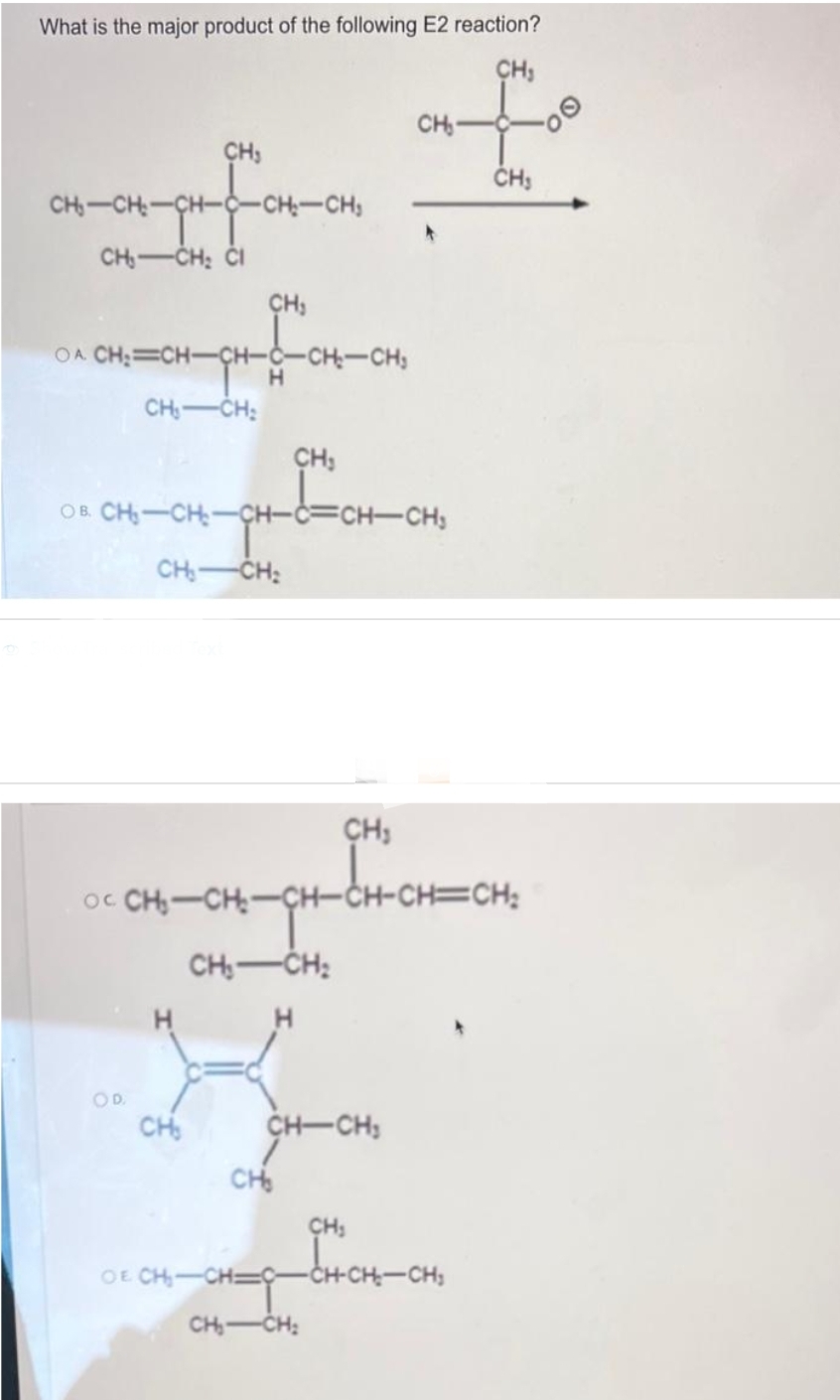
What is the major product of the following E2 reaction? CH₂ of CH₂- CH₂ CH-CH-CH CHS __ CH₁ CH₂ CI -CH₂-CH₂ CH₂ OA CHỊ=CHCH-CCHCH3 H CH₁ CH₂ CH₂ A OB. CH₂-CH₂-CH-C=CH-CH₂
Q: Macroscopic evidence that shows a chemical change did not take place. Ag (s)+HCl (aq) —-> no…
A: This question is based on reactivity of elements.
Q: 87. The ratio of the rate of effusion of Nitrogen (N₂) molecule and a carbon dioxide (CO₂) molecule…
A:
Q: How would yo perform the experiment of iron sulphide safely in a controlled manner?
A: Performing an experiment involving the formation of iron sulfide (FeS) can be done in a controlled…
Q: A 8.65 g sample of an unknown salt (MM = 116.82 g/mol) is dissolved in 150.00 g water in a coffee…
A: Moles, often represented by the symbol "mol," is the unit of measurement for the amount of a…
Q: When setting up an organic reaction for the first time, a concentration of reactants in tens to…
A: In this we have to find out the volume of the solvent required to prepare a solution that has a…
Q: Draw the major organic product of the Bronsted acid-base reaction between the compounds shown below.…
A: Bronsted bases are those species which accept proton or H+ from other compounds and form conjugate…
Q: OMe Br ON MO. Byo Me
A: This is an example of Diels-Alder reaction
Q: Heptane and water do not mix, and heptane has a lower density (0.684 g/mL) than water (1.00 g/mL). A…
A: Since heptane and water do not mix, we can assume that they form two separate layers in the…
Q: 1) HB(C5H11)2 2) H₂O₂, NaOH, H₂O 1 1) NaNH, 2) H3CH₂CBr 2
A: The first step is hydroboration reaction where we form an enol which tautomerizes to form a ketone.…
Q: A 8.65 g sample of an unknown salt (MM = 116.82 g/mol) is dissolved in 150.00 g water in a coffee…
A: When a solute dissolves in a solvent, the change in enthalpy is known as the heat of dissolution. If…
Q: 10.4B). Which structural formulas represent the same compound, and which represent constitutional…
A: Constitutional isomers are the isomers that have same molecular formula but different structural…
Q: The resistance of a 0.008 mol dm-3 solution of NaCl (aq) at 300 K was found to be 2.75 x 10³ Q. In a…
A: Molar conductivity is the conductance property of a solution containing one mole of the electrolyte…
Q: The molar mass of silver (Ag) is 107.87 g/mol. Calculate the mass in grams of a sample of Ag…
A: The mass of Ag of the sample is = 3.53 g
Q: (16) All of the following are false about reversible adiabatic process except: A) Temperature will…
A: The problem is based on Thermodynamics. Here we will discuss about the adiabatic process where no…
Q: What was/were the starting material/s? Show the formation of the product. -N. À CF3 + What was/were…
A: These compounds can be formed by Diels-Alder reaction that requires an diene and a dienophile.
Q: How many grams of BaSO4 is produced when 225g of Ba(NO3)2 is reacted with aluminum sulfate? Use…
A:
Q: Part C To normalize this wave function, you must calculate the integral ²da. What is the value of…
A:
Q: Which of the following capillaries will show a capillary depression with water? O Alumina capillary…
A: Solution - There is adhesive forces present between particles of fluid and particles of container…
Q: How is TLC useful in determining the identity of a sample?
A: TLC (Thin-layer chromatography) is a prominent technique for identifying compounds and determining…
Q: On the basis of reactivity, in how many groups metals can be classified? Write names.
A: This question is related to reactivity series. The arrangement of metals in decreasing order of…
Q: Describe the trend in acid – base behaviour of the Period 3 oxides based on the above reactions,…
A: This question is related to periodic properties. Generally, the electropositive character of the…
Q: 11. Ozone naturally decomposes to oxygen via photodecomposition. 2 03(g) →3 0₂(g) AErxn=-284.4…
A:
Q: Which of the following gives rise to a prominent peak in the mass spectrum corresponding to 18 mass…
A: we have to select the compound that shows (M+-18) signal in the mass spectrum
Q: Which of the sequences below would accomplish the following transformation? -CH₂OH- 1. PBr 2. Mg,…
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: The graph above represents the titration of a weak base with HCI. Fill in the blanks with the letter…
A: Given: Titration curve of weak base with HCl To determine: pKa of second deprotonation pH of…
Q: u H₂C eTextbook and Media Hint Part 2 CH3 Add curved arrow(s) to show a mechanism for formation of…
A: The given structure of the alkoxide ion and here we have the required solution below -->
Q: 33 $ 串 Fribourg, Department of Chemistry # s for the last electron in He are: {n, I, mi, ms} ted? 1…
A: Quantum numbers: The set of numbers used to describe the position and energy of the electron in an…
Q: 26) The major product formed in the following reaction is: Me HCI
A: Alkene gives an electrophilic addition reaction with hydrogen halide to form an alkyl halide.
Q: 15. Question: Chapter: 2 Lesson: 2 Use the Bohr model and determine the wavelength of light that…
A:
Q: Provide a name or a structure for each of the following compounds include stereochemistry as…
A:
Q: 87. The ratio of the rate of effusion of Nitrogen (N₂) molecule and a carbon dioxide (CO₂) molecule…
A:
Q: 15. What is the pH of a 1.08 10 M NaH₂PO4 solution? (H₂PO4 için Ka - 7.5 10³, K₂2-6.2 10%, K₁=1.0…
A:
Q: A doctor prescribes 500 mg of Pediazole to be taken every six hours. The pharmacy supplies a…
A:
Q: MISSED THIS? Read Section 15.6 (Pages 656 - 660); Watch KCV 15.6, IWE 15.9. Consider the following…
A: A chemical reaction can be added or subtracted like an algebraic equation. The given reaction…
Q: 2. For each of the following draw/predict what you would expect to see for the ¹H NMR and infrared…
A: Since you have asked multiple questions with multiple sub-parts, we will solve the first question…
Q: Based on the reaction a chemical change indeed took place in lead solid in aqueous hydrochloric…
A: In this experiment, lead solid is placed in aqueous hydrochloric acid. Through microscope…
Q: Consider how bond strength affects the speed of particles. Describe what happens to the kinetic…
A: Bond strength can affect the speed of particles in a couple of ways. First, stronger bonds require…
Q: Classify the following as endothermic or exothermic: An explosion A reaction in which the…
A: Given Exothermic and endothermic process
Q: D. I-tetrahedral, II-seesaw, III-trigonal planar, 12. Water is one of the most important substances…
A: We have to tell why "water is a polar" molecule. The given statements are Water is polar because it…
Q: 11. Question: Chapter: 2 Lesson: 1 Green light of wavelength 516 nm is absorbed by an atomic gas. 1.…
A: Given Wavelength= 516 nm
Q: 40 Write balanced equations for the combustion of each of the following hydrocarbons. Assume that…
A: Answer-40 Combustion reaction is an exothermic reaction and when complete combustion of the…
Q: What are the indices of the direction shown? 7 1/4 OA) 434 OB) [334] C) [434] OD) [343] OC) OE) 343
A:
Q: The molar Y concentration in a 0.600 M EDTA solution buffered to pH 10.0 (-0.35 at pll 10.0) A) 0,14…
A: Given that concentration of EDTA = 0.600 M at pH = 10, the fraction of EDTA as Y–4 is αY-4=0.35
Q: 29) The major product formed in the following reaction is: Ph——Ph (a) HO₂S Ph H₂O, H₂SO4 HgSO4 Ph
A: alkyne is converted to carbonyl
Q: Provide the missing curved arrow(s) to show the last step in the mechanism.
A:
Q: 49 Show how to convert 1-butene to these compounds. (a) Butane (c) 2-Bromobutane (b) 2-Butanol (d)…
A:
Q: Draw a condensation reaction where a molecule of cysteine combines with a molecule of lysine,…
A: Condensation Reaction: Reaction in which two molecules combine to form a single molecule by…
Q: 116. In a conductivity cell of a cell constant 0.003 cm¹ concentration of 0.01 M solution of acetic…
A: Given: Cell constant = 0.003 cm-1 Resistance, R = 20 ohm
Q: If seawater has a density of 1.03 g/cm³, what is the volume of 5800 g of seawater? cm3
A: Given -> Density = 1.03 g/cm3 Weight= 5800 g
Ee.27.
Step by step
Solved in 2 steps with 2 images

- When (s)-3-bromopent-1-ene is heated in water, which of the following compounds is not produced? A )(S)-pent-1en-3-ol B) ( R)-pent-1-en-3-ol C )pent-4-en-1-ol D)(E)-pent-2-en-1-ol E)(Z)-pent-2-en-1-olBicyclo[2.2.1]heptan-7-one + PCC (in CH₂Cl₂) => A.) Bicyclo[2.2.1]heptan-7-ol B.) Bicyclo[2.2.1]heptanoic acid C.) Cyclohexanecarbaldehyde D.) All the given choices are possible products E.) No reactionThese reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O, HgSO4 choices:A,DB,CA,B,CA,B,C,D
- Are the following the same? a) trans-2-bromocyclopentanol and 1s, 2s - 2- bromocyclopentan-1-ol b) 3,3-Dichlorocyclobutanecarbaldehyde and 3,3-Dichlorocyclobutane-1-carbaldehydeThe stereochemistry of the products of reduction depends on the reagent used, with this in mind, how would you convert 3,3-dimethylbutan-2-one [CH3COC(CH3)3] to: (a) racemic 3,3-dimethylbutan-2-ol [CH3CH(OH)C(CH3)3]; (b) only (R)-3,3- dimethylbutan-2-ol; (c) only (S)-3,3-dimethylbutan-2-ol?What is the major E2 elimination product formed from each alkyl halide?
- 1a) What’s the product in the following reaction? a. 4 b. 1 c. 2 d. 5 e. 3 1b) What reagents would you use to prepare product A with good yield? a. CH3CH2CH2CH2I; 2) Li, NH3 (liq), -33 °C b. KNH2; 2) CH3I; 3) H2, Pd/C c. HBr; 2) (CH3CH2CH2CH2)2CuLi d. NaNH2; 2) CH3CH2CH2CH2Br; 3) H2, Lindlar catalyst e. (CH3CH2CH2CH2)2CuLi; 2) H2, Lindlar catalystWhen 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observedWhen 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor products.(b) When one pure stereoisomer of 2-bromo-3-phenylbutane reacts, one pure stereoisomer of the major product results.For example, when (2R,3R)-2-bromo-3-phenylbutane reacts, the product is the stereoisomer with the methyl groups cis.Use your models to draw a Newman projection of the transition state to show why this stereospecificity is observed.(c) Use a Newman projection of the transition state to predict the major product of elimination of (2S,3R)-2-bromo-3-phenylbutane
- What is the most probable elimination product when 4-chlorocyclohexene is reacted with alcoholic KOH? 1,3-cyclohexadiene 1,4-cyclohexadiene Cyclohexane CyclohexeneWhich alkyl halide will react the fastest in an SN2 reaction (CH3)3CI CH3CH2Br CH3CH2I CH3I CH3BrWhich set of reagents are most likely to affect the E2 elimination? a. CH3CH2O-, CH3CH2OH b. (CH3)3CO-, (CH3)3COH

