Using the Steam Table, at initial pressure of 0.046 bar and final pressure of 0.107 bar, find the following: Δhfg
Using the Steam Table, at initial pressure of 0.046 bar and final pressure of 0.107 bar, find the following: Δhfg
Refrigeration and Air Conditioning Technology (MindTap Course List)
8th Edition
ISBN:9781305578296
Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Chapter46: Room Air Conditioners
Section: Chapter Questions
Problem 15RQ
Related questions
Question
Using the Steam Table, at initial pressure of 0.046 bar and final pressure of 0.107 bar, find the following:
Δhfg
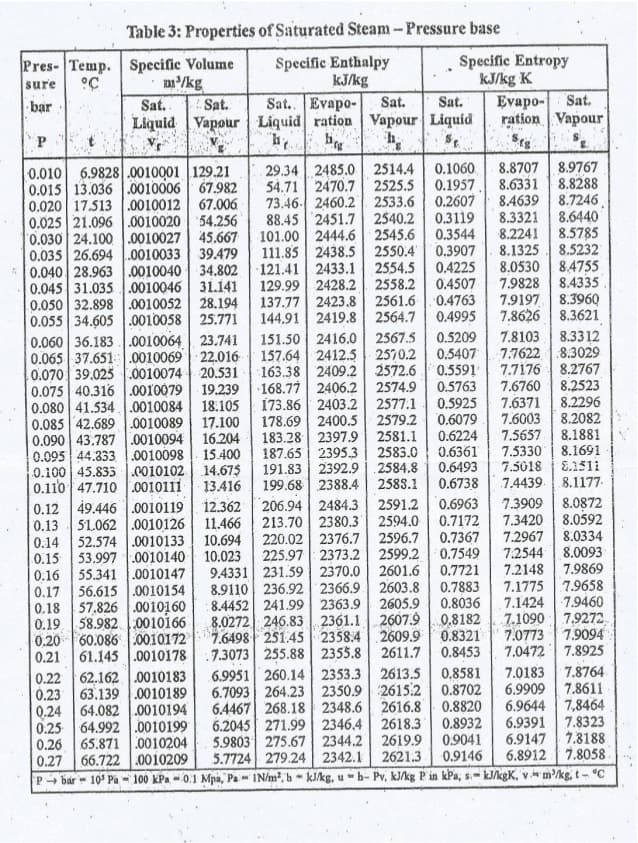
Transcribed Image Text:Table 3: Properties of Saturated Steam-Pressure base
Specific Entropy
KJ/kg K
Evapo- Sat.
ration Vapour
Pres- Temp. Specific Volume
°C
Specific Enthalpy
kJ/kg
sure
am'/kg
Sat. Evapo-
Liquid Vapour Liquid ration Vapour Liquid
bar:
Sat.
Sat.
Sat.
Sat.
29.34 2485.0 | 2514.4
54.71 2470.7
73.46- 2460.2 2533.6
88.45 2451.7 2540.2
101.00 2444.6 2545.6
111.85 2438.5 2550.4
121.41 2433.1
129.99 2428.2
137.77 2423.8
144.91 2419.8
151.50 2416.0 2567.5
8.8707
8.9767
8.8288
0.1060.
0.1957 8.6331
0.2607
0.3119
0.010 6.9828 .0010001 129.21
0.015 13.036 .0010006 67.982
0.020 17.513 .0010012 67.006
0.025 21.096 .0010020 54.256
'0.030 24.100 .0010027
0.035 26.694 .0010033 39.479
*0.040 28.963 .0010040 34.802
0.045 31.035.0010046 31.141
0.050 32.898 .0010052 28.194
0.055 34.605 .0010058 25.771
0.060 36.183 .0010064 23.741
0.065 37.651.0010069 22.016 157.64 2412.5 2570.2
0.070 39.025 .0010074 20.531
0.075 40.316 .0010079
0.080 41.534.0010084 18.105 173.86 2403.2 2577.1
0.085 42.689 .0010089
0.090 43.787 .0010094 16.204
0.095 44.333 .0010098
0.100 45.833 .0010102 14.675
0.110 47.7100010111 13.416
0.12 49.446 |.0010119 12.362
2525.5
8.4639 8.7246.
8.3321 8.6440
8.5785
8.2241
8.1325 8.5232
8.0530 84755
8.4335
45.667
0.3544
2554.5
2558.2
2561.6
2564.7
0.3907
0.4225
0.4507
0.4763
0.4995
7.9828
7.9197 8.3960
7.8626 8.3621
0.5209
7.8103
8.3312
7.7622 1.8.3029
7.7176 8.2767
8.2523
8.2296
8.2082
8.1881
8.1691
163.38 2409.2 2572.6
168.77 2406.2 2574.9
0.5407
0.5591
0.5763
19.239
7.6760
0.5925
0.6079
0.6224
0.6361
0.6493
7.6371
7.6003
7.5657
7.5330
7.5018
7.4439
178.69 2400.5
183.28 2397.9
187.65 2395.3
191.83 2392.9
199.68 2388.4
206.94 2484.3
11.466 213.70 2380.3 2594.0
220.02 2376.7 2596.7
225.97 2373.2 | 2599.2
2579.2
2581.1
2583.0
17.100
15.400
2584.8
2583.1
E.1511
0.6738
8.1177-
8.0872
8.0592
2591.2
0.6963
7.3909
7.3420
7.2967
7.2544
7.2148
0.13
51.062 .0010126
0.7172
0.7367
0.7549
8.0334
8.0093
7.9869
52.574 .0010133
0.15 53.997.0010140 10.023
0.16 55.341 .0010147
0.17 56.615.0010154
0.18 57.826 .0010160
0.19 58.982 0010166
0.14
10.694
9.4331 231.59 2370.0
8.9110 236.92 2366.9
8.4452 241.99 2363.9
0272 2405 23584
2601.6
2603.8
2605.9
2361.1 2607.9
2609.9
2611.7
0.7721
0.7883
0.8036
7.1775 7.9658
7.1424 7.9460
7.9094
7.0472 | 7.8925
0.21 61.145 .00101787.3073 255.88 2355.8
0.22 62.162 0010183
0.8453
6.9951 260.14 2353.3
6.7093 264.23 2350.9 2615.2
6.4467 268.18 2348.6 2616.8
6.2045 271.99 2346.4
5.9803 275.67 2344.2
5.7724 279.24 2342.1
2613.5
0,8581
7.0183
7.8764
6.9909 7.8611
6.9644 7,8464
6.9391 7.8323
6.9147 7.8188
6.8912 | 7.8058
63.139 .0010189
0.8702
0.8820
0.23
0.24 64.082 .0010194
0.25 64.9920010199
0.26 65.871 .0010204
0.27 66.722 0010209
2618.3
2619.9
2621.3
0.8932
0.9041
0.9146
Pbar- 10' Pa - 100 kPa -0.1 Mpa, Pa IN/m2, h kJ/kg, ub- Pv, kJ/kg P in kPa, s- k/kgK, vm/kg, t- °C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Refrigeration and Air Conditioning Technology (Mi…
Mechanical Engineering
ISBN:
9781305578296
Author:
John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:
Cengage Learning

Refrigeration and Air Conditioning Technology (Mi…
Mechanical Engineering
ISBN:
9781305578296
Author:
John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:
Cengage Learning