What is the percent composition (% w/v) of the solution created by dissolving 2 packets of sugar (5.0 grams each) into a 24 ounce glass of ice tea (density = 1.104 g/ml)? %3D XIDO 1.33% %3D 1-104 x680.40O Calculate the molality (m) of the sweet tea solution in the question above given that table sugar is sucrose, C12H22011.
What is the percent composition (% w/v) of the solution created by dissolving 2 packets of sugar (5.0 grams each) into a 24 ounce glass of ice tea (density = 1.104 g/ml)? %3D XIDO 1.33% %3D 1-104 x680.40O Calculate the molality (m) of the sweet tea solution in the question above given that table sugar is sucrose, C12H22011.
Chapter7: Solutions And Colloids
Section: Chapter Questions
Problem 7.42E
Related questions
Question
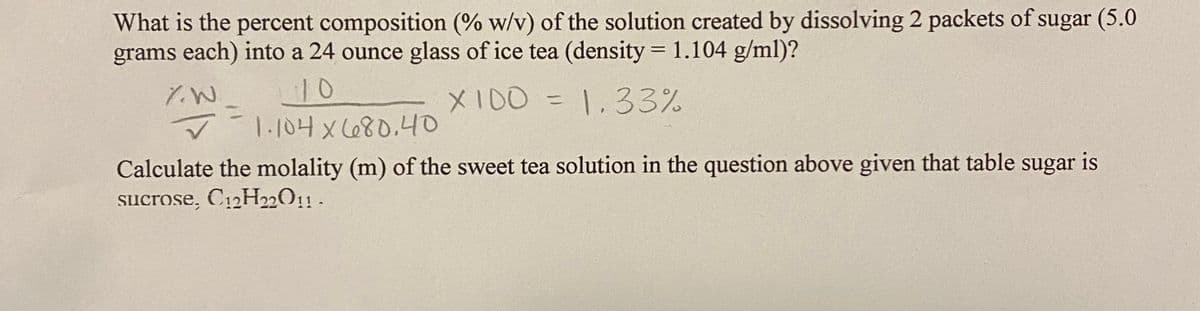
Transcribed Image Text:What is the percent composition (% w/v) of the solution created by dissolving 2 packets of sugar (5.0
grams each) into a 24 ounce glass of ice tea (density = 1.104 g/ml)?
%3D
10
1.104 x680.4O
XI00 = 1.33%
Calculate the molality (m) of the sweet tea solution in the question above given that table sugar is
sucrose, C12H22O11 .
Expert Solution
Step 1
Ice tea volume = 24 ounce
1 ounce = 29.57 mL
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
