Using the equation E = (hcRH) ()=(-2.18 x Xb calculate the energy of an electron in the hydrogen atom when n = 4. Express your answer in joules to three significant figures. ΠΨΕΙ ΑΣΦ − (−2.18 × 10–¹8 • درات EA-1.36 10. 16 J) (17) √xxx x IXI 19 Submit Previous Answers Request Answer 1920 X-10h ESSEN J X Incorrect; Try Again; 2 attempts remaining Substitute n = 4 into the given equation and solve it for E4. Since the proportionality constant (-2.18 × 1 and the principal quantum number is always positive, your answer will have a negative value.
Using the equation E = (hcRH) ()=(-2.18 x Xb calculate the energy of an electron in the hydrogen atom when n = 4. Express your answer in joules to three significant figures. ΠΨΕΙ ΑΣΦ − (−2.18 × 10–¹8 • درات EA-1.36 10. 16 J) (17) √xxx x IXI 19 Submit Previous Answers Request Answer 1920 X-10h ESSEN J X Incorrect; Try Again; 2 attempts remaining Substitute n = 4 into the given equation and solve it for E4. Since the proportionality constant (-2.18 × 1 and the principal quantum number is always positive, your answer will have a negative value.
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter16: Thermodynamics: Directionality Of Chemical Reactions
Section: Chapter Questions
Problem 16.CCP
Related questions
Question
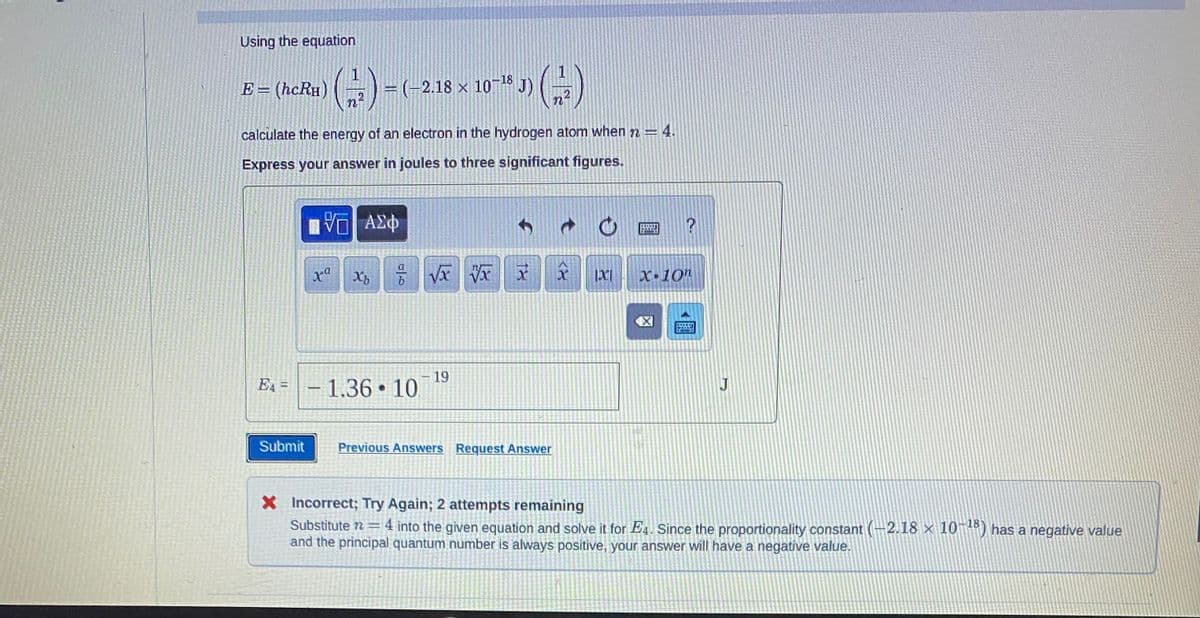
Transcribed Image Text:Using the equation
E = (hcRH)
E4=
(1)
2
calculate the energy of an electron in the hydrogen atom when 72=
Express your answer in joules to three significant figures.
ΠΨΕΙ ΑΣΦ
Submit
xª
1
=(-2.18 x 10-18
× J) (-/-)
n
X√xVx
b
Xb
- 1.36 10.
-19
X
Previous Answers Request Answer
|X|
TH
?
X-10"
J
X Incorrect; Try Again; 2 attempts remaining
Substitute 7 = 4 into the given equation and solve it for E4. Since the proportionality constant (-2.18 × 10-18) has a negative value
and the principal quantum number is always positive, your answer will have a negative value.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning