Q: number of molecules in 1.0 micromole of water
A: 1.0 mole of water = 6.02 X 1023 molecules
Q: Carbon and hydrogen form polar covalent bonds Carbon chains can be linear or branched
A: Polar covalent bond means that there is partial negative and partial positive charge on the two…
Q: Why is water considered the universal solvent? A:Water molecules are weakly bonded to other water…
A: Biomolecules are those organic compounds that are made up of carbon and hydrogen with some other…
Q: lons are: a. atoms or molecules with unequal numbers of electrons and protons b. atoms or molecules…
A: Question - lons are - a.) atoms or molecules with unequal numbers of electrons and protons b.)…
Q: Which form of water has the highest density? They are all the same a O b Ice O c Water vapor Liquid…
A: The density of water changes with changes in temperature and also with changes in salinity.
Q: Hydrogen bond lonic (electrostatic) interaction Hydrophobic interactions Disulfide bond
A: Chemical bonds are necessary for life because they bind molecules together and generate momentary…
Q: Solid particles from broken apart plant and animal cells suspended in water. A. Jellies B. Foam…
A: We are authorized to answer one question at a time since you have not mentioned which question you…
Q: Why does ice float in water?
A: It is a dipolar molecule. It is irregular with tetrahedral geometry. Each molecule in liquid water…
Q: A positively charged atom has _____. gained protons gained electrons lost electrons lost protons
A: An atom is the smallest unit of ordinary matter that forms a chemical element. Every solid, liquid,…
Q: Reactions in which water is used to break apart other molecules areknown as __________ reactions.a.…
A: A reaction is a process by which one molecule is transformed into another. In living systems these…
Q: Which of the following statements is not true? a. Water is polar. b. Water stabilizes temperature.…
A: Importance of Water.
Q: an atom whose number of electrons does not equal its number of protons is called... ion isotope…
A: Atom is the smallest unit of any matter that may be solid, liquid or gas. The core of the atom is…
Q: We say that water molecules are because they have a positive and negative end. Elements O a O b Weak…
A: Because of the unequal distribution of electrons among the atoms and the molecule's asymmetrical…
Q: The smallest unit of any substance is the . a. atom b. molecule c. cell
A: The cell is the basic structural, and function unit of all living organisms. Cells are the smallest…
Q: The mass number of an atom depends primarily on the number of protons and neutrons. positrons.…
A: An atom is the smallest unit of any matter. It has three subatomic particles; neutrons, protons, and…
Q: In a solution of NaCl and water, NaCl is the and water isthe .a. acid, baseb. base, acid c. solute,…
A: NaCl (sodium chloride) is the salt produced by the strong acid HCl and strong base NaOH. The aqueous…
Q: The charge____________ of a proton is exactly balanced by thecharge of a (an) ____________.a.…
A: An atom consists of proton, electron and neutron. Proton is positively charged. Electron is…
Q: light oxygen carbon dioxide carbohydrates water
A: Answer of the question given below...
Q: which structure is formed when fatty acid salts react with water? choices: bilayer, monolayer,…
A: A fatty acid consists of ten or more carbon atoms.
Q: Which of the following is a hydrophobic material?(A) paper (B) table salt(C) wax (D) sugar
A: Hydrophobic means the fear of water, where as hydrophilic is the liking of the water. The molecules…
Q: Sugar is composed of carbon, hydrogen, and oxygen. Sugar is an example of which of the following? an…
A: Any substance which contains two or more chemical elements and these chemical elements are…
Q: Which type of molecules is important for long-term storage of energy? sugar O carbohydrate O lipid O…
A: The nutrients that provide energy are commonly referred to as macronutrients (carbohydrates, lipids,…
Q: Which of the following statements is NOT true of biological organic molecules? None of the other…
A: A complex molecule that is primarily made of carbon atoms bonded with other elements and/or other…
Q: . A cation is a(n)a. uncharged atom.b. positively charged atom.c. negatively charged atom.d. atom…
A: A molecule is considered to be composed of two or more atoms having the same elements, which are…
Q: In a solution the part of the solution that is doing the dissolving (such as water in our cells and…
A: Every solution has two different parts, Solute Solvent
Q: Which row correctly identifies chemical elements found in fats and carbohydrates? carbon hydrogen…
A: Introduction: Those molecules that structure and perform various functions in the living body are…
Q: what is the basic unit of matter
A: Anything that has mass and is capable of occupying space is considered as matter. Different or…
Q: acids: basic building blocks of protein molecules. acid: a nucleic acid; the master copy of an…
A: Nucleic acids - These are biomolecules which make up the genetic material of the cell. They are made…
Q: In a water molecule, hydrogens are partially _____; oxygens are partially _____.
A: Hydrogens : It is the chemical element with the symbol H and atomic number 1 with a standard atomic…
Q: The tendency for water molecules to want to stick together is referred to as
A: The tendency of water molecules to stick together is referred as COHESION. The water molecules form…
Q: Monomers or Components Poly mer or larger molecule Type of linkage Carbchydrates Lipids Proteins…
A: Biomolecules are biological molecules produced by cells and organisms. The four major types of…
Q: A(n). is the smallest unit of an element that retains all the properties of that element. O molecule…
A: Elements are substances that cannot be further broken into particles. An element consists of types…
Q: A solution with a pH of 2 has a hydrogen ion concentration that is _______________ the hydrogen ion…
A: pH is a measure of the negative logarithm of the hydrogen ion concentration. Given Values: pH = 2.0…
Q: Which of the following statements is not true?a. Water is polar.b. Water stabilizes temperature.c.…
A: Water is a colorless, tasteless, odorless chemical substance which consists of hydrogen and oxygen…
Q: The 3 parts of a nucleotide are O A. water, solute, solvent O B. sugar, phosphate,…
A: Biomolecules of molecular weight above 1000 Da is known as biomacromolecules. Carbohydrates,…
Q: Which of the following choices correctly identifies each molecule in the diagram below? Molecule…
A: The given reaction is of reversible type. In this reaction, small molecules are getting joined with…
Q: vel 1) O bonds in carbon dioxide molecul 2 bonds in glucose molecules atoms of oxygen
A: Cellular respiration is the mechanism by which organisms mix oxygen with food molecules, diverting…
Q: What type of bonds occur BETWEEN water molecules? OPolar covalent Non polar covalent Hydrogen O…
A: Water molecule consists of two atoms of hydrogen linked to the same atom of oxygen.
Q: Two or more chemical compounds that have the same formula, but have different structures are known…
A: Any substance made up of similar molecules made up of atoms from two or more chemical elements is…
Q: The number of protons in a neutral (no charge) atom equals the number of ions. isotopes. neutrons…
A: An ion is a an atom or molecule which is charge, either positively or negatively, based on the…
Q: Multiple water molecules are held together by... A. Dehydration synthesis B. Covalent bonds C.…
A: Water is also known as universal solvent because of its ability to dissolve almost everything. A…
Q: ________ are to proteins as _______ are to nucleic acids.a. Sugars; lipidsb. Sugars; proteinsc.…
A: DNA contains the genetic information for the physical characteristics of the human body. The DNA…
Q: Monomers or Components Poly mer larger molecule or Type of linkage Carbohydrates Fats Proteins…
A: Biomolecules are the important organic molecules found in all living cells. They perform vital…
Q: The protons and neutrons are contained within the of the atom. Electron Cloud Atomic Center Middle…
A: All matter is made up of tiny indivisible and indestructible раrtiсles called аtоms, according to…
Q: Many people are aware that water is the most abundant substance in all organisms. Which component of…
A: The cell is the structural, functional, and biological unit of all animals. It is a self-replicating…
Q: Explain in detail why water molecules are polar. Your explanation can be as long as you wish. Your…
A:
Q: What is a polymer? * a collection of different types of molecules O a chain of the same type of…
A: A repetitive long chain of monomers is called a polymer. Monomer is basically the building block of…
Does osmosis occur if a cell is placed in an isotonic solution?
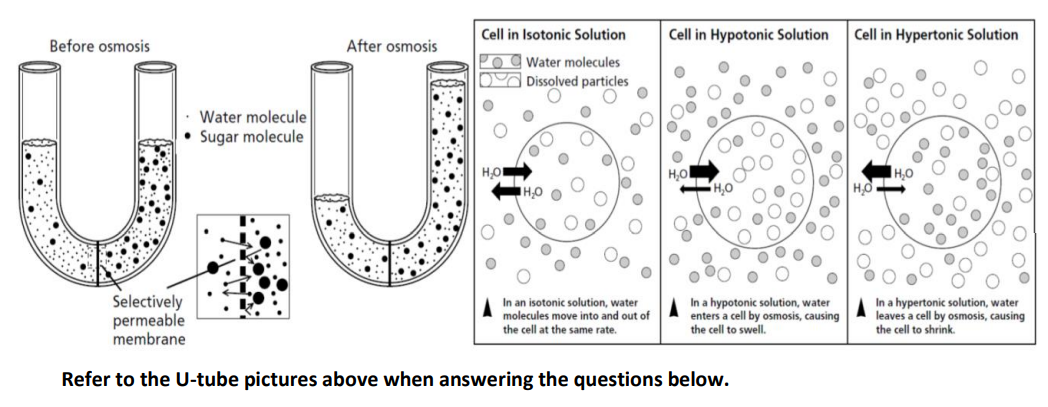
Step by step
Solved in 2 steps

- In osmosis, water flows from hypertonic/hypotonic/isotonic solutions to hypertonic/hypotonic/isotonic solutions, until both solutions are hypertonic/hypotonic/isotonic.A phospholipid bilayer separates two chambers. The chambers both contain the same volume of water. The left chamber contains 150 mM of an unknown solute A. Solute B is dissolved in the right chamber also at a concentration of 150 mM. Therefore, the initial osmolarity is the same (isotonic) between the two chambers. However, after a short time, the volume increases substantially in the right chamber. What is the most likely ranking of permeability for the molecules to cause this change? (greater permeability means that the molecule moves first across the membrane)Type of Solutes Non-permeable Permeable Solution A 300 0 Solution B 295 20 Solution C 270 30 A cell with an internal osmolarity of 295 mOsm/L (non-permeable solutes) is placed in solution A. Solution A has an effective osmolarity that is ____________ compared to the cell; after water movement has reached a steady state, the cell volume will be ___________. A) hypertonic; decreased B) hypotonic; increased C) isotonic; decreased D) hypertonic; increased A cell with an internal osmolarity of 295 mOsm/L (non-permeable solutes) is placed in solution B. Solution B has an effective osmolarity that is ____________ compared to the cell; after water movement has reached a steady state, the cell volume will be ___________. A) hypotonic; decreased B) hypertonic; decreased C) hypotonic; increased D) isotonic; unchanged A cell with an internal osmolarity of 295 mOsm/L (non-permeable solutes) is placed in solution C. Solution C has an effective osmolarity that is ____________ compared…
- Plasma membrane is permeable toa) ATPb) Glucosec) K+d) Urea1) a) Draw arrows to indicate which direction Na+ vs K+ions will tend to move.(into vsout of the cell) b) What prevents these solutes from diffusing across the membrane at a significant rate? c)If the solutes were unable to move across the membrane, would the cell be considered hypertonic, hypotonic or isotonic? d) If the solutes were unable to move across the membrane, which way would water move?(inside the cell, outside the cell or no net movement) e) How did the cell establish the differential in the shown ion concentrations in the first place?Given the following cell type and solutions, draw the cell, showing the net direction of osmosis (water flow) and say what state the cell will be in. Choose from the following terms to describe the state of the cell: Turgid, flaccid, plasmolyzed, crenulated (shriveled), lysed (burst), normal. An animal cell with an internal osmolarity of 0.32 M (total concentration of all the solutes in solution) that is placed in a solution of 0.32 M NaCl.
- When a cell is placed in a hypertonic solution,a. solute exits the cell to equalize the concentration on both sides ofthe membrane.b. water exits the cell toward the area of lower solute concentration.c. water exits the cell toward the area of higher soluteconcentration.d. there is no net movement of water or solute.Describe the chemical composition of membranes. What are the two most critical functions of membranes? What contributes to membrane fluidity and rigidity? Explain Diffusion. What are the differences between Osmosis and Dialysis? Explain the differences between Iso, Hypo, and Hypertonic conditions. If the solutions is Hypertonic then the cell would be?When a cell is placed in a hypotonic solution,a. solute exits the cell to equalize the concentration on both sides ofthe membrane.b. water exits the cell toward the area of lower solute concentration.c. water enters the cell toward the area of higher soluteconcentration.d. there is no net movement of water or solute.
- what do osmosis,diffusion,filtration and the movemenet of ions away from like charge all have in common ? in what way do the differ ?Osmosis is water movement across a semipermeable membrane. Which of the following is true about water movement across cell membranes? A. In a hypotonic solution, cells will swell. B. In an isotonic solution, cells will shrink. C. In a hypertonic solution, cells will stay the same. D. Cells can neither shrink nor swell because water cannot penetrate the plasma membrane.41. A TRANSPORT MECHANISM ACROSS A MEMBRANE WHERE THE MOVEMENT OF THE SOLUTION IS FROM HIGHER TO LOWER CONCENTRATION UNTIL SUCH TIME THAT EQUILIBRIUM IS ESTABLISHED?A. OSMOSISB. FREE DIFFUSIONC. FACILITATED ABSORPTIOND. ACTIVE TRANSPORTE. DIALYSIS 42. BIOMOLECULES ARE HYDROLYZED FIRST INTO THEIR SIMPLEST FORM BEFORE THEY CAN BE ABSORBED BY THE TISSUES. GLUCOSE ABSORPTION IS THROUGH WHICH OF THE FOLLOWING?A. OSMOSISB. FREE DIFFUSIONC. FACILITATED ABSORPTIOND. ACTIVE TRANSPORTE. DIALYSIS 43. A TRANSPORT MECHANISM THROUGH MEMBRANE WHICH NEEDS ATP?A. OSMOSISB. FREE DIFFUSIONC. FACILITATED ABSORPTIOND. ACTIVE TRANSPORTE. DIALYSIS 44. TRANSPORT MECHANISM ACROSS MEMBRANE OF GASES?A. OSMOSISB. FREE DIFFUSIONC. FACILITATED ABSORPTIOND. ACTIVE TRANSPORTE. DIALYSIS 45. A TRANSPORT MECHANISM IN WHICH SMALLER PARTICLES PASSES THROUGH MEMBRANE BUT NOT THE BIG ONE?A. OSMOSISB. FREE DIFFUSIONC. FACILITATED ABSORPTIOND. ACTIVE TRANSPORTE. DIALYSIS

