Q: What is the major product of the following reaction? A. B. H,C रे H, C OH H,SO. Heat C. D. H.C र
A:
Q: Answer and explain in detail how you arrived at that answer.
A:
Q: Which statement best describes what happens during a decomposition reaction? A B C An atom or ion in…
A: The chemical reactions are mainly classified into i) combination reaction ii) decomposition…
Q: (B) Compound 2 name:_ H3C -0 Olli Holl H N-CH3
A: The given drugs are analgesics of the drug Morphine. Compound 2 = Codeine
Q: What is the molecular formula of the following compound? Please write your answer using this format:…
A: •Q.1)=>
Q: 12. Which of the following statements is verifiably true about this equilibrium? PCIDPCl*(g) +…
A:
Q: 7.53 Which of the following nucleophilic substitution reactions will take place? NH₂ a. b. CH3CH₂I +…
A:
Q: (i) (ii) (iii) MeO₂C ZI CO₂Me D || D -D [D = ²H] N. HO₂C OH CI OMe
A: Step by step arrow mechanism of each reaction.
Q: 3) Oxygen has an unstable isotope O-17 that has a mass of 17.00454. If the mass of a neutron is…
A: Oxygen isotope(O-17) Mass of oxygen = 17.00454 Mass of neutron = 1.00898 u Mass of proton = 1.00814…
Q: Consider the following reaction: 2CH₂OH(1) + 30₂(g) → 2CO₂ (g) + 4H₂0 (1) What are the oxidation…
A: for the given reaction, we have to determine : oxidation state of elements how oxidation states…
Q: It is desired to assemble a concentration cell with a potential of 120 mV with two Zn(s) electrodes…
A: A concentration cell is a type of electrochemical cell that has two electrodes that are identical in…
Q: An 89.3 mL sample of wet O2(g) is collected over water at 21.3°C at a barometric pressure of 756…
A: Given :- Volume of wet oxygen = VwO2 = 89.3 mL Total Pressure = Ptotal = PdO2 = 756 mmHg Vapor…
Q: For a weak acid (HA) dissolved in water, which of the following describes the major species in…
A: WEAK ACID:- Weak acid are those acids which are very less dissociate when dissolved in water. •Means…
Q: A generic antacid contains sodium bicarbonate, NaHCO3, as its active ingredient. A student added a…
A: Mass of antacid = 2.000g Volume of 5% vinegar = 20 mL Volume of water = 15 mL Mass of beaker with…
Q: Q1 Explain step-wise showing all detailed solutions (a) Explain the difference in the dipole moments…
A: a) we know that the dipole moment is permanent polarity of the molecule. It determines how much a…
Q: The molecules of a certain gas sample at 353 K have a root-mean-square (rms) speed of 283 m/s.…
A: Given Gas sample having temperature = 353 K Root mean square velocity = 283 m/s Gas constant =…
Q: Lope NH₂ HO OH NH, NH, OH
A: The given functional group in boxed at the left-hand side has an alcohol functional group.…
Q: A is versatile reagent for the synthisis of a wide range of ketone. Suggest reagents and condition…
A: Aceto acetic ester (AAE) contains an activated methylene group (methylene group attached with two…
Q: Which resonance structures fulfill the octet rule for carbons and oxygen? H H A H - H H B H =0 H. 0…
A: There are two type of bonds :- Sigma Pi(π) Sigma bond is formed by head on overlapping of…
Q: Which resonance structure is the least stable? H H. C A B C OO H- H10O B H C. ====0 H. 115H Н C H CO…
A: •Here is resonance structure ate given:-
Q: A 0.235 g sample of a metal, M, reacts completely with sulfuric acid according to M(s) + H₂SO4 (aq)…
A:
Q: A sample containing 0.200 g primary standard calcium carbonate (CaCO3, FW-100.09 g/mol) was…
A: The 'hardness' of water and the concentration of calcium carbonate in various substances are…
Q: a) Draw the Lewis structure for NCI3 b) State the VSEPR notation. c) Predict the shape of the…
A: Since you have posted a question with multiple sub-parts,we will solve first three sub-parts for…
Q: Q20. Determine the direction of the reaction. (draw an arrow to show the real direction) A. Bal2 +…
A: Note: Since you have posted a question with multiple sub-parts, we will provide the solution only to…
Q: Fast pls i will give u like for sure solve this question correctly in 5 min
A: For resonance to occur there must be proper conjugation with vacant orbital of the atom . During…
Q: The rate law constant for the first-order decomposition of cyclobutane is 9.2 X 10-3 sec 1. C4H8…
A: We know for first order reaction t1/2 = 0.693/k Where t1/2 = half life and K = first order rate…
Q: Cu(IO3)2 (s) + 2e- → Cu(s) + 2 IO3-(aq) give the semi-reactions
A: •Given reaction:- Cu(IO3)2(s) + 2e- → Cu(s) + 2 IO3-(aq) •Here we have to write semi…
Q: A raindrop has a mass of 50. mg and the Pacific Ocean has a mass of 7.08 x 1020 kg. Use this…
A: Answer:- This question is answered by using the simple concept of calculation of mass and moles…
Q: Complete the table below, using the diagram of an atom shown at right. name symbol proton 0 0 0…
A: The basic unit of matter that makes up all elements is called an atom. It is composed of a nucleus,…
Q: A hypothetical metal A is stiffer than metal B, Has lower thermal expansion coefficient than B, and…
A: In the given question we have to draw the graph of the potential energy Versus inter-atomic spacing…
Q: (c) The reduction of a cyclohexanone 7 to the corresponding cyclohexanol 8 is shown below. Choose a…
A: Axial and Equatorial Positions in Cyclohexane. Careful examination of the chair conformation of…
Q: For the forward reaction, can I use H3O+ instead?
A:
Q: Given the following information: Ni²+ (aq) + 2 e → Ni (s) Eº = -0.250 V K₁ = 1.0 x 10²² for the…
A: We know logK = 16.9nE0cell
Q: Write the equilibrium constant expression for this reaction: 2 CH3COO (aq) + С₂H₂O₂(aq) → 0…
A:
Q: 12. The amount of energy required for a reaction to begin is known as d. kinetic energy a. enthalpy…
A: The amount of energy required for a reaction to begin is known as.... Answer is given below with…
Q: A. I and III B. II and III C. I and IV D. III and IV Q18. How many (single o, n,) bonds are in the…
A:
Q: сх OH OH From -OH
A: Retro-synthesis : It is the technique through which we can analyse the synthesis of target molecule…
Q: 10. What property would be appropriate to measure the reaction rate in the following reaction: N2(g)…
A: Property which is appropriate to check rate of following reaction.
Q: Question The simplest carboxylic acid is methanoic acid (or formic acid), HCOOH, which occurs…
A: Given , Volume of methanol injected =6.0×10-3mL
Q: 9. Consider the following equilibrium: 2HI(g) + 26kJ H4(g) + 14(g) If some hydrogen gas were…
A: The problem given above is actually based on equilibrium and here we will use Le Chatelier's…
Q: 1. For the following reaction, AP (H₂) / At was found to be -0.0574 atm / s. N2 (g) + 3 H2(g) →→ 2…
A: Reaction :- N2(g) + 3H2(g) -----> 2NH3(g) ∆P(H2)/∆t = -0.0574 atm/s ∆P(NH3)/∆t = ?
Q: 6. A 0.500 M solution of ascorbic acid (vitamin C) has a pH of 2.20. Find the Ka of ascorbic acid.
A:
Q: What was causing the mussel shells to erode?
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: What is the molarity of KMnO4 in a solution of 0.0849 g of KMnO4 in 0.450 L of solution? (Answer in…
A: Mass of KMnO4 = 0.0849 g Volume of solution = 0.450 L To determine: Molarity of KMnO4 solution
Q: A compound with molecular mass 71 transparent in the ultraviolet spectrum. Infra- red, medium bands…
A:
Q: Calculate the mass of fluorine (in g ) contained in 61.0 g of copper(II) fluoride.
A: 1.Copper (ll) flouride is CuF2 . 2. Given mass of CaF2 is 61. 0 g Mass of fluorine (F) =?
Q: What mass (g) of sodium hydrogencarbonate would be required to completely neutralise a typical ant…
A: An ant injects a fluid that contains 50% by volume of methanoic acid when it bites. An average ant…
Q: What is the effect of increasing temperature on the rate, rate constant and activation energy?…
A: Solution - According to collision theory as the temperature increases number of collisions increases…
Q: Fill in the missing information: symbol 2+ Mg atom or lon? check all that apply neutral atom neutral…
A: Simple rules can be used to calculate the number of protons, neutrons, and electrons in an atom.A…
Q: What volumes of 0.200 M HNO2 and 0.200 M NaNO2 are required to make 500. mL of a buffer solution…
A:
What is the nomenclature of the following compound?
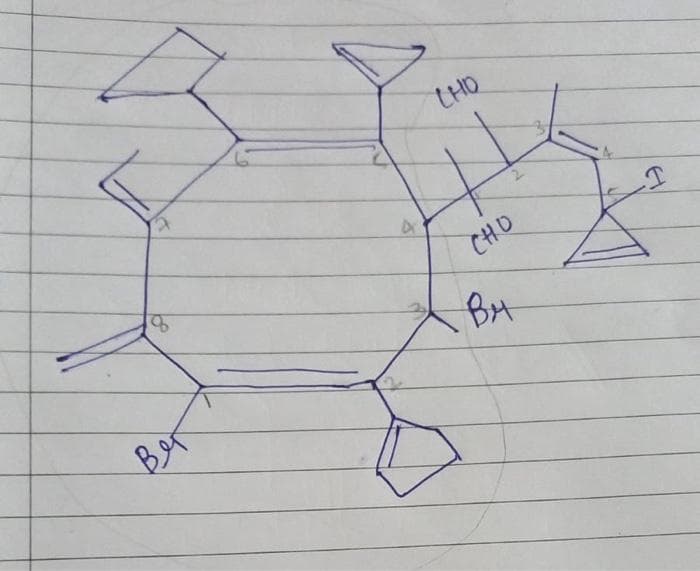
Step by step
Solved in 2 steps with 1 images


