Tre 2.) Given the final product and reagents for each step in the forward synthesis of this cyclohexane-containing internal alkyne, please propose the structures for the synthetic intermediates A-E below.
Tre 2.) Given the final product and reagents for each step in the forward synthesis of this cyclohexane-containing internal alkyne, please propose the structures for the synthetic intermediates A-E below.
Chapter16: Chemistry Of Benzene: Electrophilic Aromatic Substitution
Section16.SE: Something Extra
Problem 30MP: The carbocation electrophile in a Friede1-Crafts reaction can be generated by an alternate means...
Related questions
Question
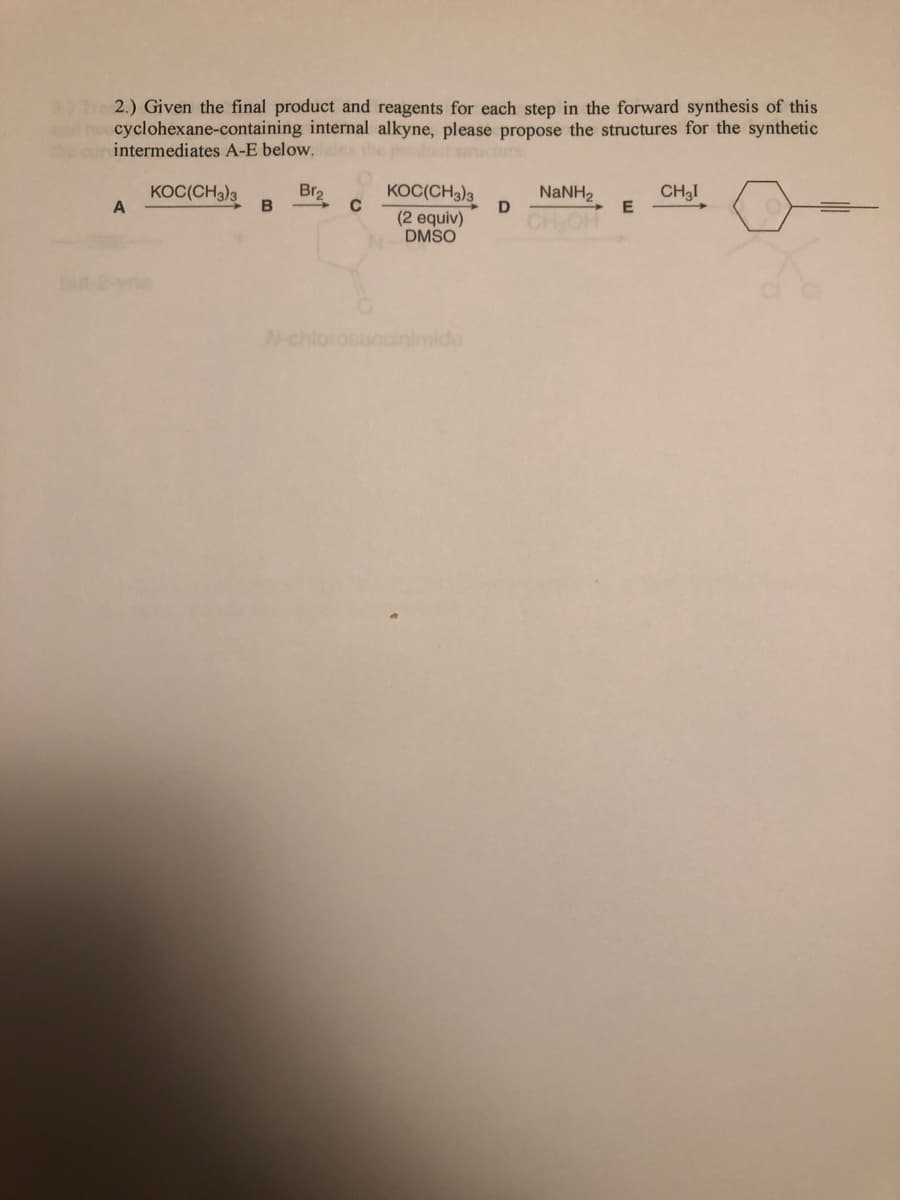
Transcribed Image Text:3) Tre 2.) Given the final product and reagents for each step in the forward synthesis of this
nd t cyclohexane-containing internal alkyne, please propose the structures for the synthetic
the cur intermediates A-E below. s
KOC(CH3)3
Br2
KOC(CH3)3
NaNH2
E
CH3I
(2 equiv)
DMSO
CHOH
but-2ayne
N-chlorosucclinimide
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

