What is the heat of a reaction, in joules, with a total reaction mixture volume of 68.8 mL if the reaction causes a temperature change of 5.1 °C in a calorimeter? Assume that the reaction mixture has a density of 1.00 g/mL and a specific heat of 4.184 J/g-°C. The calorimeter has a heat capacity of 10.0 J/°C. Type answer:
What is the heat of a reaction, in joules, with a total reaction mixture volume of 68.8 mL if the reaction causes a temperature change of 5.1 °C in a calorimeter? Assume that the reaction mixture has a density of 1.00 g/mL and a specific heat of 4.184 J/g-°C. The calorimeter has a heat capacity of 10.0 J/°C. Type answer:
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter8: Thermochemistry
Section: Chapter Questions
Problem 11QAP: When one mol of KOH is neutralized by sulfuric acid, q=56 kJ. (This is called the heat of...
Related questions
Question
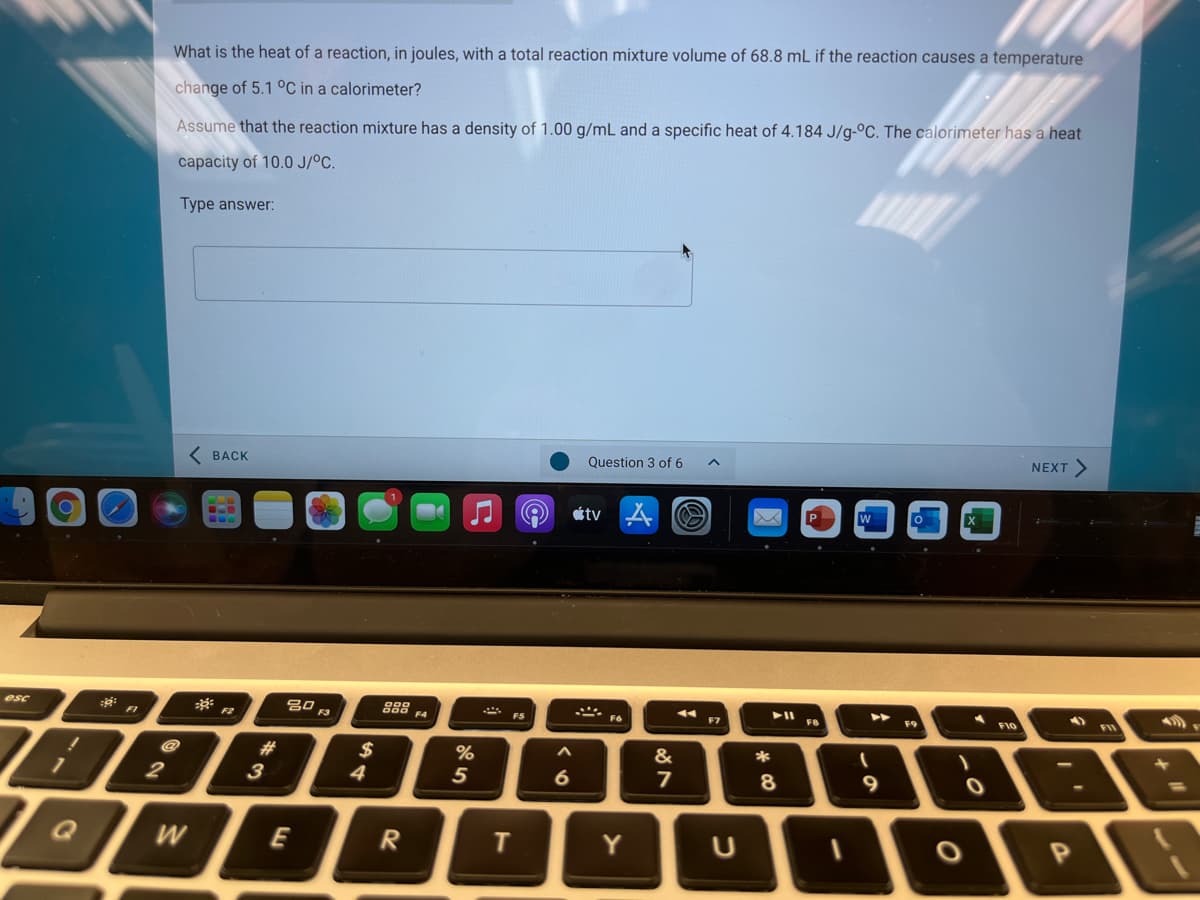
Transcribed Image Text:What is the heat of a reaction, in joules, with a total reaction mixture volume of 68.8 mL if the reaction causes a temperature
change of 5.1 °C in a calorimeter?
Assume that the reaction mixture has a density of 1.00 g/mL and a specific heat of 4.184 J/g-°C. The calorimeter has a heat
capacity of 10.0 J/°C.
Type answer:
< ВАСК
Question 3 of 6
NEXT
étv
w
g88 FA
ese
FI
F2
F6
F7
FB
F10
2$
4.
@
23
&
2
3
5
7
9
W
E
R
T
Y
P
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning