----> 2Mn2+ + 10CO2 + 8H2O Provide a detailed solution.
- A 0.5131-g sample containing KBr is dissolved in 50 mL distilled water. Titrating with 0.04614 M AgNO3 requires 25.13 mL to reach the Mohr endpoint. A blank titration requires 0.65 mL to reach the same endpoint. Report the % (w/w) KBr in the sample. [Ans. 26.19 % (w /w)]A Fajans titration of a 0.7908-g sample required 45.32 mL of 0.1046 M AgNO3 . Express the results of this analysis in terms of the percentage of BaCl2 * H2O. (Use a MW value in 4 decimal places)What volume (V) of 0.100M (moles) KMnO4 would be required to Titrate 0.23g of K2{Cu(Cu2O4)2} * H20?

