Q: Calculate the amount of activated charcoal needed to make 1 mol of Alizarin Yellow R. Assume one…
A:
Q: Under certain conditions the rate of this reaction is zero order in dinitrogen monoxide with a rate…
A:
Q: C To add an H atom in basic solution, add an H20 to the side that needs the H atom, and also add an…
A: Here we have to balance the above oxidation reaction in basic medium.
Q: alestion 7 r the following reaction at 25 C Na Hyli)+3029)2NO, 6) t2H,0) alculate AH° alculate AS°…
A:
Q: The rate of a certain reaction is given by the following rate law: =Dk[H,NH, Use this information to…
A: From the given rate equation and values we can easily find the rate constant. Just put the values in…
Q: A chemist fills a reaction vessel with 0.564 g aluminum hydroxide (Al(OH),) solid, 0.609 M aluminum…
A:
Q: A sample of oxygen gas is collected over water. If the pressure inside the collection tube is 748.2…
A:
Q: Using your knowledge about the properties of individual water molecules and your observations of the…
A: The molecular formula of a water molecule is H2O and a typical bent-shaped water molecule looks like…
Q: Rank the aniens from stromgest to weakest base B. S.
A:
Q: Consider a random mixture containing 7.00x10 particles of Na2CO3 and 5.00x10' particles of K2CO3.…
A:
Q: 3. Draw the structure of procainamide hydrochloride, mark the aromatic amino-group and suggest the…
A:
Q: Consider the galvanic cell based on the following half-reactions: Zn2+ + 2e - Zn e=-0.76 V Fe+ + 2e…
A:
Q: 2. Draw the scheme of nifedipine synthesis (name all the products).
A: The drug can be defined as a medcation. They are used to treat and cure diseases. One of the…
Q: Some measurements of the initial rate of a certain reaction are given in the table below. N2| |H2|…
A:
Q: Question 11 Which type of bond results from two atoms sharing electrons? covalent bond O ionic bond…
A: Given, Type of bond which result from two atoms sharing electrons. Covalent bond Ionic bond metallic…
Q: Enter your answer in the provided box. The combustion of how many moles of ethane (C,H) would be…
A:
Q: Match each molecular geometry with the approximate bond angle. linear [Choose ] trigonal planar […
A: Match each of the following geometry--
Q: 1. Draw the best Lewis dot structure for the anion CCly in the correct molecular geometry IInclude…
A:
Q: liter neces- 1. Calculate the minimum concentration of oxygen in milliliters sary to passivate iron…
A: Solution: In the given question we have to calculate the minimum concentration of oxygen.
Q: A sample of argon gas occupies a volume of 9.38 L at 58.0°C and 1.01 atm. If it is desired to…
A: According to an ideal gas equation PV= nRT Where P= pressure of the gas, V= volume of the gas, n= no…
Q: A solution prepared by dissolving 25.8 mg of benzene (78.11 g/mol) in hexane (86.16 g/mol) and…
A: We know that According to the Beer-Lambert law A = ε × b × c where A = Absorbance ε= molar…
Q: Q3: An equimolar liquid mixture of ethanol and water is in equilibrium with its vapor at 50° C.…
A: Given: Temperature of vapor = 50 C = (50 + 273) = 323 K For water, A = 18.30, B = 3816.44, C =…
Q: Combustion of hydrocarbons such as undecane (C,H) produces carbon dioxide, a "greenhouse gas."…
A:
Q: Question 6 For the compound. Name the rompound I dentify each chiral center Are goometric isomers…
A: Isomers are the molecules having same molecular formula but different arrangement of atoms in it.
Q: km Calculate the kinetic energy of a 3.1 x 10" kg airplane moving at a speed of 0.28 Round your…
A:
Q: Determine the value of ASgurr at 298 K, and predict whether or not the reaction is spontaneous at…
A:
Q: What would be the volume of a sample of CO2 if the sample contained 400 grams at 35°c and 785 mmHg?
A: Given :- Mass of CO2 = 400 g Temperature = 35°C Pressure = 785 mm Hg To calculate : Volume
Q: For the reaction A + B* - A* + B = -2,4 V Which of these conclusions is TRUE at standard conditions?…
A: Concept: Oxidation and reduction: Oxidation is the loss of electrons or an increase in the…
Q: -Question Completion Status QUESTION 10 Which one of the following alcohots wi yeld a yellow…
A: The iodoform test is given by an alcohol as shown in the following step.
Q: [References] Consider the galvanic cell based on the following half-reactions: Zn2+ + 2e-+ Zn e…
A:
Q: Which properties decrease as you go left to right in a period and increases as you go down a column…
A:
Q: Calculate the entropy change that occurs in the system when 56.2 g of liquid hexane (86.17 g/mol)…
A: As you asked only first question that is question number-(18) so I am going to solve this question…
Q: Zwitterions have no tendency to migrate by electrophoresis in an electric field. At which pH will…
A: We know that, zwitterions have both positive and negative equal amount of charges so that net charge…
Q: Chemistry Synthesize the following compounds from benzene:.
A: Since, you have multiple subparts questions so I will solved first three subparts questions for you…
Q: A sample of 7.0 mL of concentrated sulfuric acid, H2SO4 is used to make 250. mL of a 0.50 M sulfuric…
A: Given :- initial volume of solution = 7.0 mL Final concentration of solution = 0.50 M Final volume…
Q: Complete the following table, which lists information about the measured acid dissociation constants…
A: pKa value: pKa value negative logarithm of the acid dissociation constant (Ka). pKa = -logKa Low pKa…
Q: Based on this lipid structure, what kind of product is formed if NaOH is added to a triglyceride?…
A:
Q: Question 2 e Complex (Cu(NH,),"abserb bykt with a wanlength of 342 nm.What is the valus A Nin K3…
A:
Q: TUTOR Identify the Components of a Voltaic Cell A voltaic electrochemical cell is constructed using…
A:
Q: 2.0 mol of nitrogen gas at STP occupies what volume? O 11.2 mol O 11.2 mol/L O 44.8 mol O 11.2 L O…
A:
Q: How many moles of gas would be produced 5 grams of sodium bicarbonate reacted completely with acetic…
A:
Q: For the electrochemical cell 2 Al(s) + 3 Mn (aq) – 2 Al*(aq) + 3 Mn(s) (E° = 0.48 V, [Al**] = 1.0…
A:
Q: Draw the structure of the organic product formed when the given compounds undergo the three-step…
A:
Q: 1. Calculate the frequency and the energy of a mole of photons associated with an electromagnetic…
A:
Q: 21. At 50.0 °C, the volatile liquid hydrocarbon hexane, CH, displays a vapor pressure of 400 mm Hg.…
A: Molecular weight of icosane C20H42 = 282.55 g/mol Number of moles of non-volatile solute (icosane)=…
Q: the pH of a basic solution is 10.47. What is [H+]
A:
Q: 1- How can you prepare 5% NaOH solution ?
A: Given : Concentration of NaOH solution = 5 %
Q: The decomposition of NF3 ir
A:
Q: * 00 Al The rate of a certain reaction is given by the following rate law: =Dk[N,][HT Use this…
A:
Q: What is the mass in grams of 0.626 cmol(+) of magnesium (Mg2+), where cmol(+) is the moles of charge…
A:
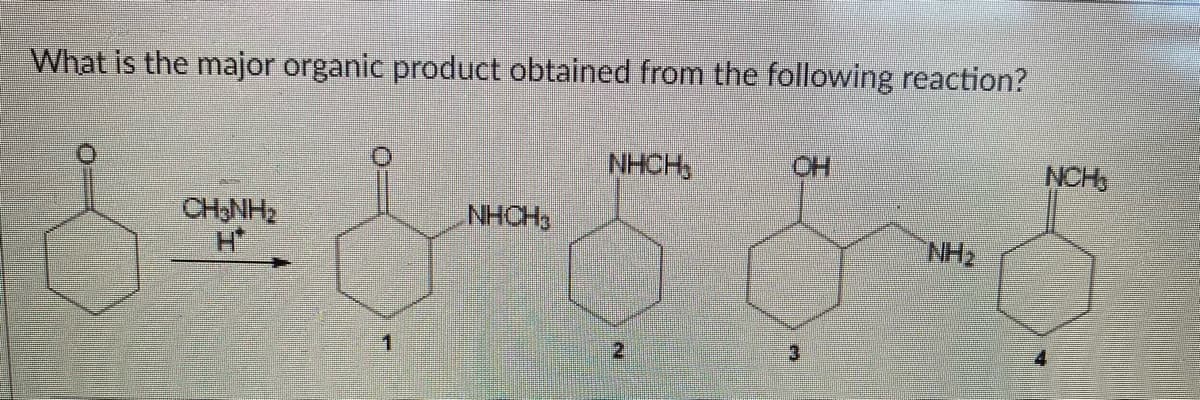
Step by step
Solved in 2 steps with 1 images

- Draw the major organic product formed in the following reaction. (The reaction stoichiometry is 1 mol reactant: 1 mol Br2.)Compounds that contain both a hydroxyl group (OH) and a carboxyl group (COOH) can undergo an intramolecular esterifi cation reaction. What product is formed when each hydroxy acid undergoes an intramolecular reaction? a. HOCH 2CH 2CH 2CH 2CO 2H b. HOCH 2CH 2CH 2CO 2HDraw the stucture of the organic reactant/s or product/s of the following reactions. ASAP!!!
- Draw the products formed when each compound is treated with CH3CH2COCl, AlCl3.Ethyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Part A Given 7.30 gg of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%% yield? Express your answer in grams to three significant figures. Part B A chemist ran the reaction and obtained 5.95 gg of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. Part C The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0%% yield. How many grams would be produced from 7.30 gg of…Ethyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.45 gg of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%% yield? Express your answer in grams to three significant figures. A chemist ran the reaction and obtained 5.50 gg of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0%% yield. How many grams would be produced from 8.45 gg of butanoic acid and excess…

