Q: Na[Ni(NH3)4(NO3)2] [Co(NH3)3(H2O)3]Cl2 H C NH₂
A: Coordination chemistry is branch of chemistry in which we deal with metal complexes. In metal…
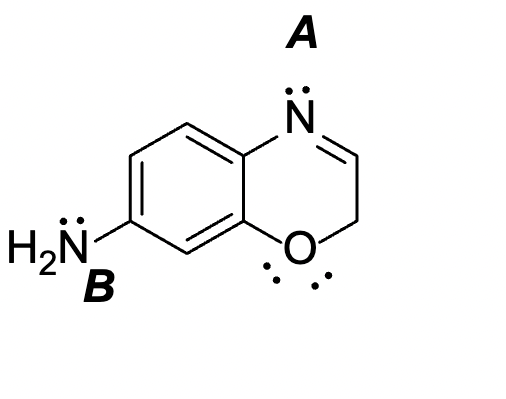
Q: 2.7 Fill in the electron lone pairs. How many are there altogether for the two structures?
A: Lewis’s structure: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a…
Q: Table 1. Listed below are the common metals with different masses. These metals also have different…
A:
Q: What is the long-hand electron configuration of tantalum, Ta?
A:
Q: Valacyclovir is an antiviral drug that slows the growth and spread of the herpes virus to help the…
A:
Q: A solution containing ethylene glycol and water has a vapor pressure of 7.96 Torr at 10 °C. Pure…
A: The vapor pressure of pure water at 10oC is = 9.21 torr The vapor pressure of the solution of…
Q: 2. Calculate the concentration of the salt stock solution you made in moles CaCl₂ per L (M). Note:…
A: 2. The concentration (molarity) of the stock solution can be calculated by dividing moles of salt by…
Q: 23. Given the following: (a) NH3 (b) NaOH (c) H₂O (d) BH3 The Bronsted Base(s) is(are) (a) NH3 (b)…
A: 3) Bronsted-Lowry acid base theory: According to this theory acid is a substance which donates H+…
Q: How many kilocalories are needed to convert 18 g of ice at 0 °C to steam at 100 °C? (Hint: The…
A:
Q: The element lithium has two stable isotopes, lithium-7 with a mass of 7.016 amu an lithium-6 with a…
A:
Q: Determine the number of hydrogen atoms connected to each carbon atom: The bond-line structure of a…
A: The labeling of carbon atoms can be done as follows:
Q: [H+] 11 [OH™] 2[CO-] + Answer Bank 2[H+] [HCO3] + 3[H+] [CO] [H,CO, ]
A: The balanced chemical equation is the equation in which the number of atoms on the both sides of…
Q: 11) Calculate the osmotic pressure associated with 90.0 g of an enzyme of molecular weight 98,000…
A:
Q: Provide proper IUPAC naming jsut part d,e, &f
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: OH لي - لي
A: For the acid -base equilibrium reaction, the equilibrium will favour to the side which contains…
Q: solution of carbonate (pKa = 6.3, 10.3) is at pH 10.1. Calculate the base:acid ratio at this pH.
A:
Q: An ion from a given element has 24 protons and 21 electrons. What is the charge on the ion? What is…
A:
Q: A reaction that has a rate only dependent on catalyst will be: A. None of the above B. Zero Order…
A: The sum of the exponent term in the rate law for a chemical reaction is known as the order of the…
Q: 26. Which of the following has a hydrophilic side chain? I.N II. L III. G I only OI and II OII and…
A: Note: According to our guidelines we are supposed to answer only one question.kindly repost other…
Q: 2 5 NON 2 NO* + NO3 → 2NO ₂ 2 3 → NO₂+O₂+NO* Find the overall rate expression for NO 2 5 ). ΝΟ 25
A: Rate is reaction is given by the slowest step. Rate of reaction should not contain the intermediate…
Q: Give the electron configuration of [Cr(NH3)sCI]Br2 using the crystal field theory of the and show…
A: Given complex is: [Cr(NH3)5Cl]Br2 NH3 is neutral ligand having charge zero. Cl and Br are anionic…
Q: Draw the constitutional isomers of the following molecular formulas.
A: Isomers are those which have same molecular formula but different arrangement of atoms.…
Q: 5. 6. CH O,N. CI СООН COOH CN
A: IUPAC nomenclature of carboxylic acid: • Identify the longest continuous carbon ring • Identify the…
Q: 100.0mL of 0.45M HBr are mixed with 50.0mL of 0.85M CaCl2 and 100.0mL of 0.80M NH3. Determine…
A: According to the question we have, The volume of HBr = 100.0 ml The molarity of the 100.0 ml HBr =…
Q: At 500 K, the rate of bimolecular reaction is 10x the rate at 400 K. Find the energy of activation…
A:
Q: 5. Give the IUPAC name for each of the following alcohols. a. Н b. С. HO ОН она НО- CH3 CH3 -OH
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: What type of hybridization does the Oxygen atom in the following molecule undergoes? CH3CH2CH₂OH…
A: Given molecule is CH3CH2CH2OH We have to find the hybridization of Oxygen in this molecule.
Q: For a second-order reaction with k= 0.0561 M-¹ s¹, what is the concentration after 400 seconds if…
A:
Q: What is the half-life of a first-order reaction if k =0.050 s¨¹? Your Answer: Answer units
A: Given that The rate constant of the first order reaction, k = 0.050 s-1 The half-life period of the…
Q: If 454 g of NH4NO3 decomposes, how much N₂O and H₂O (in grams) are formed? mass of mass of
A: Late of conservation states that sum of masses of reactant is equal to total mass of products.…
Q: WHY IS DIETHYLAMINE MORE BASIC THAN METHYLAMINE? ( PROVIDE STRUCTURE)
A: Basicity is defined as tendency of atom to donate the lone pair of electrons. More the tendency of…
Q: Which of the following represent the proper long hand form electron configuration of ruthenium, Ru,…
A: Concept based on the building up principle.
Q: If the rate constant for the decomposition reaction of cyclobutane into ethylene is 2.08 x 10-2 s1…
A:
Q: Directions: For each of the following molecules, determine the number of carbon atoms present and…
A:
Q: A student decomposes KClO3 and collects 36.5 cm3 of O2 over water at 23 oC. The laboratory barometer…
A: The relationship between the temperature, pressure, and volume of the initial and final state of an…
Q: Classify each of the following as a combination, decomposition, single replacement, double…
A: Here we are required to classify the given reaction according to its type.
Q: Assume that we used a solute with a larger molar mass than salicylic acid, would this change deltaTf…
A:
Q: How many oxygen atoms are in 0.25mol Ca(NO3)2?
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: for part b would 4-secbutyl-3,3,5-trimethylnonane be correct?
A:
Q: Use the following information to answer the next question: Mass of aluminum calorimeter: 470.0 g…
A:
Q: A solution has 5.25 mmol L-1 total phosphate (pKa = 2.1, 6.8, 12.4). If the pH of the solution is…
A: A buffer works best at a pH which is close or equal to the pKa of its acid. The pH of buffer is 7.4…
Q: Part A ▾ It takes 53.0 J to raise the temperature of an 9.80 g piece of unknown metal from 13.0°C to…
A: A.) We would use thermochemistry formula to calculate specific heat. C.) Specific heat can be…
Q: Consider a cold can of soft drink on a table in a warm room. If the cold can of soft drink is the…
A: According to the first law of thermodynamics, the energy of the universe remains constant. For a…
Q: What is the pH of these solutions? 0.100M sodium acetate CH3CH2O2Na, Kb=5.6x-10 A solution that is…
A: Given solutions are 0.100M sodium acetate CH3CH2O2Na, Kb=5.6x10-10 A solution that is both 0.050M…
Q: Solve equation
A:
Q: Which molecule below do you predict has the highest water solubility? бу б H₂N ОН ОН ОН м ОН ОН ОН Ц…
A: For the highest solubility of compounds in water. We look at the polarity of that compound. Since we…
Q: 6. Which compound is the strongest base? Justify your answer. Br HLO A. H-C-O B. CH3CH₂CH3O C.…
A: A compound which easily accepted hydrogen ion are easily donate hydroxide ion known as base .
Q: 7 Fill in the electron lone pairs. How many e there altogether for the two structures?
A: Lone pair means a pair of valence electrons that are not shared with another atom in a covalent…
Q: 33. Select the ODD one out: 000 OF OP OW OG
A: F,P,W,G are given as the codes for amino acids out of which three are similar amino acids and one is…
Q: Adipic acid is used in the production of nylon, so it is manufactured in large quantities. The most…
A:
What is the hybridization of each N atom and does their lone pairs participate in resonance?
Step by step
Solved in 2 steps with 1 images


