Q: Write a balanced equation for the combustion of liquid propanol (C3H7OH).
A: Given chemical compound is: propanol C3H7OH. Write a balanced equation for the combustion of liquid…
Q: What is the oxidation number of C in CCl4?
A:
Q: To what category of hydrocarbon does C8 H14 belong?
A: Given that, Hydrocarbon= C8H14
Q: How can wood be so strong if it consists of cellulose chain sheld together by relatively weak H…
A: INTRODUCTION: Polysaccharides is defined as the large molecule that can be breakdown into smaller…
Q: What is the balanced equation for sugar,C12H22O11, undergoes combustion?
A: The balanced equation for sugar, C12H22O11, undergoes combustion is:-
Q: What is the oxidation number of sulfur in the tetrathionate ion, S4062?
A: Oxidation number assigned to an element in chemical combination which represents the number of…
Q: Write the steps for formation of tetrachloromethane (CCl4) from the reaction of methane with Cl2 +…
A: This is the Halogenation reaction of alkane in presence of ultraviolet light. R-H (excess) + X2/…
Q: (a) When a compound containing C, H, and O is completelycombusted in air, what reactant besides the…
A: The compounds that have C and H are known as hydrocarbons. Hydrocarbons can sometimes also have…
Q: How many kilograms of CO2 are produced by the complete combustion of 3.8 kg of n-octane?
A: The balanced chemical reaction for combustion of n-octane is shown as follows:
Q: What does it mean if burning of propane gas (C3H8) in the Bunsen burner produces blue flame? Write a…
A: If propane gas burn in the presence of excess oxygen gas then it will produces blue flame it's means…
Q: How many milligrams(mg) of oxygen would it take to oxidize 100 mg of Glucose(C6H12O6)
A:
Q: Use oxidation numbers to show that the fermentation of glucose, C6H12O6 to carbon dioxide and…
A: Redox reaction is reduction-oxidation reaction where reduction and oxidation takes place.
Q: Write a balanced equation for combustion of benzoic acid, C6H5CO2H, to give CO2 and H2O. How many…
A: The combustion reaction of benzoic acid can be written as C6H5CO2H + O2 --------> CO2 + H2O
Q: Write and balance the equation for the combustion reaction of cyclobutane, C4H8. What is the…
A: The degree of unsaturation(DoU) in the compound with molecular formula, C4H8 is given by DoU=…
Q: Upon ingestion, grain alcohol,C2H6O, is metabolized into acetaldehyde, C2H4O, which is a toxic…
A: Redox reactions has two simultaneous reactions i.e. oxidation and reduction.Oxidation is generally…
Q: (a) Write separate balanced equations for the complete combustion of ethanol (C2H5OH) and of…
A:
Q: What is the balanced molecular equation for the complete combustion (in the presence of excess…
A: Combustion is a reaction in which the compound reacts with the oxygen gas to produce carbon dioxide…
Q: Write the balanced equation for the combustion of ethane, C2H6
A:
Q: (a) When a compound containing C, H, and O is completelycombusted in air, what reactant besides the…
A: (a) Combustion of a hydrocarbon occurs when a hydrocarbon reacts with oxygen gas present in air to…
Q: At the end of 2009, global population was about 6.8 billion people. What mass of glucose (C6H12O6)…
A: At the end of 2009, global population was about 6.8 billion people. What mass of glucose in kg…
Q: What mass of glucose can be produced from a photosynthesis reaction 6 CO2(g) + 6 H2O(ℓ) →…
A: The reaction described here is the photosynthesis reaction. Here carbon dioxide and water combines…
Q: Which of the following is not indicated by this reaction? KI, Н:РО ether, 25° C It occurs at room…
A: From a reaction given, we can say the reaction temperature, reactants, products, solvent etc.
Q: What is the oxidation number of the carbon in a pentanoate ion, C5H902 ? +1.0 -2.0 O -1.0 O -1.2 O…
A: In this question we have to tell the oxidation number of carbon in pentanoate.
Q: Which is the oxidizing agent in the combustion of methanol (CH3OH)? O CH3OH O CO2 O 02
A: We have given that Which is the oxidizing agent in the combustion of methanol CH3OH ?
Q: What is the oxidation state of the C atoms in C6H12O3?
A: Oxidation state or oxidation number refers to the number assigned to the atom of a chemical compound…
Q: What is the oxidation number of C in CCl4?
A: We have to find the oxidation number of C in CCl4.
Q: Given the following information about the combustion of stearic acid, balance the following…
A: The given unbalanced equation is: C18H36O2 + O2 -----> CO2 + H2O Energy released per mol of…
Q: Q4- Why, when we add Ferrocene to our fuel, it becomes eco-friendly fuel?
A: As you have asked two questions in a single attempt, I will give you the answer for the first one.
Q: What mass of glucose can be produced from a photosynthesis reaction 6 CO2(g) + 6 H2O(ℓ) →…
A: Given reaction and information, 6 CO2(g) + 6 H2O(l) → C6H12O6(aq) + 6O2(g) Moles of CO2 is 19.4
Q: The oxidation number of C in C₂O₄²⁻ is...?
A: Oxidation number :- Oxidation number, also called oxidation state. It is the total number of…
Q: The combustion of methane is represented by the equation: CH4 + 2O2 → CO2 + 2H2O a) In the above…
A: Given :- CH4 + 2O2 ---> CO2 + 2H2O To determine :- Oxidized compound in above equation and…
Q: How much O2 is required for the complete reaction of 45 g of C2H4 to form CO2 and H2O?
A: This is a combustion reaction of Ethene C2H4 as shown below
Q: Write the combustion equations for: (a) Complete combustion in theoretical air of 1 mol of…
A: The burning of hydrocarbons in the presence of oxygen is known as combustion reaction.
Q: What is the product? OH 1. LIAIHA, Et₂O 2. H3O+
A:
Q: Write the balanced chemical equation for the fermentation of sucrose (C12H22O11) by yeasts in which…
A: Balanced chemical equation of a reaction is written according to law of conservation of mass.…
Q: At the end of 2009, global population was about 6.8 billion people. What mass of glucose (C6H12O6)…
A: Global population at the end of 2009, for whom mass of glucose needed is to be calculated in one…
Q: The hydrogenation reaction of the following hydrocarbon in the presence of platinum produces
A:
Q: . Explain in your own words about the properties of the ideal antioxidant.
A: Given : The properties of the ideal antioxidant
Q: In oxidation - reduction reaction H2SO4 was used, why
A: In oxidation - reduction reaction H2SO4 was used as a
Q: How many g of CO2 will be produced by the combustion of 4 mol of ethanol?
A: The Molecular reaction equation for combustion of ethanol C2H5OH + 3 O2 → 2 CO2 + 3 H2O + ∆ From…
Q: Write the steps for formation of tetrachloromethane (CCl4) from the reaction of methane with Cl2 +…
A: To find: The steps for the formation of tetrachloromethane (CCl4) from the reaction of methane with…
Q: when solid glucose (C6H12O6) is placed in water the solid seprates into individual atoms the solid…
A: when solid glucose (C6H12O6) is placed in water the solid seprates into individual atoms the solid…
Q: 3.) At the end of 2009, global population was about 6.8 billion people. What mass of glucose…
A: Answer: Energy intake of one person per day is E1=1500 cal/person-day Since, there are 365 days in…
Q: What is the reason behind carotenoids, why do they degrade in acidic and alkaline condition?
A: Carotenoids or sometimes called as tetraterpenoids are yellow, orange, and red organic pigments that…
Q: How many water molecules are produced when C15:2-cisA5,9 is completely oxidized to CO2 and H20?
A: As per the guidelines, I will be providing solution for the first question only. Please post other…
Q: What is the reaction that represents the combustion of C6H12O2
A:
Q: The oxidation number of the carbon atom in H2PO4- is:
A: The oxidation number is used to represent the number of electrons gained or lose by an atom in a…
What is the oxidation number of oxygen, O2, in the combustion of octane, C8H18?
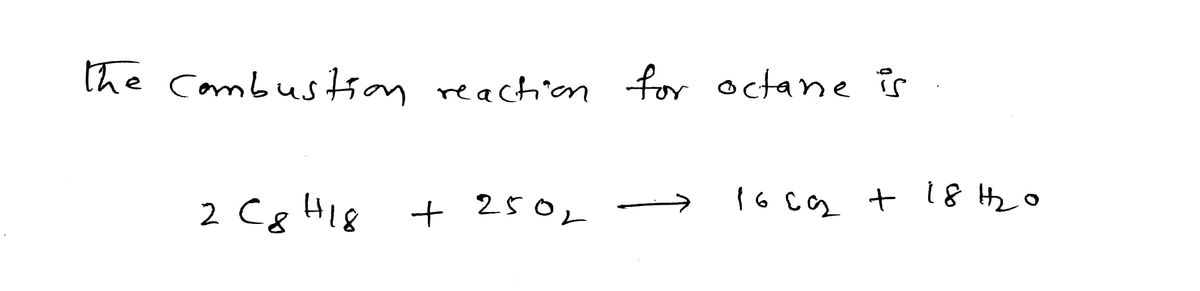
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- What is the balanced molecular equation for the complete combustion (in the presence of excess oxygen) of: C6H6 (benzene, an important industrial chemical and solvent)?What is the balance to the equation for the combustion of ascorbic acid (C6H8O6, vitamin C) in oxygen to CO2 and H2OWhat is the reaction that represents the combustion of C6H12O2
- What is the balanced molecular equation for the complete combustion (in the presence of excess oxygen) of: C21H44 (a component of paraffin wax used in candles)?Consider the equation for the combustion of acetone(C3H6O), the main ingredient in nail polish remover:Ozone is formed in the atmosphere by thecombination of oxygen molecules withA. chlorofluorocarbons.B. hydrocarbons.C. oxygen atoms.D. CO2.E. ClO.
- Suggest a net reaction for the production of nitroglycerin, C3H5(NO3)3, from glycerin, C3H5(OH)3.Octane 1C8H182 is a liquid hydrocarbon at room temperature that is a constituent of gasoline. (a) Write a balanced equation for the combustion of C8H181l2 to form CO21g2 and H2O1l2.Write a balanced chemical equation, including states of matter, for the combustion of gaseous benzene, C6H6.
- What is the oxidation number of C in CCl4?Write a balanced equation for the combustion of isooctane, C8H18, a component of gasoline.(a) When a compound containing C, H, and O is completelycombusted in air, what reactant besides the hydrocarbonis involved in the reaction? (b) What products form in thisreaction? (c) What is the sum of the coefficients in the balancedchemical equation for the combustion of one mole ofacetone, C3H6O(l), in air?

