What is the isoelectric point of AA? [Select]
Chapter10: Reconstitution Of Powdered Drugs
Section: Chapter Questions
Problem 19SST
Related questions
Question
Answer number 2
![2. What is the isoelectric point of AA? [Select ]
[ Select ]
3. What is the pka corresponding to th
2.00
[ Select ]
7.00
3.90
2.95
4. Region/point where AA is predomin 9.70
12.00](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Ff03e3e3e-dbe3-4ed0-b094-53a4b5e345c9%2F32c14f95-ebe5-4e11-aff6-5bd7f0a80b9b%2F83a5cal_processed.jpeg&w=3840&q=75)
Transcribed Image Text:2. What is the isoelectric point of AA? [Select ]
[ Select ]
3. What is the pka corresponding to th
2.00
[ Select ]
7.00
3.90
2.95
4. Region/point where AA is predomin 9.70
12.00
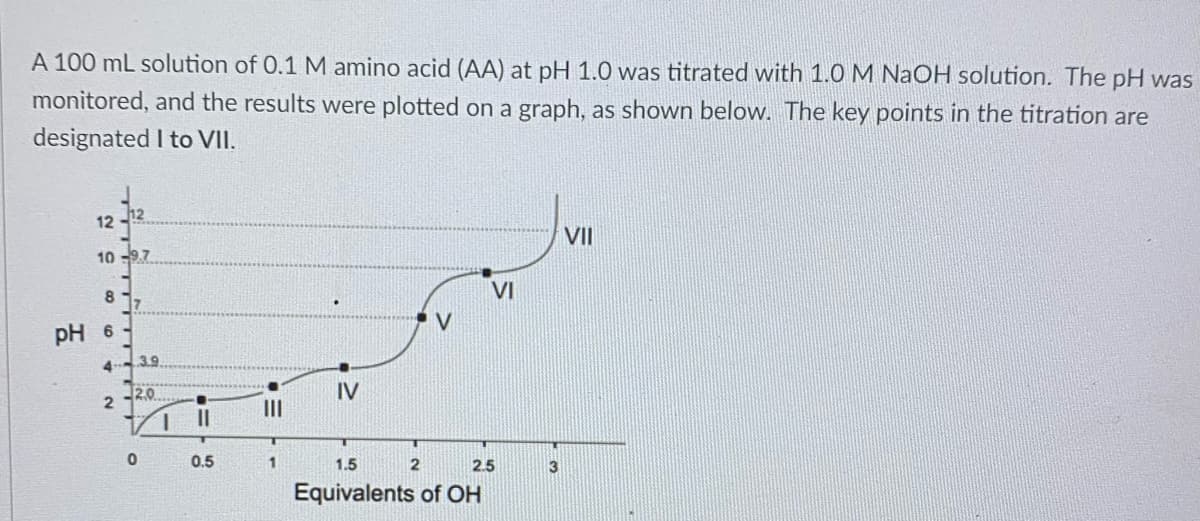
Transcribed Image Text:A 100 mL solution of 0.1 M amino acid (AA) at pH 1.0 was titrated with 1.0 M NaOH solution. The pH was
monitored, and the results were plotted on a graph, as shown below. The key points in the titration are
designated I to VII.
12
VII
10 -9.7
VI
17
V
pH 6
3.9
4
12.0
IV
II
1.
0.5
1.5
2.5
3
Equivalents of OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you



