Q: Determine the position of equilibrium for each of the following proton transfer reactions. H-CI: H…
A: Given is a proton transfer reaction.To find the position of equilibrium.Concept:Proton Transfer: It…
Q: A H₂C CH₁ 1, CH₂Li, ether 2. H₂O, H₂SO₁ CN MgBr , ether, reflux 5 h 2. CH₂OH MgBr 1. CO₂ (s), -70…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: The following table provides some information on carbon dioxide solubility in water. C P k T (mol/L)…
A:
Q: Please draw detailed mechanism of this reaction. Picture are attached:
A: Given is a reaction of aryl amine with Nitrous acid in the presence of strong acid and KI.To the…
Q: When this molecule reacts with HI, two stereoisomers are formed. Part: 0 / 3 ОН Part 1 of 3 Draw the…
A: Stereoisomers are compounds that have the same molecular formula but different spatial arrangement…
Q: 1. (Ch. 14) The hypochlorite ion (OCl-) is widely used for water disinfection. Below is the…
A: Answer:Here:
Q: Calculate K for the following reaction: given the following K values: A + 2B=C C D + B D=A+B K₁ =…
A: Characteristics of equilibrium constant are : -If the equation (having equilibrium constant K) is…
Q: Choose the best reagents and conditions to perform the following reactions (stereochemistry is not…
A: Markovnikov=> markovnikov rule explains that in addition reaction the proton(H+) added to the…
Q: do you have to subtract the amount of moles of NO2 after 2 minutes from the initial amount
A: The given reaction is a 2nd order reaction We have to tell why we have to subtract the amount of…
Q: 2NOBr(g) 2NO(g When she introduce 0.112 atm. Calculate the equilib Kp =
A:
Q: For the reaction 2 A + 4 B → 2 C + 4 D, what is the absolute magnitude of the rate of change for [B]…
A: The objective of the question is to find the rate of change of concentration of reactant B ([B])…
Q: What is the major product of the following reaction? O Br Br Br Br Br Br Br Br Br Br Br -CBr3 Br xs…
A: The objective is to determine the product of the given reaction. NBS is a controlled brominating…
Q: Predict which proton will have the most upfield signal in the ¹ H NMR spectrum. || H H Br H IV H I 3…
A: Most upfield signal is signal with lowest chemical shift.Generally sp3 hybridized carbon has lower…
Q: Explain implications of the hydrogen bonding between water molecules The images to the right depict…
A: The objective is to explain the implication of hydrogen bonding in the water molecules.
Q: 3. Determine the Miller indices for the planes shown in the following unit cell: +Z B A -IN 2/3 +y
A: After joining the crystal lattice points by straight lines, those straight lines were assumed to be…
Q: What is the maximum mass (in grams) of Magnesium Phosphate, Mg3(PO4)2, that can be obtained from…
A:
Q: Give the major products for the reaction. F3 000 F4 Cl₂ F5 (low conc.) about us | careers | privacy…
A:
Q: Draw a stable, uncharged resonance form for the structure below. Be sure to include lone pairs of…
A: In an organic reaction mechanism, the electron is transferred from a nucleophile to an electrophile…
Q: 1. Please provide the mechanism for the following transformations. If the product is not shown,…
A: As per our guidelines we are supposed to answer only the first three sub part questions hence kindly…
Q: a. Give full curved arrow pushing mecha following reaction, and draw a reaction energy diagram. b.…
A:
Q: A 50.00 ml aliquot of a solution containing Ca2+ and Mg2+ was buffered at pH 10 and titrated with…
A: The objective of the question is to calculate the molar concentration of Mg2+ in a solution. This…
Q: Highlight the most acidic of the H atoms shown in each molecule below. Note: if there are two or…
A: Acidity is defined as the ability of a molecule to release protons.If a compound easily donates…
Q: In the following multistep synthesis, fill in the missing compounds in the boxes ovided: Dilute H3O+…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Provide the correct common name for the compound shown here. O
A: To write the IUPAC name of an organic compound:we must follow the standard procedure of IUPAC…
Q: How many functional groups are in the following molecule?
A: A functional group can be defined as an atom or a group of atoms that provide some unique…
Q: Aqueous hydrochloric acid (HCI) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium…
A: From given reaction of hydrochloric acid and sodium hydroxide there is need to determine theoretical…
Q: Consider the following system at equilibrium where AH° 268 kJ, and Kc = 5.10 × 10-6, at 548 K.…
A: Equilibrium constant Kc for a reaction is defined as the product of the molar concentrations of the…
Q: Draw the skeletal structure of 1-propoxycycloheptane.
A: => For Ether , IUPAC name is written as Alkoxyalkane
Q: A test tube contains 1.0 mL of 1 mM ONP, 1.0 mL of Z buffer, and 1.0 mL of 1 M Na2CO3. The molecular…
A: To find the concentration of ONP (ortho-nitrophenol) in mg/mL, we first need to calculate the number…
Q: A solution is prepared by dissolving 371 g of sucrose (C₁2H22011) in 569 g of water. What is the…
A:
Q: Propose an efficient synthesis for the reaction below using any necessary organic and inorganic…
A: Here we are given to synthesize a cis-diol that has four carbon atoms from an alkyne that has two…
Q: Draw the major and minor product that could be formed when 1 - chloro -1,3-butadiene reacts with…
A: Given structure of (E)-1-chlorobuta-1,3-diene and acrylonitrile. Draw the major and minor…
Q: Which of the following hydrogens would you expect to be most easily abstracted from the indicated…
A: Given are organic compounds.A radical or free radical is species that has unpaired electrons.The…
Q: 3. Name the following alkynes. (a) (b) CH3CH₂CH₂CH₂CH₂CH₂C = CH CH₁ CH₁ CH₂-CH-CH-CH-CH—C—CH I CH₂…
A: The alkynes bear a triple bond between two carbon atoms. According to the IUPAC nomenclature, the…
Q: What is the most oxidized of the labeled carbon atoms in this molecule? What is the most reduced? OH…
A: Answer:Formal charge present on an atom in a specie is called the oxidation number of the atom in…
Q: Name the 2 cumulated dienės With mula 5ng If there is only one diene, leave the second space blank.…
A: The objective of the question is to name the two cumulated dienes with the formula with an…
Q: Write an acceptable IUPAC name for the compound below. Include 'cis' and trans' as part of the name…
A: The given compound is a hydrocarbon, cycloalkane. The IUPAC name of a cycloalkane can be written on…
Q: Consider the following reaction where K = 9.52x10-2 at 350 K: CH4(g) + CCl4(g)=2CH₂Cl₂(g) A reaction…
A: Note: As per the guidelines, solution of first question has been made. For the expert solution of…
Q: Calculation of Equilibrium Concentrations Using an Algebra-Simplifying Assumption What is the…
A: Given concentration of NH3 = 0.25 M K = 1.8 × 10-⁵ We have to find equilibrium concentrations
Q: How long (in seconds) does it take for the concentration of A to decrease to 0.00800 M? Enter your…
A: Answer:Here:
Q: Predict (draw) the ¹H NMR and 13C NMR spectra for each molecule. Your illustration should include…
A: 1H NMR spectroscopy is useful to determine types of protons, their signals, splitting etc and hence…
Q: Use the ΔΗ°, and ΔΗ°n information provided to calculate ΔΗ°, for IF: IF7(g) IF5(g) AH°F (kJ/mol)…
A: As we know that the enthalpy of a reaction is the difference of the standard enthalpies of formation…
Q: Consider the reaction of NO with H₂ to form N₂ and H₂O. If 3.89 g H₂ is reacted with excess NO and…
A: Mass of H2 Used = 3.89 g Mass of N2 produced (actual yield) = 20.9 g Balanced chemical equation of…
Q: What is the formal charge of "E"? A N: H 0 O-1 0-2 O +1 ;Br C :0:0 NB H
A:
Q: Consider the following system at equilibrium where AH = -87.9 kJ, and K = 83.3, at 5.00 × 10² K:…
A: Given: ∆H° = -87.9 KJKc = 83.3 T = 5.00 × 102 KLe Chateliar' principle: A reaction at equilibrium…
Q: The equilibrium constant, Kc, for the following reaction is 6.93 x 104 at 438 K. PC15 (9)PC13 (9) +…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Which of the following compounds are constitutional isomers? XX- (ii) a. (i) and (ii) b. (i) and…
A: Compounds having same chemical formula but different structures are called constitutional isomers
Q: enthalpy of for the precipitation of silver chloride is follows the reaction shown below ermine the…
A: The question is based on the concept of specific heat capacity.It is defined as the amount of heat…
Q: Show the starting diene and dienophile you could use to prepare the following molecule: Diene +…
A: The cyclic addition of diene with dienophile to form corresponding addition product is known as…
Q: If a 25.00 ml aliquot of a solution containing sulfide ions was added to 25.00 ml of 0.0139 M Cu2+…
A: The objective of the question is to find the molarity of the sulfide in the sample solution. This…
Step by step
Solved in 3 steps with 1 images

- what is the product of a reaction between C10H16(terpinene) +maleic anhydrideWhat type of reaction is involved in the following: CaC2O4 + KMnO4 (in the presence of H2SO4) _____________________________________ AsO4-3 + KI ( in the presence of CHCl3) _____________________________________ SO4-2 + Ba(C2H3O2)2 (in the presence of HCl) ______________________________________ AgSCN + FeCl3 ______________________________________Describe what each reagent does in SDS Page 1. Sodiun dodecyl sulfate 2. Polyacrylamide 3. Mercaptoethanol
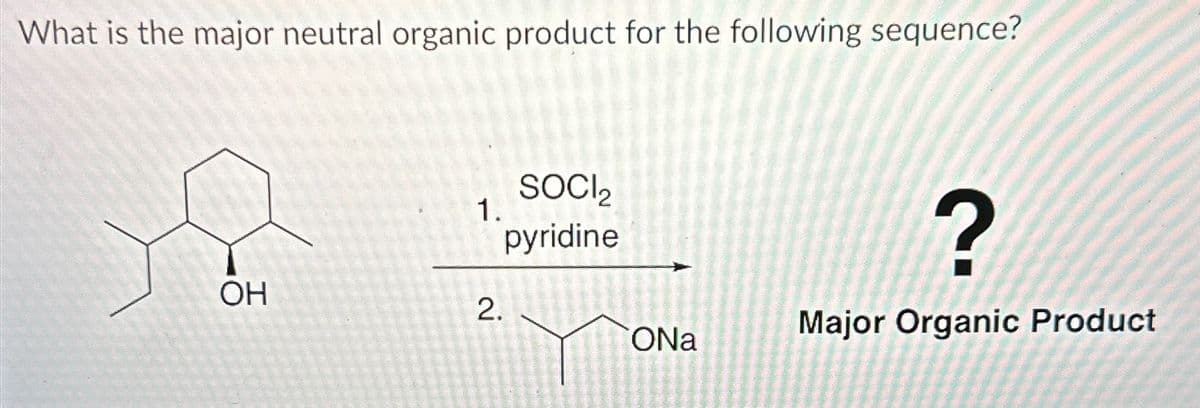
- What is the product (from the image below) of the starting material D-glyceraldehyde which will (1) produce aldaric acid upon reacting with HNO3 + H2O, NaOCH2, NH2OH, and (CH3CO)2O + NaOCOCH3 (2) produce tartaric acid upon reacting with HNO3 + H2O, NaOCH3, NH2OH, and (CH3CO)2O + NaOCOCH3What is the major neutral organic product for the following sequence?1. POCl3 Pyridine 2. Cl2H2o Major Organic Producta. For this acid-base reaction: C⊖H3 + H3O+ −−→ CH4 + H2O, the conjugate acid of carboanion (C⊖H3) is _________________ b. Complete the equation: HC≡N + NaOH −−→ ___________+__________ c. Rank the acidity of these: NH3, HF, H2O, HCl, NaH Most acidic______>_______>_______>_______>_______least acidic d. The Ka of acetic acid (found in vinega) is 1.8×10-5. Then the pKa of formic acid is
- Look at the following pairs of molecules and draw a circle around the one that has the ` hydrogen with the lowest pKa.(25) What is the major organic product for the following sequence?Methanogenic bacteria convert acetic acid (CH3COOH) to CO2(g) and CH4(g). ΔH°f, (kJ/mol) ΔG°f, (kJ/mol) CH3COOH -484.5 -389.9 CO2(g) -393.5 -394.4 CH4(g) -74.8 -50.8 Calculate ΔH°rxn. ΔH°rxn = ____ kJ Calculate ΔG°rxn. ΔG°rxn = ___kJ

