What is the product of the following reaction? HO -H H OH Pt H₂ но -H H OH CH₂OH Select one: a. H H -H H OH но -H H -OH CH₂OH b. CH₂OH H H H -OH но -H H OH CH₂OH COOH HO H H -OH HO H H OH
What is the product of the following reaction? HO -H H OH Pt H₂ но -H H OH CH₂OH Select one: a. H H -H H OH но -H H -OH CH₂OH b. CH₂OH H H H -OH но -H H OH CH₂OH COOH HO H H -OH HO H H OH
Chapter23: Carbonyl Condensation Reactions
Section23.SE: Something Extra
Problem 69AP: Fill in the missing reagents a-h in the following scheme:
Related questions
Question
Chemistry
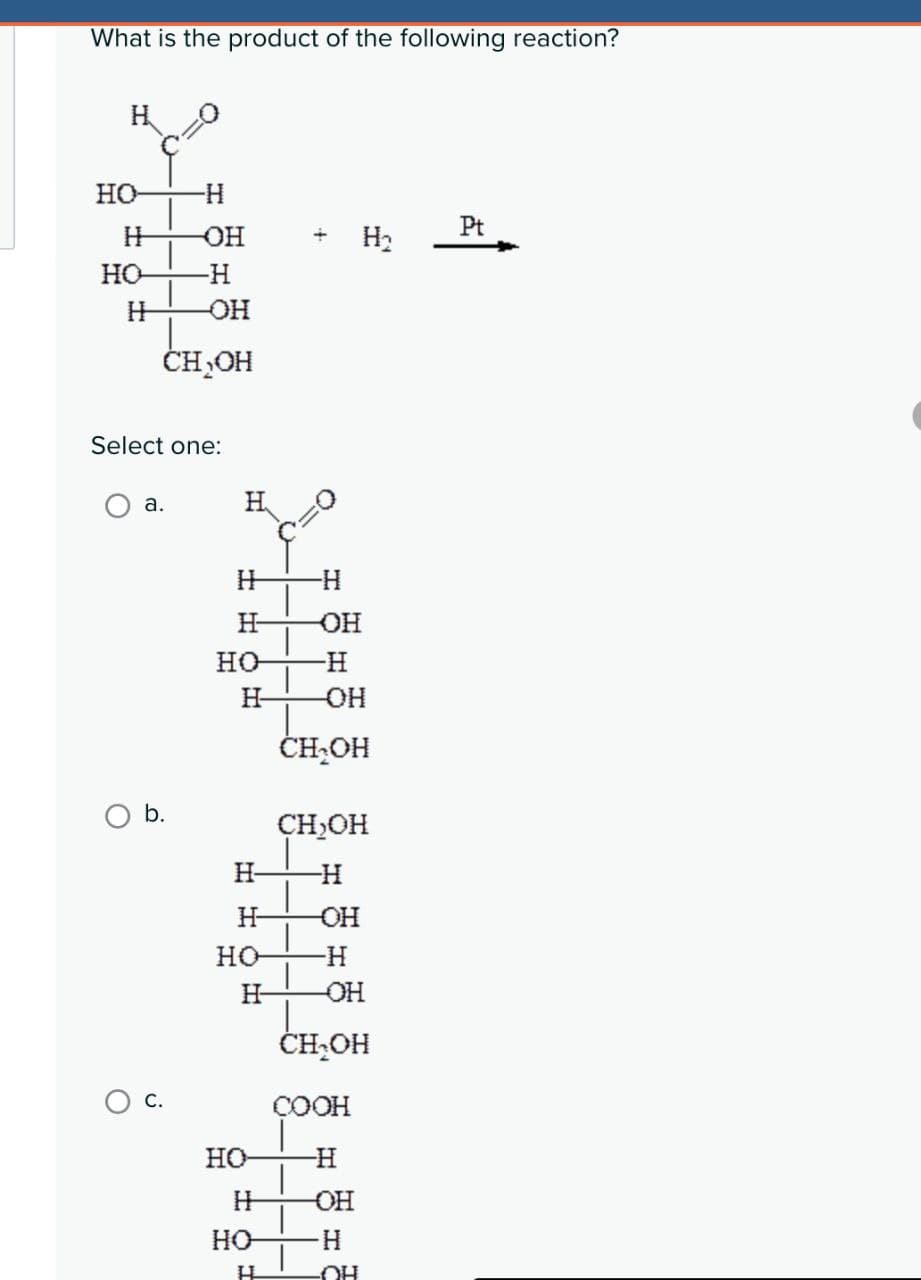
Transcribed Image Text:What is the product of the following reaction?
HO
-H
H
OH
Pt
H₂
но
-H
H
OH
CH₂OH
Select one:
a.
H
H
-H
H
OH
но
-H
H
-OH
CH₂OH
b.
CH₂OH
H
H
H
-OH
но
-H
H
OH
CH₂OH
COOH
HO
H
H
-OH
HO
H
H
OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you

