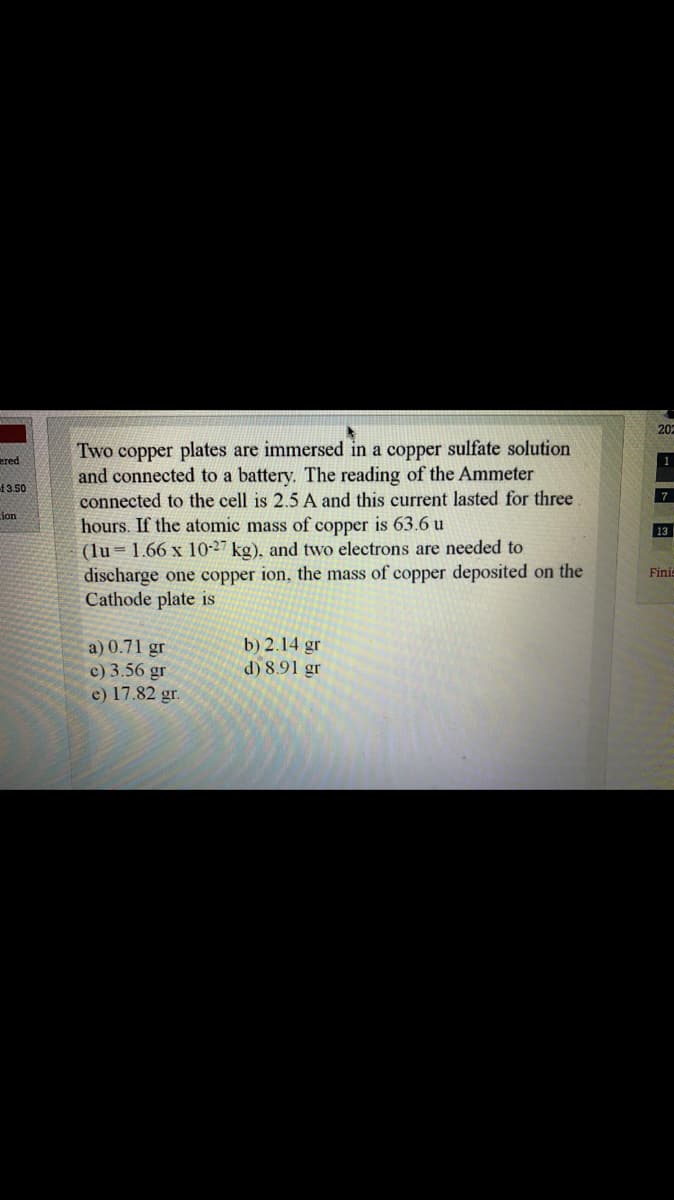
Two copper plates are immersed in a copper sulfate solution and connected to a battery. The reading of the Ammeter connected to the cell is 2.5 A and this current lasted for three hours. If the atomic mass of copper is 63.6 u (lu 1.66 x 10-27 kg), and two electrons are needed to discharge one copper ion, the mass of copper deposited on the Cathode plate is %3D a) 0.71 gr c) 3.56 gr c) 17.82 gr. b) 2.14 gr d) 8.91 gr
Two copper plates are immersed in a copper sulfate solution and connected to a battery. The reading of the Ammeter connected to the cell is 2.5 A and this current lasted for three hours. If the atomic mass of copper is 63.6 u (lu 1.66 x 10-27 kg), and two electrons are needed to discharge one copper ion, the mass of copper deposited on the Cathode plate is %3D a) 0.71 gr c) 3.56 gr c) 17.82 gr. b) 2.14 gr d) 8.91 gr
Chapter9: Current And Resistance
Section: Chapter Questions
Problem 81CP: A 10-gauge copper wire has a cross-sectional area A=5.26mm2 and carries a current of I=5.00A . The...
Related questions
Concept explainers
Ohm's law
Ohm’s law is a prominent concept in physics and electronics. It gives the relation between the current and the voltage. It is used to analyze and construct electrical circuits. Ohm's law states that the voltage across a conductor is directly proportional to the current flowing through it.
Path of Least Resistance
In a series of alternate pathways, the direction of least resistance is the actual or metaphorical route that offers the least resistance to forwarding motion by a given individual or body.
Question
Transcribed Image Text:202
Two copper plates are immersed in a copper sulfate solution
and connected to a battery. The reading of the Ammeter
connected to the cell is 2.5 A and this current lasted for three
hours. If the atomic mass of copper is 63.6 u
(lu = 1.66 x 10-27 kg), and two electrons are needed to
discharge one copper ion, the mass of copper deposited on the
Cathode plate is
ered
13.50
7
ion
13
Finis
a) 0.71 gr
c) 3.56 gr
e) 17.82 gr.
b) 2.14 gr
d) 8.91 gr
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

