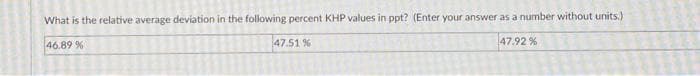
What is the relative average deviation in the following percent KHP values in ppt? (Enter your answer as a number without units.) 46.89 % 47.92% 47.51 %
Q: What is the major product from the reaction shown? OH (A) H₂O -CH₂CH3 H
A: Alkene gives hydration reactions in presence of an acid catalyst. The reaction follows an…
Q: Figure 1 represents a 1.0 L solution of sugar dissolved in water. The dots in the magnification…
A: Given,The volume of a initial solution = 1.0 LThe final volume after adding 1.0 L of water is = 2.0…
Q: How many grams of sodium chloride is needed to make 250 ml of a 125mM solution.
A: Concentration of the solution = 125 mM = 0.125 M = 0.125 mol/LVolume of the solution = 250 mL =…
Q: [References) Use the References to access important values if needed for this question. How many…
A: Given,(i) The reaction: Iron (s) + Chlorine (g ) Iron (III) chloride volume of chlorine gas =…
Q: Draw the product of the reaction shown below. Use wedge and dash bonds to indicate relative…
A: Oxidation with KMnO4 is most widely used method for the cis -hydroxylation of alkenes.
Q: A chemist prepares a solution of nickel(II) chloride (NiCl₂) by measuring out 79. g of nickel(II)…
A: Answer:
Q: Calculate the volume in milliliters of a 0.594 mol/L barium acetate solution that contains 75.0 mmol…
A:
Q: Part A 1.386 g Zn, 5.381 g I Express your answer as a chemical formula. | ΑΣΦ Part B A chemical…
A: The empirical formula is a representation of a compound's composition in the simplest whole-number…
Q: Classify the following reaction and balance the equation by entering the smallest possible integer…
A: Type of reaction = ?
Q: Interpert the IR for hydrobenzoin
A: IR spectroscopy is mainly used for the identification of functional groups present in organic…
Q: Oxygen gas reacts with powdered aluminum according to the following reaction:…
A: The given balanced chemical equation is 4Al(s)+3O2(g)→2Al2O3(s)The mass of aluminium reacts = 54.1…
Q: Draw the structure(s) of the cycloalkanes with the chemical formula C7H₁4 that DO NOT exhibit…
A: The molecular formula of the compound is C7H14. This molecular formula confirms that the molecule…
Q: What is Sº for B in the reaction 5 A 4 B if AS°(rxn) =-194.4 J/mol• K? [S° (A) = (220.0 J/ mol .K)]
A: Thermodynamics is branch of chemistry in which we deal with amount of heat evolved or absorbed by…
Q: What is the missing reactant? من ال LDA -78°C THF ? CH3
A: Given is organic reaction.In given reaction alpha alkylation of ketone is done.
Q: Help with a. Calculate the ka, concentration of H+ and pH. please show the algebraic process to get…
A: We have to calculate Ka, concentration of H+ and pH of the solutions.
Q: The expression A (g) + B (s) C (g) + D(g) O [B] O [C] Ο [A] O [D] of Keg for the following reaction…
A: The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical…
Q: Hydrogen is manufactured on an industrial scale by this sequence of reactions: CH₂(g) + H₂O(g) = CO…
A: The equilibrium constant of a chemical reaction gives a relationship between the reactants and the…
Q: Draw the skeletal structure of cis-pent-2-ene.
A: The organic compounds are named using a system that is universally accepted.The universally accepted…
Q: how to get molar enthalpy change of reaction for 2H20(g) sample: 2 H2 (g) + O2 (g) -> 2 H20 (g)…
A: The enthalpy change for the formation of one mole of a compound from its elements in their most…
Q: Liquid hexane reacts with gaseous oxygen gas to produce gaseous carbon dioxide and gaseous water. If…
A: Weight of CO2 = 67.5 gm = Experimental yield Weight of Hexane = 84.4 gmWeight of Oxygen = 98.3…
Q: Starting from the Newman projection below, rotate only the back carbon to provide the structure in…
A:
Q: What type of elimination with happen in the reaction below? Br Me Et Neither Zaitsev Hofman O…
A:
Q: Br 3C CH3 C₂H5 -H -OH Structure A Br HO H CH3 -CH3 -C₂H5 Structure B C₂H5 The relationship between…
A:
Q: What is the JUPAC Nome CH₂ CH₂ CH₂ CH₂ H H3C H Ch₂ CH₂ CH3 CH₂CH₂CH₂CH₂
A:
Q: or reaction A KOLBU or reaction B MeOH/heat [Select] MeOH/heat or reaction C EtOH/heat B C KOLBU…
A: Given are organic reactions.Reaction A is elimination reaction.Reaction B is elimination…
Q: Classify each chemical reaction: KBr(aq) + AgNO3 (aq) 1 reaction KNO,(aq) + AgBr(s) CH, OCH (1) +…
A:
Q: A weather balloon is inflated to a volume of 26.8 L at a pressure of 739 mmHg and a temperature of…
A: Initial volume of balloon = 26.8 LInitial pressure = 739 mmHg The inital temperature = Final…
Q: need to dilute 20.0 mL of a 1.40 M solution of LiCN to make a 0.0555 M solution of LiCN. What is the…
A: Initial Molarity (M1) = 1.40 M Final Molarity (M2) = 0.0555M Initial volume (V1) = 20 mL Let…
Q: Draw three (generic) amino acids in a row using arrows to show where the peptide bonds will form.…
A: The question is based on the concept of amino acids.We need to write 3 amino acids and explain the…
Q: To preview image for reaction click here B B To preview the image for energy diagram click here BL…
A: In given reactions alkyl bromide is converted into alkene in presence of methanol and heat.So, this…
Q: Draw the major product of this reaction. Ignore inorgani- byproducts and the alcohol side product. O…
A: The PhMgBr is called Grignard reagent, acts as a nucleophile or sometimes as a base (if acidic…
Q: What is the product of the following reaction? HQ Br X A C NaH НО B D *****
A: In the above reaction, the organic compound as starting material contains bromine atom and hydroxyl…
Q: A B Energy Br Energy -Br TM CH3OH heat ||| CH3OH heat Reaction Coordinate Energy+ Energy Reaction…
A: The given reactions are shown below.We have to answer the below given questions.
Q: Draw the major monobromination product when the alkane shown is subjected to radical bromination at…
A: The reaction of n-butane with bromine in the presence of light is an example of a photochemical…
Q: What is the product of the following reaction sequence? Д HO HO I НО CI OH Li (2 eq) IV Cl H II – L…
A: Given is organic reaction.Alkyl halide reacts with lithium to form organo lithium reagent.
Q: Using Cahn-Ingold-Prelog rules, rank these substituents from highest priority to lowest priority.…
A: Given,The substituents are:
Q: For the following reaction step, indicate which pattern of arrow pushing it represents. H proton…
A: A reaction mechanism step in which a proton is removed from one species and accepted by another…
Q: Which is the major organic product of the reaction below? OA OO B C OD A OH H₂0 ? HO B OH Тон 4 OH D
A:
Q: The following chemical reaction takes place in aqueous solution: CuSO4(aq) + (NH) S(aq) → CuS(s)+…
A:
Q: All of the following are anti-addition reactions with alkenes EXCEPT? 1. MCPBA, 2. H3O+ All…
A: A syn addition is an addition reaction of an alkene in which the net reaction is the addition of two…
Q: you carry out the reaction between table salt ( NaCl ) and ammonium phosphate ( (NH4)3PO4 ) in 100.0…
A: The reaction between sodium chloride and ammonium phosphate is a double replacement reaction,…
Q: 2. Are these compounds activated towards EAS (do they have an ERG?) or are they deactivated towards…
A: Given are two organic compounds.Electron donating groups act as activating groups.Electron…
Q: What are the units of k, for the following rate laws? b.k=kr[A] a) rate = kr[A]º b) rate = kr[A] c)…
A: Unit of rate constant = (concentration)1-n time-1 n = overall order of the reaction
Q: A chemist prepares a solution of potassium iodide (KI) by measuring out 180. g of potassium iodide…
A: The formula to calculate concentration of solution is => concentration of solution = moles /…
Q: Use the References to access important values if needed for this question. A dollar bill is found to…
A: Dimensional analysis is used to convert a physical quantity given in one type of unit to some other…
Q: Consider the reaction shown below. Classify compound Il as which of the following: OH 0:f H₂C CH3 40…
A: According to Bronsted -Lowry acid base concept, an acid is a substance which can donate proton(H+)…
Q: What is the major organic product of the following reaction? CH3 OH 1. (R)-CBS reagent 2. H2O "I ОН…
A: The reaction you provided involves the use of (R)-CBS (Corey-Bakshi-Shibata) reagent and water (H2O)…
Q: 1. IS THE RELATIONSHIP: the same molecule, enantiomers, or dia skriemers H3CH₂C H3C H H b) но C\ р 0…
A: If two stereoisomers are non-superimposable images of one another, then they are said to be…
Q: Calculate the root mean square (rms) average speed of the atoms in a sample of xenon gas at 0.13…
A: The root mean square (rms) of the atoms in a sample of xenon gas is given by the average speed of…
Q: Starting from the left side moving to the right side of the molecule identify the chiral…
A: Rules to Prioritise substituents present at chiral centre according to Cahn -ingold prelog system…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- An analysis of city drinking water for total hardness was done by two students in the laboratory and produced the following results (in ppm CaCO3): Student A: 228.3, 226.4, 226.9, 227.1, and 228.6. Student B: 229.5, 226.1, 230.7, 223.8, and 227.5What is the 95% confidence interval for the mean?A volumetric calcium analysis on samples of the blood serum of a patient believed to be suffering from a hyperparathyroid condition produced the following data: mmol Ca/L = 3.10, 3.08, 3.28, 3.15, 3.26, 3.12, 3.14, 3.18, 3.25, 3.11, 2.95. Calculate: Mean Standard deviation Coefficient of variation What is the 95% confidence interval for the mean of the data, assuming no prior information about the precision of the analysis? Apply the Q test to the following data sets to determine whether the outlying result should be retained or rejected at the 95% confidence level. ( work on the first measurements according to your case)Analysis of several plant-food preparations for potassium ion yielded the following data: Sample Percent K+ 1 3.90, 3.96, 4.16, 3.96 2 4.48, 4.65, 4.68, 4.42 Find the mean ; standard deviation variance and RSD for each sample.
- In quality management, it is very important that you have a good background knowledge in both descriptive and inferential statistics. Since you already took up Biostatistics and Epidemiology during your Second Year, answer BRIEFLY the following questions on basic statistics: 1. Compare and contrast the three most common measures of central tendency mean, median and mode. 2. Differentiate standard deviation from coefficient of variation. 3. What is T-test?What is the relative error in the ppt of the following data if the accepted value is 70.05 M? 70.24 M, 70.22 M and 70.10 MAnalysis of several plant-food preparations for potassium ion yielded the following data: Sample Percent K+1 6.02, 6.04, 5.88, 6.06, 5.822 5.29, 5.13, 5.14, 5.28, 5.20 The preparations were randomly drawn from the same population.(a) Find the mean and standard deviation s , variance , andCV for each sample.
- Estimate the absolute deviation(or uncertainty) for the results of the following calculations. Round each result so that it contains only significant digits. The numbers in parentheses are absolute standard deviations. Finally, write the answer and its uncertainty.a.) Y= 6.75 (± 0.03) + 0.843(±0.001) - 5.021 (±0.001) = 2.572b.) Y= 19.97(± 0.04) + 0.0030(±0.0001) + 4.29(±0.08) = 24.263c.) Y= 143(± 6) - 64(±3) = 5.9578 x10-21249 (±1) +77 (±8)A powder was prepared containing 3.00% NaCN and 97.00% NaCl. A sample obtained from that mixture containing 7.374×10^5 particles weighs 10.0 g. Determine the number and percent relative standard deviation of NaCN particles from a sample of the mixture weighing 6.30 g. I need nNaCN particles and %RSDTo test the quality of the work of a commercial laboratory, duplicate analyses of apurified benzoic acid (68.8% C, 4.953% H) sample were requested. It is assumed that therelative standard deviation of the method is sr → = 4 ppt for carbon and 6 ppt forhydrogen. The means of the reported results are 68.5% C and 4.882% H. At the 95%confidence level, is there any indication of systematic error in either analysis?
- The results obtained by two analysts for the lead content of samples obtained at different certified points in Rustenburg are as follows: Analyst 1 – 79.22, 79.41,79.66, 79.45 Analyst 2 – 79.09, 79.08, 79.25, 79.13, 79.10, 79.19 Using the equation, calculate the following 1.the standard deviations 2 .the mean values of the two sets of data and 3. s-pooledThe following results were obtained in the replicate determination of the lead content of a blood sample: 0.752, 0.756, 0.752, 0.751, and 0.760 ppm Pb. Calculate the standard deviation and variance of this set of dataFind the mean sand standard deviation of each sample.





