Unknown Number Color in H₂O 3 purple Unknown Spot(s) Color in Ammonia Spot #1 Spot #2 Spot #3 Blue yellow N/a Color after CaS (aq) Black yellow N/A Distance Spot Moved 57cm 5cm N/A Distance Solvent Front Moved 7.8 cm 6.5cm NA Calculated R 0.8974 0.76923 N/A Identity of Metal Ion
Unknown Number Color in H₂O 3 purple Unknown Spot(s) Color in Ammonia Spot #1 Spot #2 Spot #3 Blue yellow N/a Color after CaS (aq) Black yellow N/A Distance Spot Moved 57cm 5cm N/A Distance Solvent Front Moved 7.8 cm 6.5cm NA Calculated R 0.8974 0.76923 N/A Identity of Metal Ion
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter13: An Introduction To Ultraviolet-visible Molecular Absorption Spectrometry
Section: Chapter Questions
Problem 13.11QAP: The equilibrium constant for the conjugate acid-base pair HIn+H2OH3O++In is 8.00 10-5. From the...
Related questions
Question
Please help with the last boxes of "identity of metal ion"
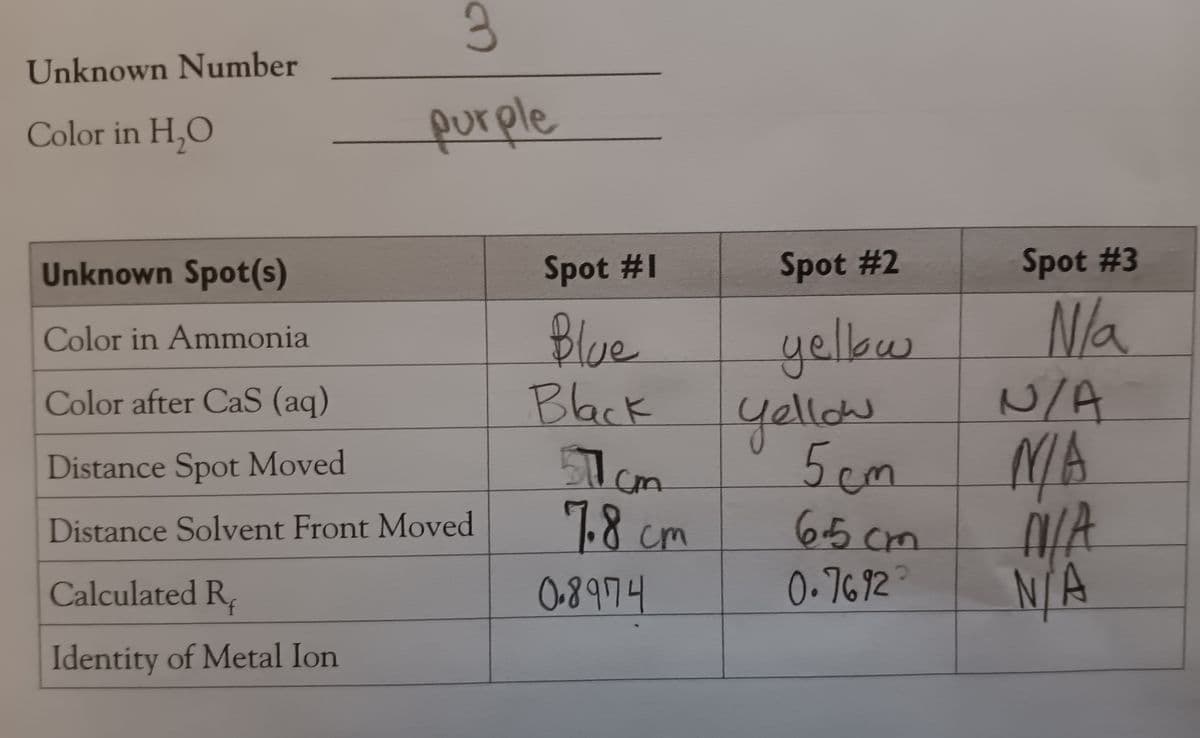
Transcribed Image Text:Unknown Number
Color in H₂O
3
purple
Unknown Spot(s)
Color in Ammonia
Spot #1
Spot #2
Spot #3
Blue
yellow
N/a
Color after CaS (aq)
Black
yellow
N/A
Distance Spot Moved
57cm
5cm
N/A
Distance Solvent Front Moved
7.8 cm
6.5cm
NA
Calculated R
0.8974
0.76923
N/A
Identity of Metal Ion
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

