Chapter24: Amines And Heterocycles
Section24.SE: Something Extra
Problem 40MP
Related questions
Question
Transcribed Image Text:Check
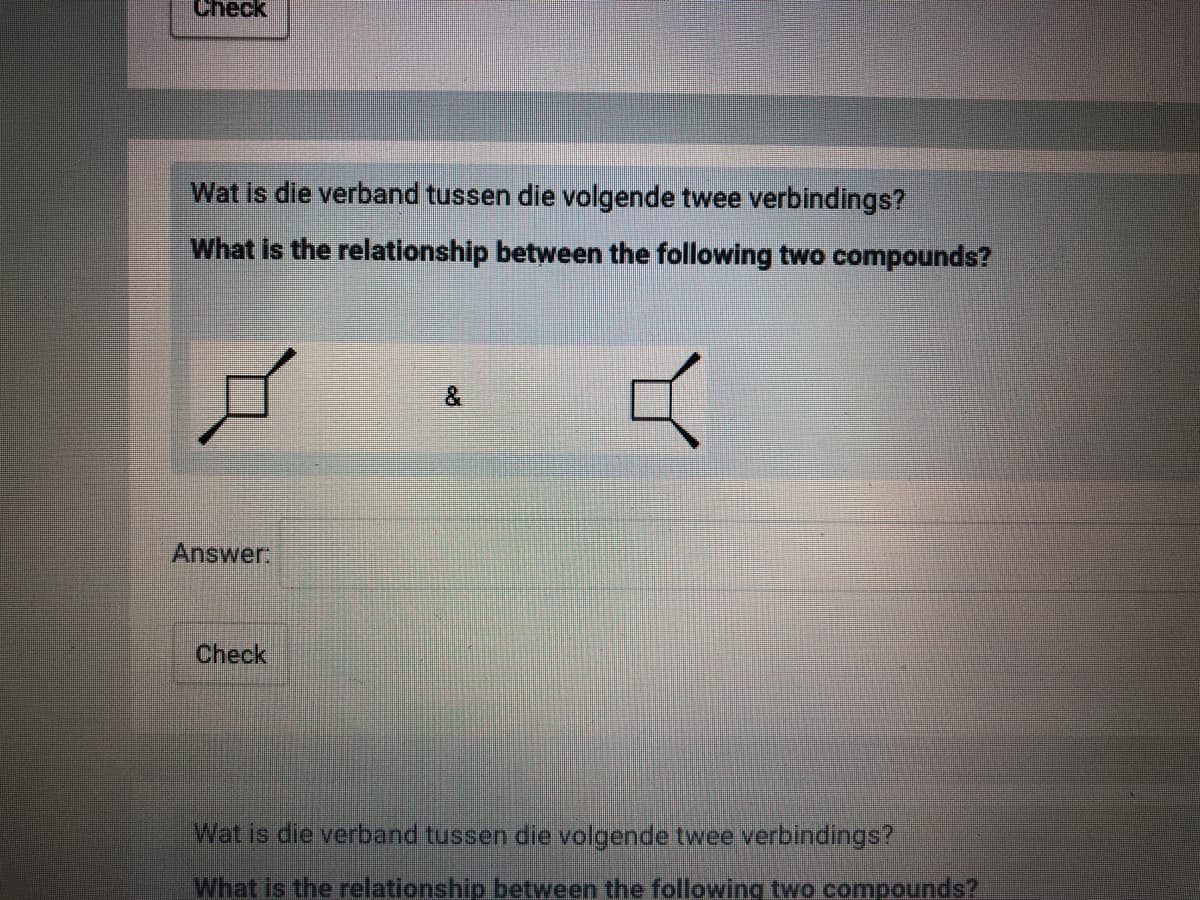
Wat is die verband tussen die volgende twee verbindings?
What is the relationship between the following two compounds?
Answer:
Check
Wat is die verband tussen die volgende twee verbindings?
What is the relationship between the following two compounds?
Transcribed Image Text:put tor Chegg Study |
2021-13623-178: SAM Ceng
+
1/ Natural Sciences
Chemistry And Polymer Science / Chemie- Chemistry - 144
Konsep Vasvrae Co
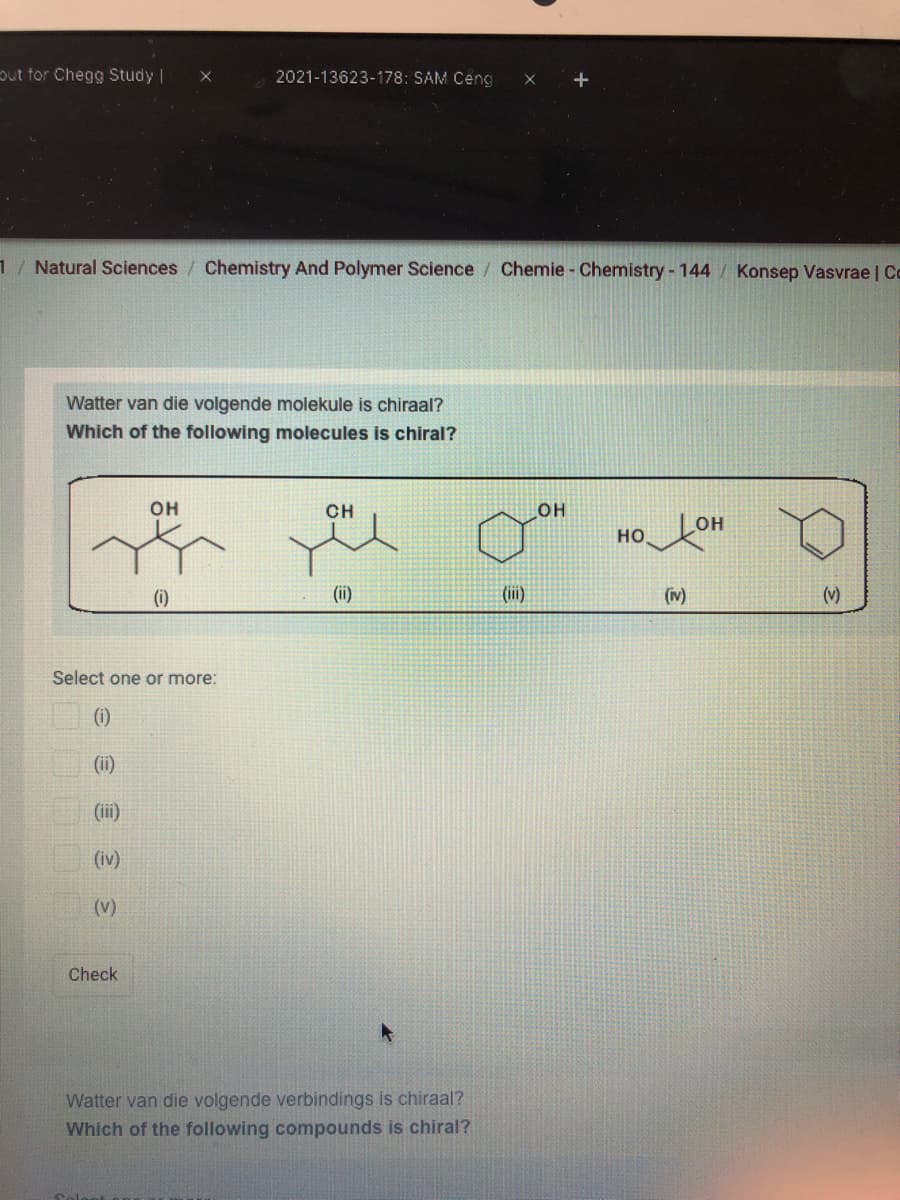
Watter van die volgende molekule is chiraal?
Which of the following molecules is chiral?
он
CH
он
он
но.
(i)
(ii)
(ii)
(v)
(v)
Select one or more:
(1)
(ii)
(iii)
(iv)
(v)
Check
Watter van die volgende verbindings is chiraal?
Which of the following compounds is chiral?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

