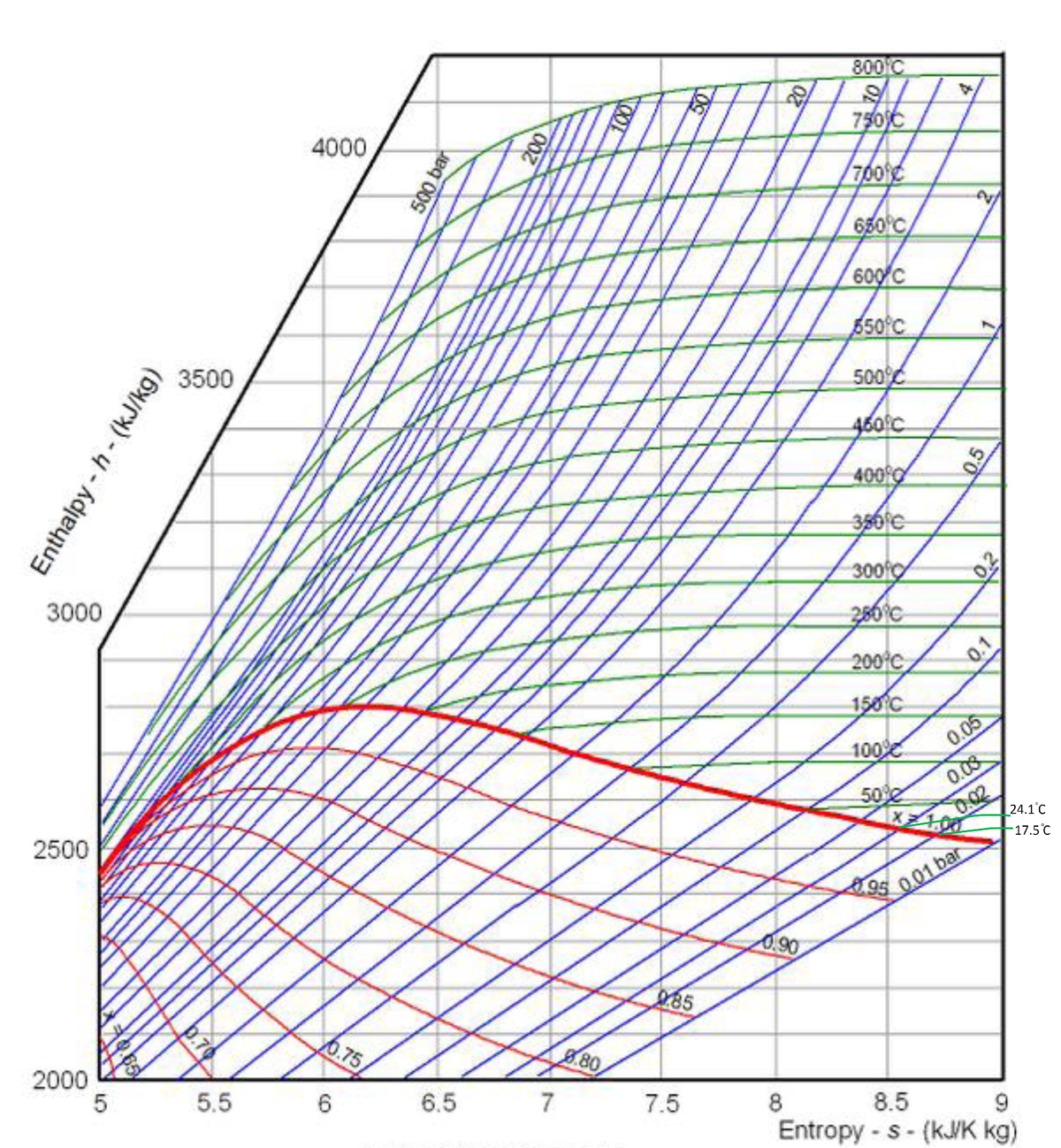
Use only the MD below to solve this Item; use it by plotting data and process to determine items being asked. Steam enters a turbine at 117.4308 atmgage and 932 deg. F, and expands isentropicaly to 177.17 inches of Hg. For 3.10 kg/sec of steam, what is (a) the ideal work done (KJ) if ∆KE = 0 and actual exhaust enthalpy is 2700 KJ/kg. Ans:_________________ (b)ne = Ans:___________ (c)Determine: ideal steam moisture at exhaust. Ans:________________
Use only the MD below to solve this Item; use it by plotting data and process to determine items being asked. Steam enters a turbine at 117.4308 atmgage and 932 deg. F, and expands isentropicaly to 177.17 inches of Hg. For 3.10 kg/sec of steam, what is (a) the ideal work done (KJ) if ∆KE = 0 and actual exhaust enthalpy is 2700 KJ/kg. Ans:_________________ (b)ne = Ans:___________ (c)Determine: ideal steam moisture at exhaust. Ans:________________
Principles of Heat Transfer (Activate Learning with these NEW titles from Engineering!)
8th Edition
ISBN:9781305387102
Author:Kreith, Frank; Manglik, Raj M.
Publisher:Kreith, Frank; Manglik, Raj M.
Chapter5: Analysis Of Convection Heat Transfer
Section: Chapter Questions
Problem 5.2P: 5.2 Evaluate the Prandtl number from the following data: , .
Related questions
Question
100%
- Use only the MD below to solve this Item; use it by plotting data and process to determine items being asked. Steam enters a turbine at 117.4308 atmgage and 932 deg. F, and expands isentropicaly to 177.17 inches of Hg. For 3.10 kg/sec of steam, what is
(a) the ideal work done (KJ) if ∆KE = 0 and actual exhaust enthalpy is 2700 KJ/kg. Ans:_________________
(b)ne = Ans:___________
(c)Determine: ideal steam moisture at exhaust. Ans:________________
(d) Actual steam quality at exhaust Ans: ________________
(e) Actual steam temp at the exhaust. Ans.__________
(f) Degree SH of the steam at turbine inlet. Ans_____________
(g) change in entropy during the actual process. Ans:__________________
Transcribed Image Text:800 C
4000
750C
700%
650°C
600 C
650°C
3500
500
460°C
400
356°e
300f
3000
286°C
200°c
150°C
100
0.05
0.08
0.02
X100
50
24.1°C
2500
-17.5°C
001 bar
95
0.90
85
0.75,
2000
,80
5.5
6
6.5
7.5
8
8.5
Entropy - s- (kJK kg)
Enthalpy - h- (kJ/kg)
500 bar
002
0.5
7.
0.1
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning