Q: Parts per million (ppm) is used with very low concentrations in solution. The formula in getting…
A: The question is based on the concept of solutions. PPM is a method of expressing concentration of…
Q: [References] INTERACTIVE EXAMPLE Calculating Temperature Using Charles's Law A gas occupies 4.48 L…
A: We have : Initial Temperature (T1) = 43.3 °C Volume (V1) = 4.48 L Final Temperature (T2) = ?…
Q: QUESTION 26 In the IUPAC name for the following compound, the -Br group is located at what position…
A: According to IUPAC rules the first locant any group or multi bond should get minimum number in IUPAC…
Q: ffer solution is prepared by combining 0.848 moles of formic acid and 0.719 moles of sodium formate.…
A: Mole formic acid= 0.848 Mole sodium formate = 0.719 Mole of NaOH = 0.126 Ka = 1.8×10-4. Pka=…
Q: 1. LIAIH4 2. H₂O* 3. SOCI₂
A: Lithium aluminum hydride reduces the carbonyl compounds into alcohols. In this reagent, a hydrogen…
Q: a) b) Fill in the reagents above/below the arrow in the following reactions. c) CN d) CO₂H LỊCH OH…
A: The different sets of reagents lead to the formation of different products from the same starting…
Q: Calculate the radius ratio of ZnO and AgI and predict its crystalline latice.
A: The radius ratio is a dimensionless quantity that is calculated by dividing the radius of the cation…
Q: How many chemically inequivalent carbons are there in this molecule? Select an answer and submit.…
A: Since you asked multiple questions so as per Q&A guidelines of portal I solved full questions…
Q: For each of the following complex ions: a) [Ni(SCN)2(H₂O)2] b) [Mn(acac)2Br2]²- (i) (ii) (iii) (iv)…
A:
Q: HO. N₂ IZ N .Me Me
A: Name of the functional group attached to the aromatic ring ?…
Q: Electrolysis is the process of using electrical current to cause a nonspontaneous reaction to occur.…
A:
Q: 8. Identify the following reaction types (e.g. condensation, addition, etc.). Y- Y OH CI F2 300 80…
A: The type of reaction depends upon nature of substrate and nature of reagent.
Q: Classify each of the following as a strong acid or a weak acid. Indicate how each should be written…
A: Strong acid completely dissociates while weak acid partially dissociate in aqueous solution. 1)…
Q: Formula: CgH140 108 80- 89 48- -83 4080 3500 CB 8962 2918.87- 3880 2880.88 Spectroscopy Reference…
A: The question is based on the concept of organic spectroscopy. we need to analyse the spectral data…
Q: 12 3 2.2 2.1 2.0 1.9 1.8 1.7 1.6 50- IR 100+ 2 4000 1 integration 11 10 9 3000 00 8 2500 7 2000 22…
A: The question is based on the concept of organic spectroscopy. we need to analyse the spectral data…
Q: ant stalo Assign each set of protons (A-F) to the NMR peaks. 4.5 2H 4.0 2H 3.5 3.0 ethyl…
A: The question is based on the concept of organic spectroscopy . we need to assign fragments to…
Q: EA 27 A) the most stable conformation of the Identify following compound. CH₂CH3 CH 3 B)
A: In substituted cyclohexane , the bulky group tend to occupy equatorial position to avoid 1,3 diaxial…
Q: Please plot this in excel as a smooth line scatter chart with the Volume on the X axis and the pH on…
A: The graphs that show the association between two variables in a data set are called scatter plots.…
Q: Identify the type of solution produced in a solution that has 100 g of water as its solvent based on…
A: The question is based on the concept of solubility. solubility follows a basic principle "like…
Q: draw the structure of glutamic acid/glutamate at a pH of 5.
A:
Q: A chemist titrates 150.0 mL of a 0.3588 M trimethylamine ((CH3),N) solution with 0.2376M HBr…
A: we have to calculate the pH of solution at equivalence point
Q: A slightly soluble salt is added to water and a small fraction of the solid dissolves. Once the…
A: A sparingly soluble salt is dissolved in water, we need to discuss about how the equilibrium behaves…
Q: A 5.00 g quantity of a diprotic acid was dissolved in water and made up to exactly 2.25 x 10² mL.…
A:
Q: IZ NÄY LiAlHy (Ht workup)
A: LiAlH4 is a strong reducing agent that reduces amide (-CONH2 ) to amine (-CH2NH2)
Q: What is the solubility of PbCl₂ in 0.020 M NaCl? Ksp = 1.59 x 10-5 Keep 3 total significant figures.
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: The poison strychnine is a weak base with Kb = 1.8 x 106. What will be the equilibrium pH of a…
A:
Q: Check the box under each compound that exists as a pair of mirror-image twins. If none of them do,…
A:
Q: EA 33) What are the configurations of carbons 2 and 3 respectively in the Fischer projection shown ?…
A:
Q: Questions 1-4 refer to the same strong acid/strong base (SA/SB) titration. A 25.00 mL solution of…
A: This question is based on the titration of strong acid and strong base where as we keep adding Base…
Q: is the Arrhenius definition of an acid and a base? O a An acid contains hydrogen; a base contains a…
A: The concept of Arrhenius acid base is applicable for only aqueous solution.
Q: What is the pH of a solution that is 0.855 M in hydrosulfuric acid (H₂S)? Ka1 = 8.9 x 108 and Ka2 =…
A:
Q: EA 28 H H CH3 H H H CH3 H The structure on the left is:
A: Cyclohexane is a cycloalkane that contains 6 carbons.
Q: For a particular redox reaction, SO3 is oxidized to SO2 and Cu2+ is reduced to Cu*. Complete and…
A: Redox word is the combination of two word, reduction and oxidation, therefore, any reaction in which…
Q: Consider the pair of reactions. Draw the organic products, then predict the type of substitution…
A:
Q: Mercury oxide (HgO) readily decomposes when heated. If it takes 90.8 kJ to decompose one mole of…
A:
Q: Calculate the composite modulus for polyester reinforced with 30 vol % Al2O3 whiskers under…
A: Given = E polyester = 6900 MPa % polyester = 100-30= 70 % E Al2O3 = 430*10^3 MPa % Al2O3 = 30%…
Q: Consider these two entries from a fictional table of standard reduction potentials. X²+ (aq) + 2e →…
A: Species having higher value of reduction potential has higher tendency to get reduced. While…
Q: Questions 1-4 refer to the same strong acid/strong base (SA/SB) titration. A 25.00 mL solution of…
A: Equivalence point is a point at which enough base is added to completely neutralize acidic solution.…
Q: Calculate the molar solubility of Fe(OH)₂ in a solution buffered at pH 9.00. Be sure your answer has…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: Which of the following reagents would NOT convert the shown alcohol into the alkene a) 1) TsCl, py…
A:
Q: 124 Does the [CoBr214]³ complex have an optical isomer? W Using the valence bond theory and hybrid…
A: A compound will only be optically active if it doesn't have plane of symmetry , center of symmetry…
Q: Identify which two alcohols are needed to make the ether shown here. CH-CH2-CH2-O-CH-CH, CH3 CH3 A)…
A: Williamson ether synthesis: Alkoxide reacts with alkyl halide to form ethers. Alkoxide ion is…
Q: 8. Identify the following reaction types (e.g. condensation, addition, etc.). CI I OH dilute NaOH…
A: The basic organic reaction types are addition, elimination, substitution, and rearrangement…
Q: Check the box under each compound that exists as a pair of mirror-image twins. If none of them do,…
A: The compounds with the same molecular formula but different bond connectivity are structural…
Q: Questions 15-20 refer to the same weak base/strong acid (WB/SA) titration. The K₁ of pyridine is 1.7…
A: The question is based on the titrations. we are titrating a weak base with a strong Acid. we need…
Q: 5. Balance the following redox reactions in the given conditions. You do not need to show all of…
A:
Q: During a calorimetry experiment, the specific heat of a metal is unknown. A hot piece of metal is…
A:
Q: 32. Which one of the following is not a part of osmosis? a. osmotic pressure c. overall flow of…
A: Osmosis: The process in which solvent from solution of lower concentration is transferred to…
Q: What are the products of a reaction between an acid and a base? O a Carbon dioxide and a salt O b…
A: The reaction between Acid and base is known as Neutralization reaction.
Q: In the analysis of the kinetics of the reaction A+B → products, which follows the reaction rate law…
A: Answer: Rate law given for the reaction is: ν=k[A]a[B]b Here: ν=rate of reactionk=rate constant…
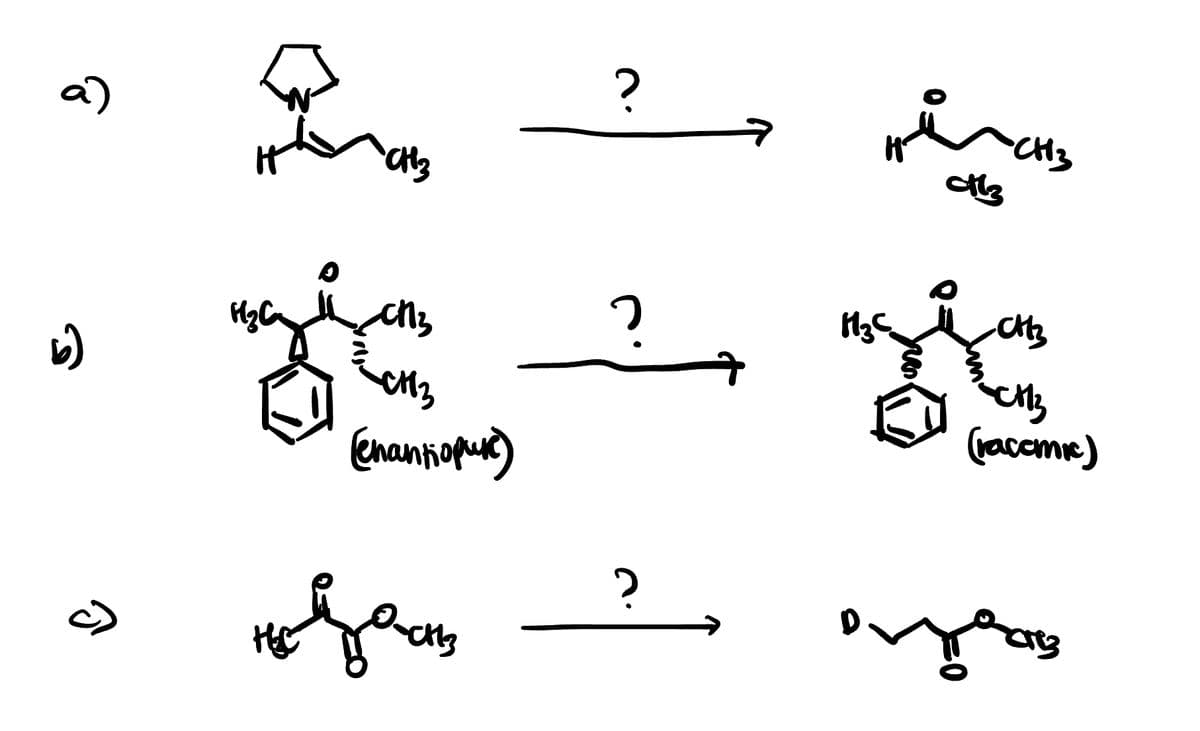
What are the reagents?
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images


