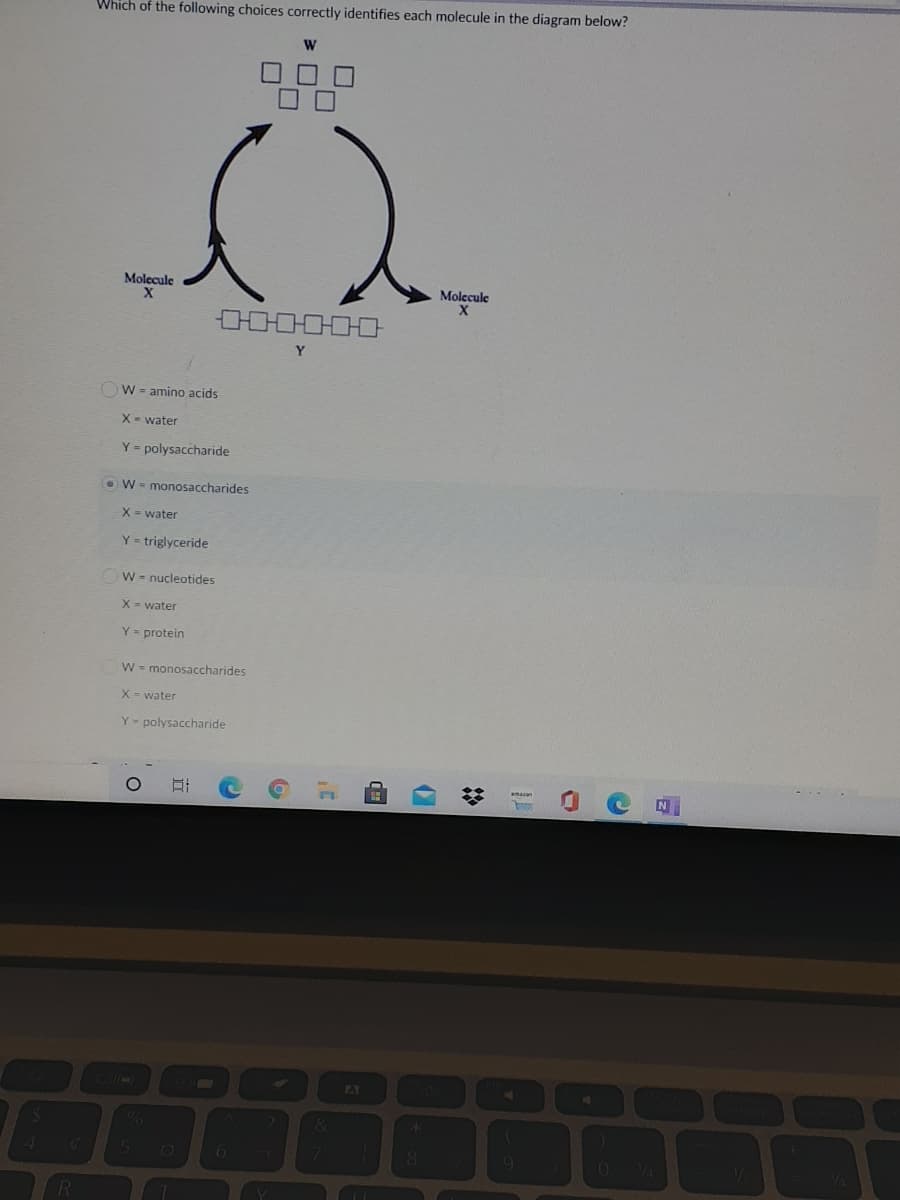
Which of the following choices correctly identifies each molecule in the diagram below? Molecule Molecule
Q: number of molecules in 1.0 micromole of water
A: 1.0 mole of water = 6.02 X 1023 molecules
Q: Why is water considered the universal solvent? A:Water molecules are weakly bonded to other water…
A: Biomolecules are those organic compounds that are made up of carbon and hydrogen with some other…
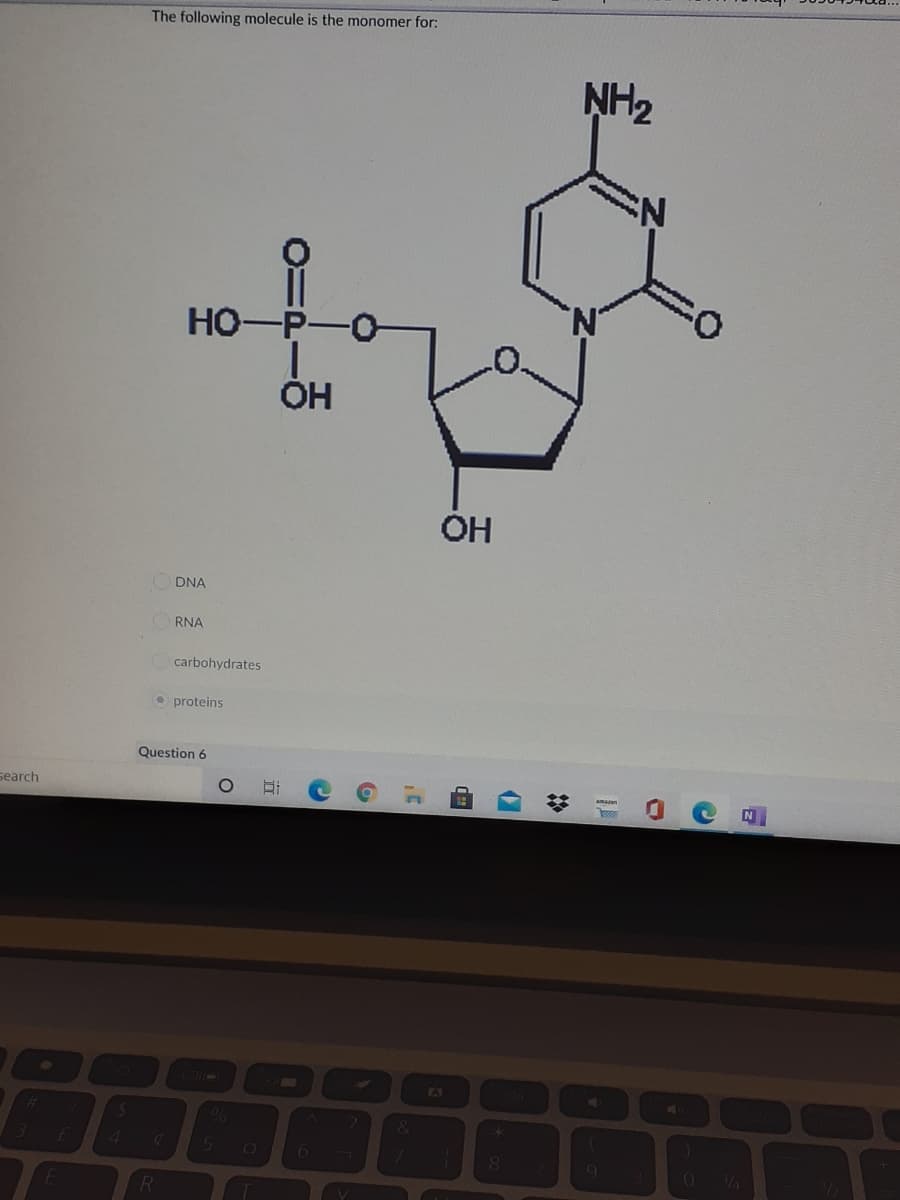
Q: A biological molecule is shown here. What is the most likely classification of this molecule? HO…
A: The four classes of biological macromolecules are nucleic acids, carbohydrates proteins and lipids.…
Q: Examine the water molecule in figure 2.14 of the textbook . What type of bond occurs between each…
A: The compounds and molecules are formed by combining one type or more than one type of atoms through…
Q: Explain the each of the following properties making water important to life
A: Water is a molecule composed of three atoms, two hydrogen atoms and one oxygen atom. It is an…
Q: Solid particles from broken apart plant and animal cells suspended in water. A. Jellies B. Foam…
A: We are authorized to answer one question at a time since you have not mentioned which question you…
Q: Please select the correct statement below. 1. Polar molecules can interact with water via hydrogen…
A: Biomolecules are organic molecules made up of mainly carbon and hydrogen but there are other…
Q: Which of the following is not a characteristic of water due to hydrogen bonding? Solvent Adhesion…
A: Hydrogen bonding : Hydrogen bonding is primarily defined as a type of electrostatic force of…
Q: Water molecules are characterized by several properties, which of the following is not a property of…
A: Water is colourless, odourless and tasteless liquid essential for all living organisms. The boiling…
Q: Define the following terms:a. saponificationb. wax esterc. waxd. phospholipide. polar head group
A: Lipids are molecules that contain hydrocarbons and make up the building blocks of the structure and…
Q: Explain the process that is occuring in
A: According to Central dogma of Molecular Biology, information flows from DNA to RNA and then two…
Q: Which one of the following terms best describes a phospholipid. It consists of а: A. polar head and…
A: All plasma membranes contain proteins as well as lipids and the basic structure of the plasma…
Q: Which of the following is not composed of repeatingsubunits?a. starchb. proteinc. nucleic acidd.…
A: Biological molecules like nucleic acids (deoxyribonucleic acid (DNA) and ribonucleic acids (RNA),…
Q: Which of the following outcomes is most likely to occur if pure phospholipids are added to water?…
A: A phospholipid molecule has two hydrophobic fatty acid tails and one hydrophilic phosphate moiety,…
Q: Example Elements Found in the Molecule Function Biological Macromolecule Carbohydrates Protein EDUC…
A: Biomolecules are organic macromolecules that play an important role in the proper functioning of the…
Q: When a hydrophobic substance like a hydrocarbon is dissolved in water, aclathrate cage of ordered…
A: When randomness of molecules increases, entropy also increases. When randomness decreases, entropy…
Q: Cells are composed of chemicals and both the structure and function of cells are regulated by the…
A: Both living and nonliving cells obey the same laws of chemistry and thermodynamics.
Q: Explain why two identical atoms joined by a covalent bond cannot form a hydrogen bond.
A: Electronegativity, is the tendency of an atom to attract shared electrons (or electron density) to…
Q: Which of the following statements is NOT true of biological organic molecules? None of the other…
A: A complex molecule that is primarily made of carbon atoms bonded with other elements and/or other…
Q: what is the basic unit of matter
A: Anything that has mass and is capable of occupying space is considered as matter. Different or…
Q: Which molecules is useful for holding proteins and fats in suspension with eachother? O…
A: Fats and lipids are considered as the fuel source, which provides high calories, which is utilized…
Q: In this gel image, click on the band that represents the molecule that is largest in size (mass).
A: Gel electrophoresis is a technique used to separate different pieces of DNA on the basis of their…
Q: What has occurred to this molecule to cause it to change as shown in the diagram? Covalent bonds…
A: Double-stranded DNA is the genetic material present in most organisms. In a double-stranded DNA, two…
Q: Label the different types of phases shown in the picture
A: Mitosis Mitosis is the division phase of a cell cycle. In this phase of the cell cycle, the cell…
Q: At which pH would the structure shown be predominate? H₂N-CH- - H OpH 1 pH 3 pH 8 pH 11 0 0
A: Glycine has the simplest structure among all the amino acids. This side chain of amino acid has most…
Q: Which one of the following is universal solvent? Select one: Ethanol Acid Water Methanol
A: A solvent is a substance that has the ability to dissolve solutes. The mixture of solvent and solute…
Q: ch the terms with the labeled structure in the image.
A: Greater Vestibular Gland - O Mons Pubis - J Labia - N Vagina - H Ovary - C
Q: Many characteristics of water make life possible. Which of the following is NOT one of the reasons…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Refer to figure 2.15 to draw a picture of on NaCl molecule interacting with several water molecules.…
A: A chemical bond: It is a bonding between the atoms or ions due to the force of attraction. For…
Q: Which structure is formed when fatty acid salts reacts with water? O Clathrate O Micelle O Monolayer…
A:
Q: How can you tell what type of molecule something is by it is spelling?
A: Atoms bond together to form molecules. Prefixes and suffixes are used to tell the type of molecules.
Q: Organic molecules are often assembled by polymerization. Which is an exception to this statement? a…
A: A polymerization is a chemical event in which relatively small molecules, known as monomers, produce…
Q: Rg thế chârt above, what type of molecules (carb, lipjd, nucleic acid or protein) are these? 12…
A: Macromolecules are large biomolecules such as proteins, carbohydrates, lipids, and nucleic acids.…
Q: part of the structure (see
A: the image refers embryology of digestive system formation of primitive gut primitive gut tube is…
Q: Water molecules Dissolved particles Water molecule Sugar molecule HO H,O H,O
A: If cell is places in a solution Isotonic to the cytoplasm of the cell, then osmosis doesnot occur…
Q: Water is an example of a compound. Which of the following is the best description of all compounds?…
A: INTRODUCTION Water is an inorganic, transparent, tasteless, odorless, and nearly colorless chemical…
Q: `N*
A: The plasma membrane is a lipid bilayer structure that encloses the content of the cell of the…
Q: Which of the following is correct about the properties of water? Three of the qualified choices…
A: Water is also a chemical compound that is composed of one atom of oxygen and two atoms of hydrogen.…
Q: hich molecule has the most atoms?
A: N2O2 has 4 atoms N=2, O=2 CO2 has 3 atoms., C=1, O=2 CO=2 atoms., C=1, O=1 H2O=3 atoms., H=2, O=1
Q: A. Fill in the table by comparing the following biological molecules. Biological Molecules Basic…
A: Every living organisms have cells which is the smallest structure and functional unit of the…
Q: Which of the following combinations describes the electron in the modern model of an atom? Onegative…
A: Atom It is a particle of matter that constitutes the nucleus and the orbits. The nucleus contains…
Q: name the class of large biological molecule for the following molecule. circle & label an example of…
A: Testosterone is a male hormone produced by the human body. Dihydrotestosterone is a naturally…
Q: Use the following diagram to answer the question: Which of the molecules have hydrogen bonding? (B…
A: In the given images, we are shown different biomolecules. There are many biomolecules present in the…
Q: sing the graphic above, answer the following questions. Vhich of the substances above has the…
A: pH (power of hydrogen) is used to know the acidic or basic level of a particular liquid or compound.…
Q: I' H I "H HO Choose all of the following are true of the given structure? (Choose all of the correct…
A: The major biological macromolecules are nucleic acid, proteins, lipids and carbohydrate. The four…
Q: The molecule shown below is NH2 HO N. Но но
A: Deoxyribonucleic acid (DNA) is a molecule comprised of two polynucleotide chains that wrap around…
Q: What is the name of the biological molecule that provides instruction the cells of all living…
A: Biomolecules or biological molecules are those which are found in living organisms.
Q: The questions in the left hand column correspond to the diagram in the right hand column. Read each…
A: Shown molecule is a disaccharide made up of 2 sugar monomers.
Q: Explain in detail why water molecules are polar. Your explanation can be as long as you wish. Your…
A:
Step by step
Solved in 2 steps

- the tripeptide is ILYwhy is CH3-CH-CH2-CH2-CH3 non imposable ? I OHWhat does it mean for an amino acid to be polar? Please explain what qualities about an amino acid might make the amino acid polar. The disease known as sickle cell anemia is due to a genetic mutation which replaces glutamic acid, a polar amino acid with valine. Please explain how this affects the hemoglobin.
- Which abbreviation is this following peptide? GKH QGR HK GHK GHLDraw leucine and identify any chiral carbon atoms with arrows.The following sequence of amino acids are part of adult hemoglobin from the N-terminus. Draw the skeletal structure of this peptide. Val-His-Leu-Thr-Pro-Glu-Glu-Lys-Ser-Ala-Val-Thr
- D-Erythrose and D-threose are referred to as _____________ because their structures only differ in the configuration at one asymmetric carbon atom.How does sickle cell hemoglobin differ from normal hemoglobin at the fourth level of protein structure (the sum of all the folded protein chains)?draw the following amino acid chains and give the single and three letter abbreviations for cysteine
- What amino acid residue interacts with molecular oxygen (O2) in hemoglobin? a. Phenylalanine b. Serine c. Valine d. Histidine1-33. Indicate whether each of the following statements about stereoisomers is true or false.How does sickle cell hemoglobin differ from normal hemoglobin at the quaternary level of protein structure (the sum of all the folded protein chains)?

