Toluene (CoH5CH3) has a vapor pressure of 0.038 atm, while benzene (CoH6) has a vapor pressure of 0.13 atm. If 0.89 moles of toluene and 0.77 moles of benzene are mixed, what is the vapor pressure of the resulting solution (assume 25°C throughout)? Report to two decimal places regardless of sig figs.
Toluene (CoH5CH3) has a vapor pressure of 0.038 atm, while benzene (CoH6) has a vapor pressure of 0.13 atm. If 0.89 moles of toluene and 0.77 moles of benzene are mixed, what is the vapor pressure of the resulting solution (assume 25°C throughout)? Report to two decimal places regardless of sig figs.
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter10: Properties Of Solutions
Section: Chapter Questions
Problem 129CP: In some regions of the southwest United States, the water is very hard. For example, in Las Cruces,...
Related questions
Question
100%
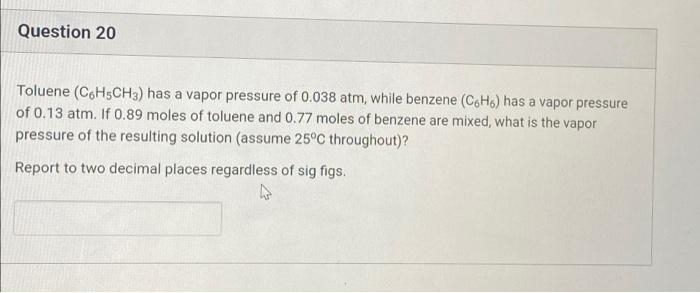
Transcribed Image Text:Question 20
Toluene (C6H5CH3) has a vapor pressure of 0.038 atm, while benzene (CoH6) has a vapor pressure
of 0.13 atm. If 0.89 moles of toluene and 0.77 moles of benzene are mixed, what is the vapor
pressure of the resulting solution (assume 25°C throughout)?
Report to two decimal places regardless of sig figs.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole