What is the pH of a 0.25 M solution of acetic acid, Ka = 1.8 x 105. What percentage of the acid is dissociated?
What is the pH of a 0.25 M solution of acetic acid, Ka = 1.8 x 105. What percentage of the acid is dissociated?
Basic Clinical Laboratory Techniques 6E
6th Edition
ISBN:9781133893943
Author:ESTRIDGE
Publisher:ESTRIDGE
Chapter1: The Clinical Laboratory
Section1.8: Laboratory Math And Reagent Preparation
Problem 8RQ
Related questions
Question
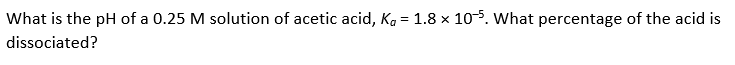
Transcribed Image Text:What is the pH of a 0.25 M solution of acetic acid, Ka = 1.8 x 105. What percentage of the acid is
dissociated?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Recommended textbooks for you



Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning



Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning