Two neutral objects are sketched below, in black and white. There are positive and negative electric charges inside these objects, and at least some of those charges can move around. Now suppose a positive charge comes close to these objects, as shown in the sketch. Shade the objects red anywhere you expect them to become more negatively charged, and shade the objects blue anywhere you expect them to become more positively charged.
Two neutral objects are sketched below, in black and white. There are positive and negative electric charges inside these objects, and at least some of those charges can move around. Now suppose a positive charge comes close to these objects, as shown in the sketch. Shade the objects red anywhere you expect them to become more negatively charged, and shade the objects blue anywhere you expect them to become more positively charged.
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter20: The Representative Elements
Section: Chapter Questions
Problem 1RQ: What are the two most abundant elements by mass in the earths crust, oceans, and atmosphere? Does...
Related questions
Question
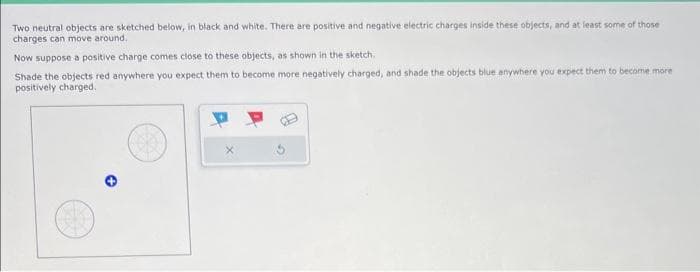
Transcribed Image Text:Two neutral objects are sketched below, in black and white. There are positive and negative electric charges inside these objects, and at least some of those
charges can move around.
Now suppose a positive charge comes close to these objects, as shown in the sketch,
Shade the objects red anywhere you expect them to become more negatively charged, and shade the objects blue anywhere you expect them to become more
positively charged.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning