Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter14: Acid-base Equilibria
Section: Chapter Questions
Problem 113E: Draw a curve for a series of solutions of HF. Plot [H3O+]total on the vertical axis and the total...
Related questions
Question
Using the data table 1 please fill the data table 2
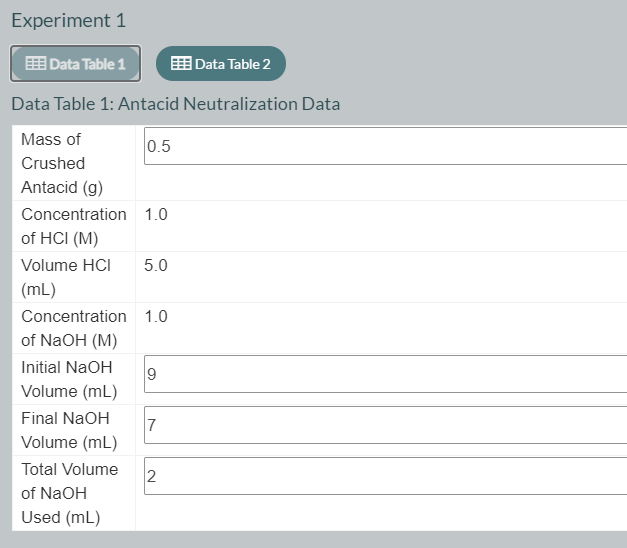
Transcribed Image Text:Experiment 1
E Data Table 1
E Data Table 2
Data Table 1: Antacid Neutralization Data
Mass of
0.5
Crushed
Antacid (g)
Concentration 1.0
of HCI (M)
Volume HCI
5.0
(mL)
Concentration 1.0
of NaOH (M)
Initial NaOH
Volume (mL)
Final NaOH
7
Volume (mL)
Total Volume
2
of NaOH
Used (mL)
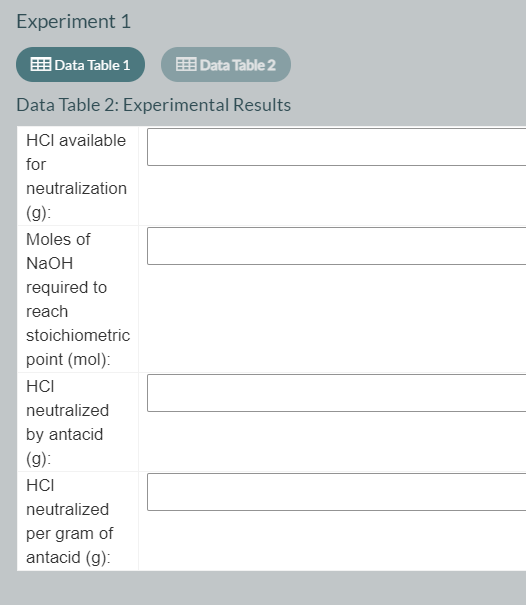
Transcribed Image Text:Experiment 1
Data Table 1
EE Data Table 2
Data Table 2: Experimental Results
HCI available
for
neutralization
(g):
Moles of
NaOH
required to
reach
stoichiometric
point (mol):
HCI
neutralized
by antacid
(g):
HCI
neutralized
per gram of
antacid (g):
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax