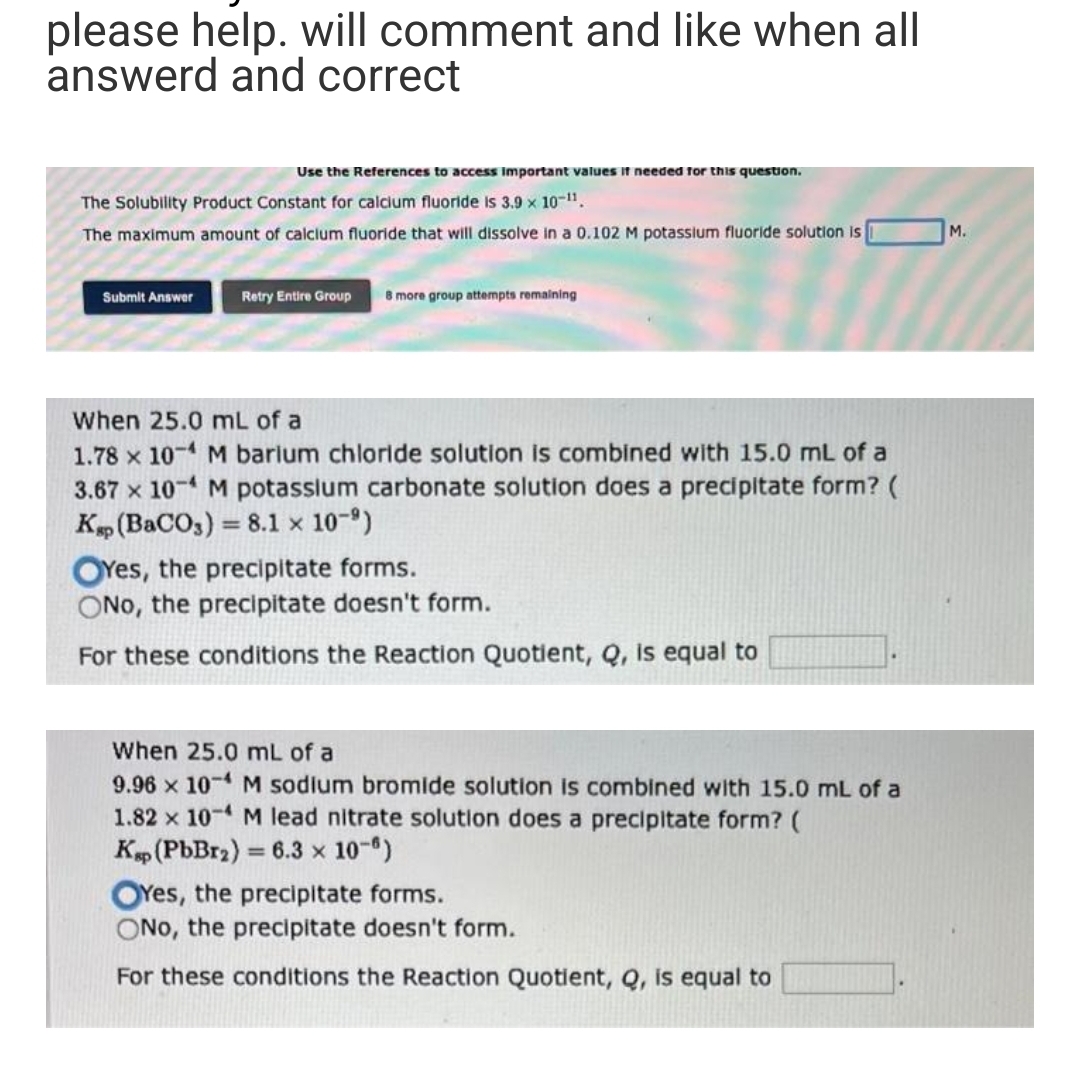
Use the References to access important values if needed for this question. The Solubility Product Constant for calcium fluoride is 3.9 x 10-¹¹. The maximum amount of calcium fluoride that will dissolve in a 0.102 M potassium fluoride solution is Submit Answer Retry Entire Group 8 more group attempts remaining When 25.0 mL of a 1.78 x 10-4 M barium chloride solution is combined with 15.0 mL of a 3.67 x 10-4 M potassium carbonate solution does a precipitate form? ( Kap (BaCO3) = 8.1 × 10-⁹) OYes, the precipitate forms. ONo, the precipitate doesn't form. For these conditions the Reaction Quotient, Q, is equal to When 25.0 mL of a 9.96 x 10-4 M sodium bromide solution is combined with 15.0 mL of a 1.82 x 10-4 M lead nitrate solution does a precipitate form? ( Ksp (PbBr₂) = 6.3 × 10-6) OYes, the precipitate forms. ONo, the precipitate doesn't form. For these conditions the Reaction Quotient, Q, is equal to M.
Use the References to access important values if needed for this question. The Solubility Product Constant for calcium fluoride is 3.9 x 10-¹¹. The maximum amount of calcium fluoride that will dissolve in a 0.102 M potassium fluoride solution is Submit Answer Retry Entire Group 8 more group attempts remaining When 25.0 mL of a 1.78 x 10-4 M barium chloride solution is combined with 15.0 mL of a 3.67 x 10-4 M potassium carbonate solution does a precipitate form? ( Kap (BaCO3) = 8.1 × 10-⁹) OYes, the precipitate forms. ONo, the precipitate doesn't form. For these conditions the Reaction Quotient, Q, is equal to When 25.0 mL of a 9.96 x 10-4 M sodium bromide solution is combined with 15.0 mL of a 1.82 x 10-4 M lead nitrate solution does a precipitate form? ( Ksp (PbBr₂) = 6.3 × 10-6) OYes, the precipitate forms. ONo, the precipitate doesn't form. For these conditions the Reaction Quotient, Q, is equal to M.
Chapter11: Solving Equilibrium Problems For Complex Systems
Section: Chapter Questions
Problem 11.5QAP
Related questions
Question
Transcribed Image Text:please help. will comment and like when all
answerd and correct
Use the References to access important values if needed for this question.
The Solubility Product Constant for calcium fluoride is 3.9 × 10-11.
The maximum amount of calcium fluoride that will dissolve in a 0.102 M potassium fluoride solution is
Submit Answer
Retry Entire Group 8 more group attempts remaining
Fil
When 25.0 mL of a
1.78 x 10-4 M barium chloride solution is combined with 15.0 mL of a
3.67 x 10-4 M potassium carbonate solution does a precipitate form? (
Kap (BaCO3) = 8.1 × 10-⁹)
OYes, the precipitate forms.
ONo, the precipitate doesn't form.
For these conditions the Reaction Quotient, Q, is equal to
When 25.0 mL of a
9.96 x 10-4 M sodium bromide solution is combined with 15.0 mL of a
1.82 x 10-4 M lead nitrate solution does a precipitate form? (
Ksp (PbBr₂) = 6.3 × 10-8)
M.
OYes, the precipitate forms.
ONO, the precipitate doesn't form.
For these conditions the Reaction Quotient, Q, is equal to
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

