Chapter20: Applications Of Oxidation/reduction Titrations
Section: Chapter Questions
Problem 20.28QAP
Related questions
Question
Transcribed Image Text:dation state of -1
Constants Periodic Table
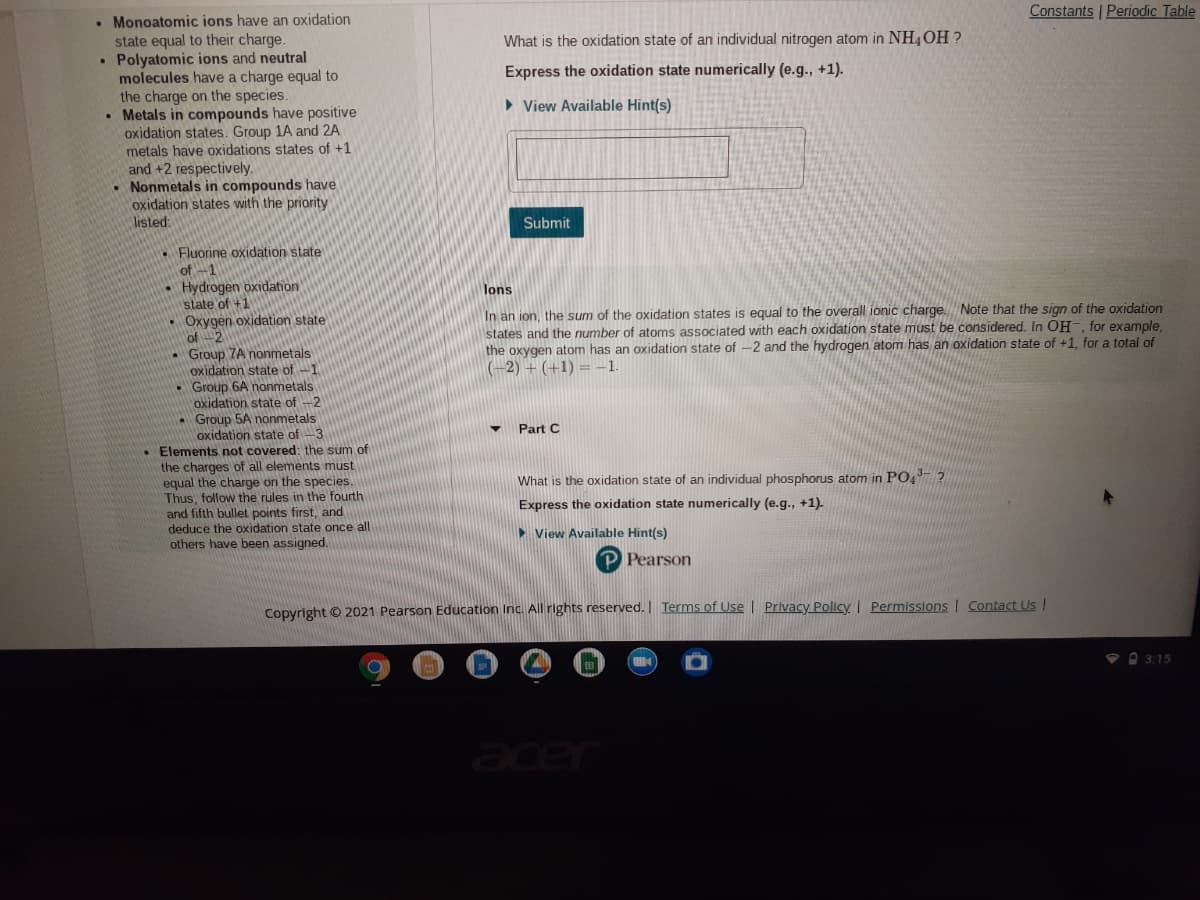
• Monoatomic ions have an oxidation
state equal to their charge.
• Polyatomic ions and neutral
molecules have a charge equal to
the charge on the species.
• Metals in compounds have positive
oxidation states. Group 1A and 2A
metals have oxidations states of +1
and +2 respectively.
• Nonmetals in compounds have
oxidation states with the priority
listed:
What is the oxidation state of an individual nitrogen atom in NH4OH ?
Express the oxidation state numerically (e.g., +1).
> View Available Hint(s)
Submit
. Fluorine oxidation state
of-1
Hydrogen oxidation
state of +1
Oxygen oxidation state
of 2
7A nonmetals
lons
In an ion, the sum of the oxidation states is equal to the overall ionic charge. Note that the sign of the oxidation
states and the number of atoms associated with each oxidation state must be considered. In OH, for example,
the oxygen atom has an oxidation state of -2 and the hydrogen atom has an oxidation state of +1, for a total of
(-2) + (+1) = –1.
• Group 6A nonmetals
oxidation state of -2
- Group 5A nonmetals
oxidation state of -3
• Elements not covered: the sum of
the charges of all elements must
equal the charge on the species.
Thus, follow the rules in the fourth
and fifth bullet points first, and
deduce the oxidation state once all
others have been assigned.
Part C
What is the oxidation state of an individual phosphorus atom in PO,- ?
Express the oxidation state numerically (e.g., +1).
> View Available Hint(s)
P Pearson
Copyright © 2021 Pearson Education Inc. All rights reserved. Terms of Use | Privacy Policy | Permissions | Contact Us |
V O 3.15
acer
Transcribed Image Text:E O Home - AP Student..
6 Elk Grové Unifled
Constants | Peniodic Table
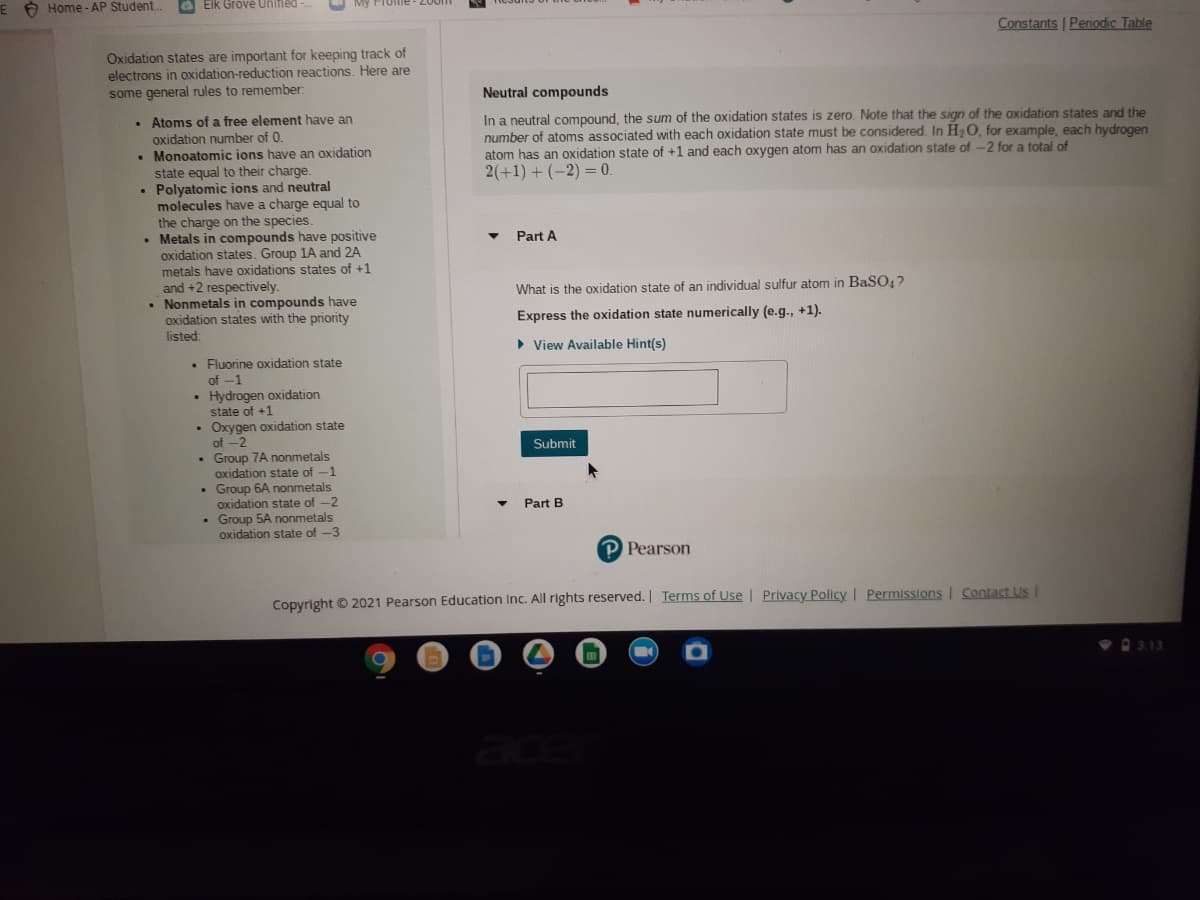
Oxidation states are important for keeping track of
electrons in oxidation-reduction reactions. Here are
some general rules to remember:
Neutral compounds
• Atoms of a free element have an
oxidation number of 0.
• Monoatomic ions have an oxidation
state equal to their charge.
Polyatomic ions and neutral
molecules have a charge equal to
the
In a neutral compound, the sum of the oxidation states is zero. Note that the sign of the oxidation states and the
number of atoms associated with each oxidation state must be considered. In H2O, for example, each hydrogen
atom has an oxidation state of +1 and each oxygen atom has an oxidation state of -2 for a total of
2(+1) + (-2) = 0.
%3D
charge on the species.
• Metals in compounds have positive
oxidation states. Group 1A and 2A
metals have oxidations states of +1
and +2 respectively.
• Nonmetals in compounds have
oxidation states with the priority
listed
Part A
What is the oxidation state of an individual sulfur atom in BaSO,?
Express the oxidation state numerically (e.g., +1).
• View Available Hint(s)
• Fluorine oxidation state
of -1
Hydrogen oxidation
state of +1
Oxygen oxidation state
of -2
Submit
Group 7A nonmetals
oxidation state of -1
Group 6A nonmetals
oxidation state of -2
Group 5A nonmetals
oxidation state of -3
Part B
P Pearson
Copyright © 2021 Pearson Education Inc. All rights reserved. Terms of Use | Privacy Policy I Permissions | Contact Us I
O 3.13
ace
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning