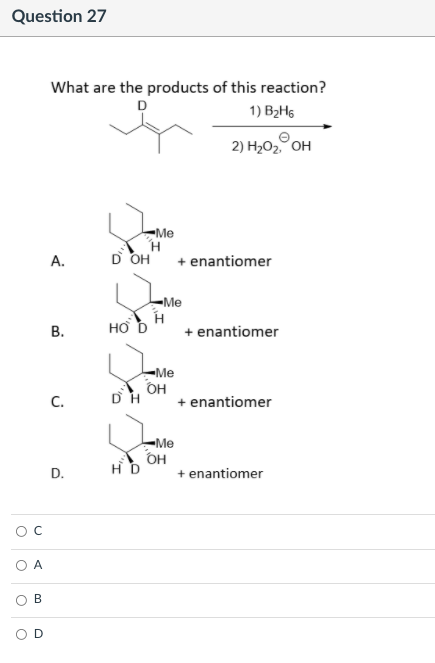
What are the products of this reaction? D 1) B2H5 2) H2O2 OH "Ме A. D OH + enantiomer -Me В. но ъ + enantiomer -Me OH + enantiomer С. -Me OH HD + enantiomer D. O A B.
Q: (E)-4-methyl-2-pentene + HBr 1-methylcyclohexene + ICI (hint: treat ICI as Br2) HO…
A:
Q: (a) (b) (c) ОН -OH OH ? -CH₂OH ? (d) О CHOH CH₂OH ? ? moilosot not in ее 2010 о о (e) H (f) -CH₂CI ?…
A:
Q: What is the type of alkyl halide below? CI O a. Tertiary O b. Primary O c. Methyl halide O d.…
A:
Q: 9. Identify all functiona l groups (excluding alkanes) for the given molecule (S) b.count all…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: The reaction below is commonly used as a laboratory preparation of cyclohexene. The Forward and…
A: Chain termination step is the last step of free radical mechanism.in this step, free radicals react…
Q: 6. oraw the viructure for each compound. a. (3R)-3-methylnexane 6. (4R, 55)-4,5- diethyl octane 4) 1…
A:
Q: CrO3 CH3CH2CH2OH CH3CH2COH CH3CH2COCH2CH2CH3 A problem often encountered in the oxidation of primary…
A: Ester produced as a by-product because the propanol reacts reversibly with propanoic acid and the…
Q: CH,OH b. СНО CH,OH CHO a. с. C=0 H-C-OH C-O H-C-OH Н-С-ОН Н-С-ОН Н-С-ОН Н-С-ОН CH,OH Но-С-Н Н-С-ОН…
A: The saccharides containing an aldehyde group are called Aldoses while those with a ketone group are…
Q: What is/are the product(s) of this reaction? 1. Hg(OAc)2 CH;OH 2. NaBH, HO enantiomer OCH3 OCH3 OCH3…
A:
Q: 7. Choose the correct option. * 1 point NO2 AIC13 CI Major product + H;C CH3 H;C CH3 D) NO2 O NO2 B)…
A: Nitrobenzene does not give friedel craft reaction with acyl cation due to presence of nitro group in…
Q: H-C三C- H 2) 1- bromo-5-methyl hexone 1) NANH2 / /qued NHs/-78 °C A (CgHI6) 2) 1-bromodecane P-2 (Ne,…
A: reactions
Q: CH3CH2NH2 (с) 2 eq CH3CH2OH (d) H30* (е) -CH2-CH2-C-H КKMnOд, cold dilute
A: The given reactions are some of the well established reactions exhibited by carbonyl functionality.
Q: ALKENE/ ALKYNE REAGENTS(S) AND MAJOR PRODUCTS(S) CONDITIONS ? OH
A:
Q: lo i. LIAIH4 i. deprotection CH2=CHCOCH3 protection ii. H3O* ii. dehydration K The synthesis of K…
A: We can protect ketone group by the help of ethylene glycol.
Q: What is the structure of B? and of C? H CH3 H3O* H20 B + H CH3 1) ВНз-THF CH3 2) H2O2 HO H20 H CH3
A: The product are of the reaction are B = Tertiary butyl alcohol C = 2-methylpropan-1-ol
Q: What is the product? OCH3 1. DMSO, (CICO)2 HO, 2. EtgN, CH,Cl, CH, CH.
A:
Q: Order the following in increasing priority A. -C, -CH, -OL B. -CH3, -CH2OH, -CH2CH3 C. -C≡CH,…
A: During nomenclature of molecules or while assigning R/S nomenclature of an organic molecule. The…
Q: BY BY2, hD NOOH Cae) ( Cyclopentane) SM2 он pcC, THE BY BY2 ACOH t'Buok JEBuott E2 Reoction ( dip -…
A: In the first step bromination takes place in presence of sunlight with the production of free…
Q: (a-b) (c-d) (e-f) OH CI CI HORE ? NH pyridine -OH HAM S CI LIAIH[OC(CH3)3]3 1. LIAIH4 2. H₂O+ 1.…
A: Lithiumaluminiumhydride is a reducing agent. It release hydride ion (H-). According to Q&A…
Q: он а. HNO3, H2SO4 b. SO3, H2SO4 с. СН3СH2CH2CІ, AICI3 d. (CH3CH2)2CHCOCI, AICI3 e. Br2, FeBr3 f.…
A:
Q: (b) Complete the following reactions : W (i) H3C D /H CH3 NO₂ og H B 09 Br₂/Dioxane COCH3 A
A: Indole show Electrophilic Aromatic substitution reaction.
Q: CIH CIH CIH 27. What is the monomer used to prepare -ċ-ċ-Ċ-Ċ-Ċ-Ċ- H CHH CHH CH; Oa. CH2=CHCI Ob.…
A: Cut the repeating unit and put the double bond to get monomer.
Q: .CH2CH OH ACID OH ACID ? CH3CHCH3 CH3CHCH3 ? САTALYST САTALYST ОН OH H,SO4 i) CH;CHCHCH2CH2CH3 +…
A:
Q: IV. 1. 2. 3. H Fill-in the missing the reactant, reagent, or major product. 1. Hg(OAc)2, H₂O 2.…
A:
Q: (CH2 Kuli DNOOHA 2) Atdal condonaten of From fragments of 4 carbons or fewer 3) HoD
A: Aldol condensation is the condensation between two carbonyl compounds to give a β-hydroxy carbonyl…
Q: what Ps the UPAC name: OH a) 3-methylpent-1-en-5-01 3-methylpent-s-en-1-01 3-methylpent-4-en-)-01 8)…
A:
Q: 3) Draw the ECSF structures of (CH3)3CO® (tert-butoxide ion) and CH3O® (methoxide ion). Why is…
A:
Q: Identify the type of reaction: CH3-CH2-CH2-CH2-CH3 + 802 5CO2+6H2O+energy
A: The given reaction is, CH3-CH2-CH2-CH2-CH3 +8 O2 -----> 5CO2 + 6H2O + energy In the above…
Q: Reaction Of ALIPHATIC AL COHOL conc H2SO4 CH;COOH conc H2SO4 + reflux CH3 Na CH;ĊCH,CH3 OH + PCI5…
A:
Q: C1H CI H CIH 27. What is the monomer used to prepare c-c-c-C-C-C- H CHH CHH CH; Oa. CH2=CHCI Ob.…
A: Correct option is C.
Q: The answer choices give me chemical reactions like: C6H11FO3S + C5H11+ + H2O ---> C5H10 + C5H11+ +…
A: Given reaction is We need to write overall reaction
Q: CH3 + KMNO4 CH3 CH2 85% H¿PO4 CH3 CH3 Q2. In the Preparation of Alkenes from alcohols the acid (H,A)…
A: we have to draw the product:-
Q: Draw the structure for an alkene that gives the following reaction product. 1. Hg(OАc)2, Н2О ?…
A:
Q: Addition of HCl to alkene X forms two alkyl halides Y and Z. Label Y and Z as a 1,2-addition product…
A: To find: 1,2 addition and 1,4 addition product of the given reaction
Q: 3. What is the configuration of the alkene PAUL? PERO. cis PEOR. trans HO POER. E REPO. Z PAUL
A:
Q: CH- C- CH, + Clh →ら-CH2C) CH3 63% 1 CHS Why this still CH;-C-CHs CH3 is the major 37% product?
A:
Q: Name the alkene below. Use ONLY E/Z designators to indicate stereochemistry. H H H3C CH3 CH;CH2CH2…
A:
Q: H2NOC CONH2 CONH2 R N= H2NOC Co 'N N. H2NOC .CONH2 NH НО O. O=P- ОН R = 5'-deoxyadenosyl, CH3, OH,…
A:
Q: 2n-BuLi ThE 30 c° NHCOZ-Buc 2+-1B4 me thent mel-70 54% CHO 79% 5-azaindole whate the Mechanits is
A:
Q: Q. The final product of this synthesis is a non-steroidal anti-inflammatory drug (NSAID). It has…
A: At first chlorination occurs to give chlorobenzene. Then nitration gives ortho nitro chlorobenzene…
Q: 9. Classify the following reactions as addition, elimination, substitution or rearrangement: KOH a)…
A: a .It's elimination reaction .KOH being strong base it abstracts H+ and Cl-leave simultaneously…
Q: Explain why the addition of HBr to alkenes A and C is regioselective, forming addition products B…
A:
Q: 2Me hv .CO2Et RT "CO,Me Ar ELOH R Ar 2 Ar- CO2ET 1 R. CO,Me R. CO,Me CF3SO3H CH2CI2, A RT CO2M CO2ME…
A: Given, When succinate 1 is irradiated in EtOH, it produces cis-dihydronaphthalene (2). When…
Q: tehich of Ahe followsng has the Jorrgest mumber of n कटपक कड ?हतविल C1) phoseho80u S-30 xp laim (1)…
A: Atomic number = number of proton Number of proton = Number of electron Number of neutron = mass…
Q: Determine the product of the ff. rxn Br OH- -N. CH3 А. В. С. D. Br OH -N. CH3 -N. CH3 N-
A:
Q: Kenalpasti sebatian A hingga E. Identify compound A to E. В H2SO4 но NH2 1) A D 2) H,O* NAOH PCC,…
A:
Q: 2014 male luler Yomul nach paoden Hnanle , A= 271 (s) Comprunds ond Y yhoore he Compound X has 8-…
A: This Question is based upon NMR Spectroscopy.
Q: H2/Ni cis but-2-ene
A: Given,
Q: Identify A, B, C, and D CHy-CH-C-CI so, AICI3 H,SO, B Pd/H2 COOH Br
A:
Q: ethane + H. alkene X + Y ethanol I3D a) State the identification of i. Alkene X; ii. Compound Y b)…
A:
Step by step
Solved in 2 steps with 2 images

- (a) What happens when CH3—O—CH<sub3 is heated with HI?(b) Explain mechanism for hydration of acid catalyzed ethene :CH2 = CH2 + HzO CH3—CH,—OHIn addition to organic halides, alkyl tosylates (R'OTs. Section 9.13) also react with organocuprates (R2CuLi) to form coupling products R – R'. When 2° alkyl tosylatee are used as starting materials (R2CHOTs), inversion of the conguration at a stereogenic center results. Keeping this in mind, draw the product formed when each compound is treated with (CH3)2CuLi.In addition to organic halides, alkyl tosylates (R'OTs. Section 9.13) also react with organocuprates (R2CuLi) to form coupling products R – R'. When 2° alkyl tosylatee are used as starting materials (R2CHOTs), inversion of the configuration at a stereogenic center results. Keeping this in mind, draw the product formed when each compound is treated with (CH3)2CuLi.
- When (CH3CH2)3CBr is added to CH3OH at room temperature, the product is(CH3O)C(CH2CH3)3. Propose a mechanism(s) for the reactions leading to theseproducts and use curved arrows to show the movement of electrons.Why does the alpha hydrogen to a ketone have a lower pka value than the alpha h to an alkene. Draw a picture to explain ur a waysThank you for the answer, would it be possible to follow the path I have attached as well srarting from methanol ---> CH3Br -----> CH3MgBr-------> (H3C)CH-CH2OH ----> E1 Isobutalene
- 2. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 3Draw all of the substitution and elimination products formed from thegiven alkyl halide with each reagent: (a) CH3OH; (b) KOH. Indicate thestereochemistry around the stereogenic centers present in the products,as well as the mechanism by which each product is formed.a) Which reaction yields hexan-1-ol?
- Draw the substitution product formed (including stereochemistry) when (R)-hexan-2-ol is treated with each series of reagents: (a) NaH, followed by CH3I; (b) TsCl and pyridine, followed by NaOCH3; (c) PBr3, followed by NaOCH3. Which two routes produce identical products?When (CH3CH2)3CBr is added to CH3OH at room temperature, the product is (CH3O)C(CH2CH3)3. Propose a mechanism(s) for the reactions leading to these products and used curved arrows to show the movement of electrons.Alkyl halides undergo elimination reactions with Brønsted–Lowrybases. The elements of HX are lost and an alkene is formed. Explain this ?

