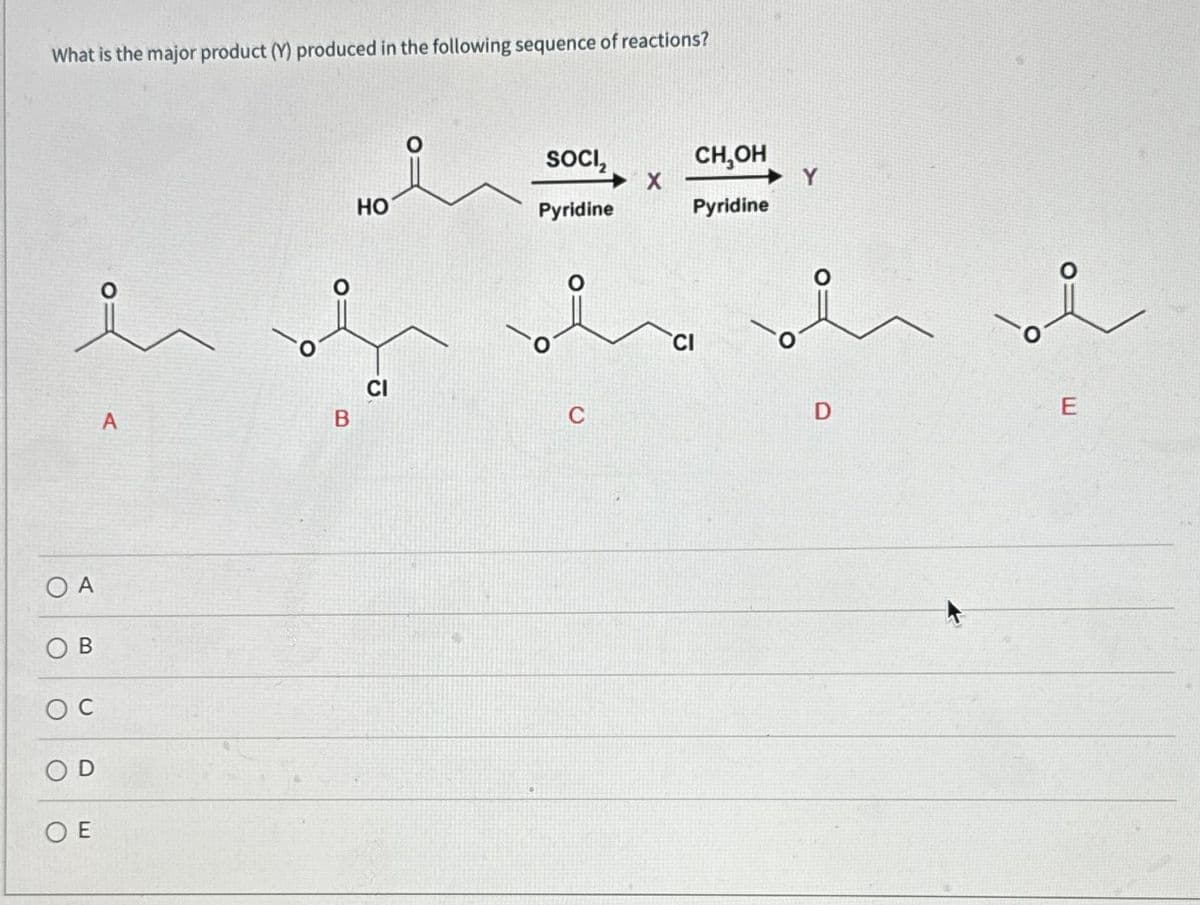
What is the major product (Y) produced in the following sequence of reactions? ů COA B OC E A HO of CI B SOCI₂ Pyridine X CH,OH Pyridine Y D E
Q: Which of the following has a zero dipole moment? A) CICN B) SO₂ C) NCI, Pls E) 0,
A: To determine the molecule with a zero dipole moment, we need to consider the individual bond…
Q: QUESTION 4 Which of the following will be the major product after a prolonged incubation of a…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Calculate energy content in kcal for food containing fat 16g carbohydrates 27g Protein 8g in kcal
A: The question is based on the concept of biomolecules.We need to calculate total energy content of…
Q: Draw the product of this reaction. Ignore inorganic byproducts. H HO H HO HO H OH H H CH₂OH Cu²+…
A: Given that, the reaction is:
Q: The Tb = 1090.00°C Tm = 649.00°C Specific heat solid = 1.017 following information is given for…
A: Given data:Calculate the amount of the heat required to raise the temperature.
Q: The solubility of Mg(OH)₂ is measured and found to be 8.85×10-³ g/L. Use this information to…
A: The solubility of magnesium hydroxide = The solubility product of magnesium =?
Q: Decide whether each chemical reaction in the table below is an oxidation-reduction ("redox")…
A: We are given 3 reactions 2Ca(s) +02(g)- 2Ca0(s)C(s)+O2(g)→CO2(g)CO2(g)+2H2O(l) =HCO3−(aq)+H3O+(aq)We…
Q: 5H. Based on the following practice problem, if either of the reactions is nonspontaneous but can…
A: For spontaneous reactions Gibbs free energy change is negative. ΔG = ΔH - T ΔS
Q: What is the abbreviated name of the compound shown below. NH₂ O AMP O ADP O ATP N N py OHOH None are…
A:
Q: Given an aqueous solution of 0.80 M propanoic acid (CH3CH2COOH, Ka = 1.33 x 10-5), calculate the…
A: The question is based on the concept of the pH of the solution.It is defined as a negative logarithm…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. Na2Cr2O7 33 Acetone,…
A: It is a oxidation reaction.Na2Cr2O7 is a strong oxidising agents it oxidises a primary alcohol to…
Q: dicate which acid derivative will have the highest and lowest te of hydrolysis. Choose the correct…
A: The question is based on concept of nucleophilic acyl substitution reaction.we need to identify the…
Q: Explosions cause damage through the by-products of exothermic chemical reactions. Which is not one…
A: A reaction in which the energy is released into the surroundings is called an exothermic reaction.…
Q: 1. H3O+, heat H 2. Neutralizing work- up o
A: Aldol condensation is a type of organic reaction in which an enol or an enolate ion reacts with a…
Q: By titration, it is found that 58.5 mL of 0.177 M NaOH(aq) is needed to neutralize 25.0 mL of…
A:
Q: Balance the following redox reaction in acidic solution. Br (aq) + HNO₂(aq) Br₂(!) + NO(g) Br (aq) +…
A: A reaction involving both oxidation and reduction is called redox reaction. In redox reaction, one…
Q: Use the References to access important values if needed for this question. When the following…
A:
Q: Consider the following particulate-level representation. The larger spheres represent N, and the…
A: Answer:In the chemical equation sign of arrow represents the sign of equal and on the left hand side…
Q: Considering stereochemistry, draw the resulting E2 elimination product when H. is removed. H H I…
A: In E2 elimination there is simultaneous removal of the leaving group and hydrogen atom in presence…
Q: A mixture of krypton and helium gases, in a 6.73 L flask at 88 °C, contains 9.60 grams of krypton…
A:
Q: Consider the following 1H NMR Spectrum for the following Compound. Which protons) are represented by…
A: Each unique proton in a compound produces a signal in hnmr spectra.The value of the signal depends…
Q: The enthalpy of solution (AH) of NaNO3 is 20.4 kJ/mol. If 9.15 g NaNO3 is dissolved in enough water…
A:
Q: A 29.4 mL sample of 0.272 M trimethylamine, (CH3)3N, is titrated with 0.385 M hydroiodic acid. After…
A:
Q: Rank alpha particles, beta particles, positrons, and gamma rays in terms of increasing penetrating…
A: Penetrating power refers to a particle's ability to pass through matter or barriers. It is a measure…
Q: Possible cation/s present (can be one or more): Cu2+, Ca2+, Fe3+, NH4+, Zn2+ Provide a flowchart…
A: (a) Cu2++ NaOH --> Cu(OH)2 Blue precipitate Cu2+ + 4NH3 --> [Cu(NH3)4 ]+2 Blue (b) Ca+2 +…
Q: H₂C Consider the two resonance forms of acetate ion shown and the curved arrow notation showing…
A: Resonanace form of acetate ion is given.
Q: b. лод HO
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: In the acid catalyzed esterification of compound 1, several possible intermediates are shown.…
A:
Q: A special gas mixture used in bacterial growth chambers contains 1.00 % by weight CO₂ and 99.0 % 0₂1…
A:
Q: Given the molecule below, the bond angles marked x, y, and z are __ (x), _____(y), and respectively.…
A:
Q: Problem 26 of 34 Submit Curved arrows are used to illustrate the flow of electrons. Follow the…
A:
Q: A student ran the following reaction in the laboratory at 671 K: H₂(g) + 12 (9) 2HI(g) When she…
A: Given here an equilibrium chemical reaction and the initial amount of reactant in terms of their…
Q: While water exhibits capillary attraction with capillary tubes, mercury exhibits capill repulsion…
A: Capillary attraction or repulsion in capillary tubes is primarily determined by the balance between…
Q: The following skeletal oxidation-reduction reaction occurs under basic conditions. Write the…
A: Answer:
Q: Polymers may be composed of thousands of monomers. Draw three repeat units (trimer) of the polymer…
A: Polymerization is a process to create polymers by adding monomer units. In the given question it has…
Q: A student wishes to determine the chloride ion concentration in a water sample at 25 °C using a…
A: A redox reaction is a reaction where an oxidation and reduction reaction occurs at two different…
Q: The solubility of Al(OH)3 is measured and found to be 2.22×10-7 g/L. Use this information to…
A:
Q: the the strongest force between the charges, select "2" beside the sketch with the next strongest…
A: According to Coulomb's law, the force of attraction or repulsion between two charged bodies is…
Q: Write a second resonance structure for the following carbocation. +
A: The given compound is shown below.We have to draw the resonance structure.
Q: What is the organic by-product in a haloform reaction? A) an ether B) a haloform C) an alcohol D) an…
A:
Q: the standard reaction free energy of the following chemical reaction: P4 (s) +20HF (g) → 4PF5 (g) +…
A:
Q: A certain hydrocarbon had the molecular formula C₁8H30 and contained two triple bonds. Ozonolysis…
A:
Q: When the following skeletal equation is balanced under basic conditions, what are the coefficients…
A: Answer:- This question is answered by using the simple concept of balancing the redox reaction.
Q: tementary reaction 2H₂O(g) + 2H₂(g) + O₂(g) proceeds at a certain temperature until the partial…
A: Given: 2H2O(g) ⇔ 2H2(g) + O2(g)Partial pressure of H2O (pH2O) = 0.013 barPartial…
Q: Using the ionization constants (K, and K) tables in your online textbook's Appendix, determine which…
A: Whether equilibrium lies on the right side or left side depends upon the acidity of the reactant and…
Q: Consider the reaction and information below: Reaction A+4+4 e- A (s) B+2+2 e-B (s) A+B (s)= A (s) +…
A:
Q: The chemical environment of carbons can be deduced form their chemical shifts. Deduce the relative…
A: Each unique carbon in a compound produces a signal in CNMR spectra.The value of the signal depends…
Q: 10. CHALLENGE. Avogadro's number has been determined by about 20 different methods. In one approach,…
A:
Q: OC is the most likely product of the following E2 reaction? CH₁ Br Br NaOCH₂CH₂ CH,CH₂OH CH₂ CH₂
A: In E2 mechanism, the E2 stands for bimolecular elimination, which involves a one-step mechanism in…
Q: MeO- OH OMe OH прав OMe OMe MeO
A: Reagents are chemical compounds of organic or inorganic nature added to a reaction to bring out a…
Please don't provide handwriting solution
Step by step
Solved in 3 steps with 3 images

- Draw the organic product(s) of the reaction of phenylacetaldehyde with KMnO4. H3O+Which of the following does not support a nucleophilic attack of a covalent catalysis? a. Hydroxyl b. Sulfhydryl c. Imidazole d. Amino e. MethylWhat products are formed when the following molecule reacts with catalytic H2SO4, H2O, and heat? Please show the mechanism for the reaction.
- When the rate constants for the hydrolysis of several morpholine enamines of para-substituted propiophenones are determined at pH 4.7, the r value is positive; however, when the rates of hydrolysis are determined at pH 10.4, the r value is negative. a. What is the rate-determining step of the hydrolysis reaction when it is carried out in a basic solution? b. What is the rate-determining step of the hydrolysis reaction when it is carried out in an acidic solution?Provide the primary product and the mechanisms of the reaction shown below.Draw the structure of unsaturated carbonyl compound(s) formed when 3-Hydroxy-2,4-diphenyl-butyraldehyde is heatedin i) a basic ii) an acidic solution. iii) Write the complete mechanism for the dehydration reaction that takes place in the basic solution.
- Reduction of an alkyl azide results in the formation of —-. A. an imine B. an oxime C. a tertiary amine D. a secondary amine E. a primary amineSelect the appropriate reagent and TWO possible substrates for the following reductive amination.The first step in the mechanism of reduction of an aldehyde or ketone with either LiAlH4 or NaBH4 is nucleophilic attack. Which of the following is the electrophilic site in this first step?
- What starting materials are needed to synthesize the following azo compound?Draw what compounds would result from complete hydrolysis of the following heterocyclic compound.What is the mechanism of this transformation? Super confused on this mechanism any help is much appreciated Apparently the first step is formation of an iminium cation through the condensation of a primary amine with dihydroxyacetone phosphate.

