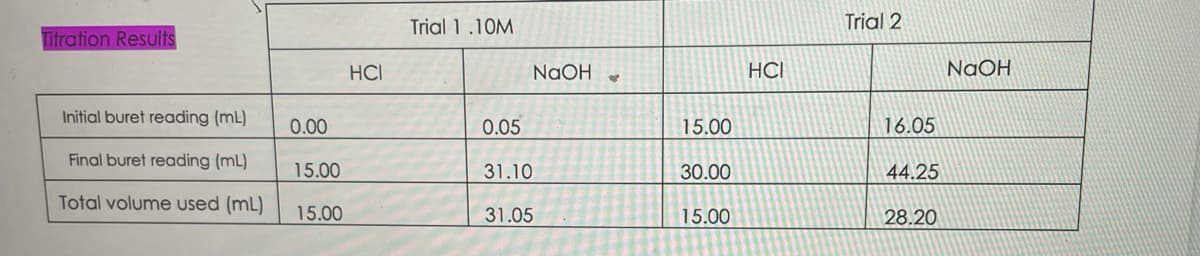
Titration Results Initial buret reading (ml) Final buret reading (ml) Total volume used (mL) 0.00 15.00 15.00 HCI Trial 1.10M 0.05 31.10 31.05 NaOH SP 15.00 30.00 15.00 HCI Trial 2 16.05 44.25 28.20 NaOH
Q: Which of the following compounds is an carboxylic acid? A) B) CH3C-OH CH3CH₂CH C) D) CH3CH₂C-O-CH3…
A:
Q: Consider the reaction of a 20.0 mL of 0.220 M CsHsNHCI (Ka = 5.9 x 10-) with 12.0 mL of 0.215 M…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: Which of the following processes yields a AS = - ? O2 NO(g) + O2(g) (11> 2 NO2(g) NaCIO3(s)---> Na…
A: If ∆ng for a reaction is + then ∆S for this reaction is also + If ∆ng for a reaction is - then ∆S…
Q: The following equation is used to prepare gas M. 2A(g) + 3X(g) ⇌ 2M(g) A mixture of A and X was…
A:
Q: Compared to hydrogen , 1/1H, the element Helium , 4/2 Has : A - more mass and is larger in size B-…
A: Compared to 11H , the element helium 2He4 has :
Q: Which of the following unbalanced reactions will proceed to completion: CuCl₂ (aq) + NaOH (aq) -->…
A:
Q: Name the following organic compound: da CH₂ CH₂ H3C CH₂ OH 2-oxo-5-pentanol O 4-oxo-1-pentanol O…
A:
Q: Two possible Lewis structures for the cyanate ion are shown below, with formal charges (FC)…
A: All the related discussions have been done in the following step.
Q: Determine the osmotic pressure in atm at 45.8 °C of an aqueous solution that is 0.502 M NaNO3. (R =…
A: Given :- concentration of NaNO3 solution = 0.502 M R = 0.0821 atm.L / K.mol T = 45.8°C To…
Q: Blood alcohol content is a measure of alcohol in the blood as a percentage. It is calculated in…
A: A BAC of 0.08% means that 0.08 g of alcohol is present in 100 mL of blood So the required…
Q: Which element has the following configuration: [Xe]6s²4f¹12 Enter the chemical symbol for the…
A: Part C Given :- electron configuration = [Xe]6s24f11 To determine : chemical symbol for this…
Q: 1 24 27 Fe: Select the spectator ions in the following equation: CaCl2 (aq) + (NH4)2SO4 (aq) -->…
A:
Q: Which of the following five compounds produced the IR spectrum below? LOH NH₂ OH OH you A 4000 *****…
A: Given is IR spectra
Q: the sodium acetate - acetic acid buffer keep the pH: O a. -4.8 b. =8.9 O C. -1.8 O d. = 7.7
A: pH of an acidic buffer is given by : pH = pKa + log ([salt]/[acid])
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. OH DMSO, (COCI)2 Et3N,…
A:
Q: The following plot shows the pH curves for the titrations of various acids by 0.10 M NaOH (all of…
A:
Q: What are the amino acids that can give a positive Ceric ammonium nitrate test? Oxidation number of…
A: We have to tell the name of the amino acids that give the positive Ceric ammonium nitrate test. The…
Q: If we have a container with a partial pressure for oxygen of 2.64, a partial pressure for nitrogen…
A: Given :- Partial pressure of oxygen = 2.64 Partial pressure of Nitrogen = 2.51 Partial pressure of…
Q: Painly What product forms at the cathode during the electrolysis of molten KBr ? K+ (1) Br-(1)…
A: Electrolysis Process in which electric energy converted into chemical energy Cathode undergoes…
Q: Only part c and d please
A: Part c) Is zinc more reactive, or less reactive than iron? Explain your choice. Part d) A…
Q: What is the answer to this problem? At what pressure (in atm) would 230 g of disulfur monoxide gas…
A:
Q: 5. What is the molality of KBr in a solution prepared by dissolving 2.21 g of KBr in 299 g of water?…
A: Molality of KBr can be calculated as follows
Q: 1.50 mol of calcium chlorite is dissolved in enough water to produce 4.50 L of solution. K₁ = 2.9 x…
A:
Q: Given the following information, calculate the reaction velocity at saturating substrate…
A: Michaelis-Menten constant, KM = K-1 + K2K1 = 20 + 300010 = 302 sec-1 Maximum reaction velocity, Vmax…
Q: 50. Calculate the kinetic energy of an electron ejected by a photon with wavelength 2.07×102 nm from…
A: We have to predict the kinetic energy of the electron.
Q: Use the observations about each chemical reaction in the table below to decide the sign (positive or…
A: The observations given are,
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. PhMgBr 2. H3O+
A:
Q: Out of the following, which is incorrect? been pleed (2 lortrymond (b a) A buffer solution is one…
A: A buffer solution is a mixture of two solutions - a weak acid and its conjugate base. It has a…
Q: caves. Balance the following redox reactions. and identify the elements that are being reduced or…
A: Hii there, As there are multiple sub parts posted. we are answering first-three sub parts . If you…
Q: Write the complete decay equation for the given nuclide in the complete X notation. Refer to the…
A: 1) Write electron capture reaction of 75Se ---
Q: What is the pH of a buffer solution, prepared by mixing of 20 mL 0.1 M NaH₂PO4 solution and 100 mL…
A:
Q: Fill the missing reagents. 0= → in synthesis. ? alcohol MM
A:
Q: Answer the following question by matching the numbers with the descriptio Numbers may be used more…
A: 1) The number of orbitals in a 4d subshell or sublevel is
Q: A) Determine if the following changes in oxidation state are reduction or oxidation How many Changes…
A: Oxidation is an increase in the oxidation state Reduction is an decrease in the oxidation state
Q: Synthesize the o-form of amino benzoic acid from benzene.
A: Synthesis of o-form of amino benzoic acid from benzene is given below
Q: consider the titration of 100.0ml of 0.200M HONH2 by 0.100M HCl (Kb for HONH2 = 1.1x10^-8) a.…
A: NH2OH is a weak base with Kb = 1.1*10-8 Given the initial concentration of NH2OH = 0.200 M
Q: What is the mechanism of the phosphate buffer action? Write down the equations of these reactions.
A:
Q: 5. Identify the product(s) from the complete reduction of the unknown below and LiAlH4 followed by…
A: Molecular formula : C6H12O2 ( DBE = 6-(12/2)+1 = 7-6 = 1. DBE = 1 is carbonyl stretch of ester.
Q: The reaction below represents radioactive decay and has a rate constant of 0.04 sec-1. What is the…
A: Given that, rate constant k= 0.04 s-1 Reaction velocity r = ? [A] = 10 mM
Q: Use the information in the ALEKS Data tab to sort species reducing power Ni (s) choose one ✓ Ag(s)…
A: ->The specie which has high reduction potential that tends to reduce itself and oxidize other…
Q: Feedback Consider the titration of 100.0 mL of 0.8 M H₂A by 0.8 M KOH for the next three questions.…
A: Answer: This is a question based on acid-base titration of polyprotic acid (H3A) against a strong…
Q: The decomposition of N₂O5 proceeds according to the following equation: 2N2O5 (g) →→ 4NO2(g) + O₂(g)…
A:
Q: The reaction 3Ox(g) + M(g) ⇋ 4N(g) has Kc = 67.10 Calculate Kc for the reaction: 2N(g) ⇋ 0.5…
A:
Q: Calculate both the pH and pOH for each of the solutions with the following hydrogen ion or hydroxide…
A:
Q: 5. Continued H₂C-CC-H H₂C-CC-H H₂, Lindlar's cat. NBS, hv HBr. R-O-O-R Cl₂ 1 cq. HCI 1) NINH, 2)…
A: Reaction 1 : hydrogenation gives cis product. Reaction 2 : Allylic bromination. Reaction 3 : HBr…
Q: Using these metal ion/metal standard reduction potentials: Cd²+ (aq)| Cd(s): -0.38 V; Zn²+ (aq…
A:
Q: change 18h Dry nswer contains a unit symbol. Round your answer to 3 significant digits. μ ロ・ロ X 010…
A: Given, □Hfus = 4.39 kJ= 4390 J T = -112 K = 161 K Mass of CS2 = 400 g Molar mass = 76 g/mol Mole…
Q: arrange the following ionic compounds in order of decreasing amount 10 INT
A: Arrange the following ionic compounds in order of decreasing amount of energy released in lattice…
Q: Draw 4-ethyl-2,2,3,5-tetramethylnonane How many H atoms are bonded to carbon 2? Answer: and number…
A: Draw the structure and determine number of H atoms are bonded to Carbon-2 ?
Q: What is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka = 6.8 × 10⁻⁴) and 0.200 mol of NaF to…
A: In this question we have to tell the PH of the buffer solution after adding 0.04 mol of HCl.
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

- Data Table 3: Complexometric Titration of Soft Water 5 ml of water 1ml of buffer Hard Water Trial 1 Hard Water Trial 2 Hard Water Trial 3 Initial Syringe Reading (mL) 1ml 1ml 1ml Final Syringe Reading (mL) 77ml 79ml 75ml Volume of EDTA Consumed (mL) .23ml .21ml .25ml Water Hardness ppm CaCO3 (mg/L) Average ppm CaCO3 (mg/L):Data Table 3: Complexometric Titration of Soft Water 6ml of water used (1ml of buffer and 5ml of soft water) Hard Water Trial 1 Hard Water Trial 2 Hard Water Trial 3 Initial Syringe Reading (mL) 1ml 1ml 1ml Final Syringe Reading (mL) 77ml 79ml 75ml Volume of EDTA Consumed (mL) .23ml .21ml .25ml Water Hardness ppm CaCO3 (mg/L) Average ppm CaCO3 (mg/L):Part A: Standardization of a Sodium Hydroxide Solution Titration 1 Titration 2 Titration 3 Mass of 125 mL flask 45.849g 46.715g 44.953g Mass of flask and KHP 46.849g 47.745g 46.003g Initial buret reading (mL) 0.5 ml 0.5 ml 0.5 ml Final buret reading (mL) 27.8 ml 26.5 ml 26.7 ml Volume of NaOH used (mL) 45.11 ml 45.06 ml 45.14 ml Calculations Titration 1 Titration 2 Titration 3 Moles of KHP Moles of NaOH Molarity of NaOH Average Molarity of NaOH: _______________
- Molarity of titrant (NaOH): 0.4550 M HC2H3O2 (aq) + NaOH (aq) → NaC2H3O2 (aq) + H2O (l) Trial # First Second Third Fourth Initial buret reading 0.15 mL 2.43 mL 1.32 mL 0.58 mL Final buret reading 18.62 mL 20.87 mL 20.03 mL 19.14 mL Volume of titrant used 18.47 mL 18.44 mL 18.71 mL 18.56 mL 4) Calculate the molarity of the acetic acid in the vinegar solution (Show your work). use FW for moles-->grams acetic acid. Molarity acetic acid = _____________ M 5) Calculate the weight % of acetic acid in the vinegar. How does this compare with the % listed on the label (5.00%)? (For this calculation assume that density of vinegar is 1.03 g/mL and of course, show your work). Weight % = ___________ 6) If you didn’t get the same weight % of acetic acid as listed on the vinegar label (5.00 %), what are two things (be specific) that could’ve happened during the experiment that could explain the variation from the expected weight %? To do…Data Table 1: Complexometric Titration of Hard Water 6ml of water used (5ml of hard water and 1ml of buffer) Hard Water Trial 1 Hard Water Trial 2 Hard Water Trial 3 Initial Syringe Reading (mL) 1ml 1ml 1ml Final Syringe Reading (mL) 0.55ml .50ml .33ml Volume of EDTA Consumed (mL) 0.45ml .50ml .77ml Water Hardness ppm CaCO3 (mg/L) Average ppm CaCO3 (mg/L): photo shows equation that needs to be used to solveData Table 2: Complexometric Titration of Tap Water photo shows equation to find water hardness 6 ml of water used (5 ml of tap water 1ml of buffer) Hard Water Trial 1 Hard Water Trial 2 Hard Water Trial 3 Initial Syringe Reading (mL) 2ml 2ml 2ml Final Syringe Reading (mL) .61ml .69ml .58ml Volume of EDTA Consumed (mL) 139ml 131ml 142ml Water Hardness ppm CaCO3 (mg/L) Average ppm CaCO3 (mg/L):
- Data Table for Weak Acid Vol. of Titrant pH Avg. Vol. ∆pH/∆vol 0 2.881 0.25 0.622575 0.5 3.193 0.75 0.453769 1 3.420 1.25 0.32668 1.5 3.583 1.75 0.251434 2 3.709 2.25 0.20456 2.5 3.811 2.75 0.173202 3 3.897 3.25 0.150977 3.5 3.973 3.75 0.134517 4 4.040 4.25 0.12191 4.5 4.101 4.75 0.112002 5 4.157 5.25 0.104057 5.5 4.209 5.75 0.097586 6 4.258 6.25 0.092253 6.5 4.304 6.75 0.087819 7 4.348 7.25 0.084111 7.5 4.390 7.75 0.081001 8 4.431 8.25 0.078391 8.5 4.470 8.75 0.076209 9 4.508 9.25 0.074397 9.5 4.545 9.75 0.072912 10 4.582 10.25 0.071719 10.5 4.617 10.75 0.070794 11 4.653 11.25 0.070117 11.5 4.688 11.75 0.069675 12 4.723 12.25 0.06946 12.5 4.757 12.75 0.069467 13 4.792 13.25 0.069696 13.5 4.827 13.75 0.070152 14 4.862 14.25 0.070845 14.5 4.897 14.75 0.071787 15 4.933 15.25 0.072999 15.5 4.970 15.75 0.074507 16 5.007 16.25 0.076346 16.5 5.045 16.75 0.078561 17 5.085 17.25 0.081211 17.5 5.125 17.75 0.084372 18…A 50.00 (±0.02) mL portion of an HCl solution required 29.71(±0.02) mL of 0.01963(±0.0032) M Ba(OH)2 to reach an end point with bromocresol green indicator. ? of HCL = 29.71?? ? 0.01963 ???? ??(??)2 ?? ? 2 ???? ??? ???? ??(??)2/ 50.00?? = 0.02333 ? Calculate the uncertainty of the result (absolute error).Calculate the coefficient of variation for the result.Trial 1: Initial pH= 3.92mL Mass of KHP and Paper= 0.868g Mass of paper= 0.357g Mass of KHP= 0.511g Trial 2: Initial pH= 4.09ml Mass of KHP and Paper= 0.870g Mass of paper= 0.359g Mass of KHP= 0.511g 1)Use Kb, the number of moles of C8H4O4^2- at the equivalence point, and the total volume at that point to calculate the pH for each sample at the equivalence point. Compare these calculated results with the experimental results.
- How will the analyte concentration be altered if the titrant volume recorded is greater than required? cannot be determined from the information given It will be reported lesser than the actual concentration. It will be reported the same as the actual concentration. It will be reported greater than the actual concentration.A standard serum sample containing 102 meq/L chloride was analyzed by coulometric titration with silver ion. Duplicate results of 101 and 98 meq/L were obtained. (a) Calculate the mean error (b) Calculate the relative error (c) Calculate the relative accuracyThe water hardness of tap water has been determined to be 120 (= 120 mg CaCO3 per 1 L of water). After the tap water has run through the ion exchange column, titration of a 20.00 mL sample used only 0.70 mL of 0.0100 M EDTA, to reach the endpoint. Calculate the effectiveness of the ion exchange column: what is the % of the calcium, removed by the ion exchange column? Provide your answer in % and without decimal. ( Please type answer note write by hend)



