Q: A chemist designs a galvanic cell that uses these two half-reactions: standard reduction potential…
A: In an electrochemical cell, the reduction of the chemical species takes place that is having the…
Q: Part E How many pi bonds are present in the molecule below: Πν ΑΣφ ? Submit Reguest Answer
A:
Q: What is the order of reactivity for the following compounds toward reaction with methylmagnesium…
A: In Grignard reaction, the nucleophile, CH3- attacks the partially positively charged C of -C=O of…
Q: Consider a collection of N, rubidium-87 (rubidium is atomic number 37) atoms confined inside a cube…
A: Bose-Einstein condensation is a matter of state in which most of the particles (mainly Bosons)…
Q: Which of the following molecules is responsible for this IR spectrum?
A:
Q: Answer the following chemical reaction 1. Iron reacts with oxygen gas and forms solid iron(II)…
A: Reactants are the substances which are present at the start of any reaction. Products are the…
Q: structure
A: In iupac name ,first we select longest parent carbon chain which includes main functional group
Q: 20. How many grams of Ca metal are produced by the electrolysis of the molten CaBr2 using a current…
A: The reduction reaction taking place in cathode is as follows, Ca2++2e → Ca Molar mass of Ca is 40…
Q: Which one of the following salts, when dissolved in water, produces the solution with pH less then…
A: When a salt is made up of strong acid and weak base, then acid remains excess after…
Q: Performance task 1) ermine if the decomposition of potassium chiorate (KCIO3) is spontaneous or not.…
A: Spontaneous reactions can be defined as the reactions that occur by itself without any external…
Q: 12. What occurs during the electrolysis of a molten salt? A. Electricity is produced by a…
A: Electrolysis is a process which takes place in the electrolytic cell. In this process a chemical…
Q: Which compounds will react with each other in the presence of NAOH to give the following product? oe…
A: In this question, we will see the starting materials for the give aldol condensation product. You…
Q: Give IUPAC names for the following compounds. Abbreviate ortho (0), meta (m) and para (p), no…
A: (1) Ortho-bromopropyl benzene
Q: The Lewis dot formula for NOCI, shows 1. I. three single covalent bonds. 2. II. one unshared pair of…
A: Given-> NOCl
Q: QUESTION 2 To establish a standard curve for a Blg standard curve using Bradford, the…
A: Correct option is F. Explanation: The Bradford assay is very useful and fast, but its chemistry is…
Q: он HO ČHO ABC DEF GHI
A: ->Reaction of ether with HI produce alkyliodide and alcohol. ->For strength of acids we see…
Q: For the reaction 2C03+ (aq) + 2CI- (aq)→2C0²+(aq) + Cl2 (g). E° =0.483 V what is the cell potential…
A: Cell potential is calculated using Nernst equation .
Q: O https://. 4. 10The blood brain barrier protects the brain from harmful molecules and also…
A: The answer is as follows:
Q: The following are ways to prepare aldehydes EXCEPT a) oxidation of primary alcohols b) oxidation…
A:
Q: An aqueous solution contains 0.27 M hydrocyanic acid. One Liter of this solution could be converted…
A: ->Given Molality of HCN = 0.27 M Volume = 1 L ->The buffer solution formed by it must…
Q: A sample of helium gas occupies a volume of 6.06 L at 40.0°C and 0.870 atm. If it is desired to…
A:
Q: If 27.5 g of MgCl₂ are dissolved 500.0 g of water at 20.0 °C in an insulated container, a…
A: Given that, The mass of the MgCl2 is 27.5 g. The mass of the water is 500.0 g. So, the total mass of…
Q: Can you make solutions for numbers 1-3? like the one that shows the oxidation number of every…
A:
Q: 27. The value of E° for the following reaction is 0.189 V. What is the value of Ecell given the…
A: Given the standard cell potential, Eo = 0.189 V [H+] = 0.1 M [NO3-] = 0.6 M [Fe3+] = 1.0 M [NO] =…
Q: oues- ow mamy moles of HCI ane in 53.4mL of a L.ll M HeL solution 2
A:
Q: overall reaction in an electrochemical cell i
A:
Q: 27. The value of E° for the following reaction is 0.189 V. What is the value of Ecell given the…
A: Here we have to find Ecell of the following Galvanic cell.
Q: number of moles
A:
Q: 10. What is the standard cell potential of the following electrochemical cell at 25°C: Ni(s) I…
A: 10. Given that, an electrochemical cell reaction is Ni(s)|NiCl2(aq)||Cu(NO3)2(aq)|Cu(s). We have to…
Q: Calculate the theoretical cell potential of the following cells. If the cell is short-circuited,…
A:
Q: Consider a Galvanic cell constructed from the following half cells, linked by an external circuit…
A:
Q: calculation of AG° in Kj/mol
A:
Q: Itis Known that at 120°c k = 10ms %3D -7 At 27°c Fz= 10'st Eace) = 80 kcal mot Deterrmine the…
A: As we know, for parallel reaction, we have following equations: [B]=-k1[A∘](k1+k2)e-…
Q: ANALYSIS (Performance Task # 2) 1. What does the second law of thermodynamics imply about the state…
A: 1. The second law of thermodynamics implies that the state of entropy of the entire universe, as an…
Q: For a certain chemical reaction, the standard Gibbs free energy of reaction at 5.00 °C is 142. kJ.…
A: The free energy change of the reaction is = 142 kJ/mol The temperature of the reaction is = 5.00oC =…
Q: 18. For a voltaic cell using Ag/Ag* (1.0 M) and Zn/Zn* (1.0M) half cells, which of the following…
A: This question is from Electrochemistry topic.
Q: A sample of xenon gas occupies a volume of 9.28 L at 68.0°C and 339 torr. If the volume of the gas…
A:
Q: can you double check the letter and the answer you have circled they are the opposite?
A:
Q: 26. Chromium often is electroplated on other metals and even plastics to produce a shiny metallic…
A: Here we have to determine the mass of chromium deposited by passing 10 amp current for 5.36 hour.
Q: The titration reaction: 2Ce4+ + 3I- ⇌ 2Ce3+ + I3- is an example of: Precipitation titration…
A: The titration reaction: 2Ce4+ + 3I- ⇌ 2Ce3+ + I3- is an example of - Iodometry. In the above…
Q: Calculate the average rate using the graph below. 1.2 1.0 0.8 [HO 0.6 0.4 0.2 10 20 30 40 50 60 70…
A: To calculate the average rate of the reaction.
Q: Which of the following is the major product of the following reaction? (i) CH3CH2L¡ (excess) (ii)…
A:
Q: Hello, I entered that and for some reason it's still incorrect. I'm so confused
A: Mass defect is given by the formula ∆m = [mass of protons + mass of neutrons] - [mass of atom]
Q: The reaction that forms most of the acid in acid rain is A s02 (g) + H20 (1) – H2SO3 (aq) (в) сl2…
A: Acid rain is a natural phenomenon in which rain water combines with different non metallic oxides…
Q: What is the expected product A for the following Claisen condensation reaction? 1. NaOEt H3C. A OEt…
A: Claisen condensation reaction- It is an carbon carbon bond forming reaction. It occurs between two…
Q: Question For the following coordinction (omples:(Co (H,0), CIB-3 Give the name Determine the…
A:
Q: [d] Do the same with these five words: beta, neutron, uncertainty, decay, electron.
A: [c] Write a single sentence, using all of these five words plus other words, that makes scientific…
Q: 7. Which of the following expressions correctly shows the calculation of AG° in Kj/mol for a Voltaic…
A:
Q: Nucleophile acetone + Substrate Choose from the pool of choices below: SH Br АВС DEF HS Br GHI JKL
A:
Q: Can you state what happened here in essay form?
A: Carboxylic acid reacts with an amine to form an amide. The reaction proceeds by nucleophilic…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Which product would likely be the major product for the following reaction?a. Cb. Dc. Ad. BPredict the major products of the following reactions. (a) the tosylate of cyclohexylmethanol + excess NH3(b) n-butyl tosylate + sodium acetylide, H¬C‚C:- +NaWhich product would likely be the major product for the following reaction?a. Ab. Cc. Dd. B
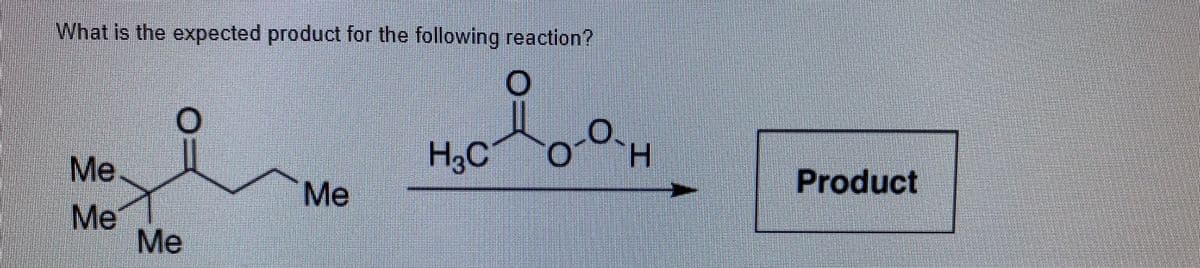
- What is the expected product for the reaction below?Propose a mechanism to account for the products formed in the following reaction: The peroxide works as light, hν. So, read “NBS/hν.”Predict the products of the following reactions.(a) cyclohexylmethanol + TsCl>pyridine (b) product of (a) + LiAlH4(c) 1@methylcyclohexanol + H2SO4, heat (d) product of (c) + H2, Pt

