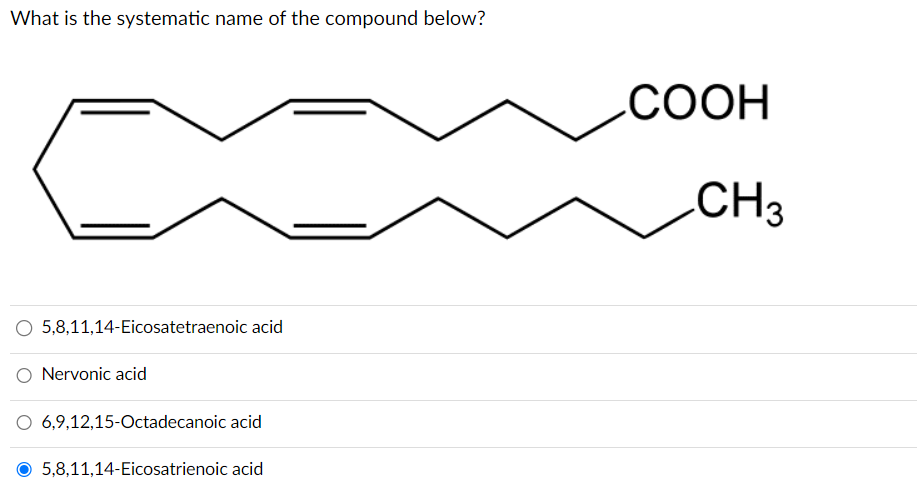
What is the systematic name of the compound below? O 5,8,11,14-Eicosatetraenoic acid O Nervonic acid O 6,9,12,15-Octadecanoic acid 5,8,11,14-Eicosatrienoic acid COOH CH3
Q: The body uses ketone bodies as fuel “during times of extreme starvation” and that ketosis “is an…
A: Acetyl CoA produced from beta oxidation of fatty cids has two possible fates: enter the TCA cycle…
Q: 1.0.1 mL of a protein solution of concentration of 11 mg/mL was diluted to a total volume of 4.0 mL…
A: The four types of biological macromolecules are nucleic acids proteins lipids and carbohydrates.…
Q: Explain the role/importance of the localization of GTPase-activating protein (Ran-GAP) in BOTH…
A: The Ran protein is a member of the Ras superfamily of small GTPases. Ran regulates nucleocytoplasmic…
Q: Explain in depth the Regulation of Glycogen Metabolism by Insulin and Glucagon.
A: Glycogen is a multibranched polysaccharide of glucose that serves as a form of energy storage in…
Q: What will happen if the enzyme renin is mixed with 1N HCL before adding milk? Little coagulation O…
A: Rennin is a proteolytic enzyme or protein digesting enzyme also know as chymosin. If the enzyme…
Q: Write a note on immunoglobulins. Write briefly about conjugated proteins.
A: The immune system is a complex network of cells, tissues, and organs that work together to protect…
Q: Define carbohydrates. Write their biological functions.
A: Introduction: Carbohydrates are one of the essential biomolecules that play a vital role in many…
Q: Which amino acids are in the figure?
A: Amino acids are the building blocks of proteins. They are organic compounds made up of an amino…
Q: 38. Which test can distinguish between sesame oil and corn oil? a. Burchard test b. Baudouin test c.…
A: Fatty acids (FA) are building blocks of fats and oils in our body, it consists of a carboxylic acid…
Q: Activity B. You perform a titration of an amino acid and obtain a plot. 21- What is equivalent…
A: Amino acids are biomolecules that have an amino group and a carboxyl group linked to the same carbon…
Q: What are eicosanoids? Mention their biomedical importance.
A: Introduction: The below answer provides an explanation of eicosanoids, their functions, and their…
Q: Provide an example of covalent catalysis in the mechanism of chymotrypsin. What is the nucleophile…
A: Chymotrypsin is a digestive enzyme that hydrolyzes peptide bonds, specifically at the carboxyl side…
Q: 6. Which of the following process are mainly enthalpy driven and which are mainly entropy driven? a.…
A: An entropy-driven process is a process that occurs spontaneously due to an increase in the entropy…
Q: Draw a two-stranded anti-parallel -sheet where each strand is 5 amino acids long. Use “R” to show…
A: The β-sheet is one of the most common secondary structures in proteins. The β-pleated sheets are…
Q: Define phospholipids. Classify them with suitable examples and state their functions.
A: Introduction:Phospholipids are an important class of lipids that make up a major component of cell…
Q: How many amino acids make up the unknown protein? How many nucleotides in the mRNA transcript? How…
A: Proteins are polymers of amino acids linked by peptide/amide bonds by the condensation reaction…
Q: Given the following enzyme pathway, A→B→C→D The AGO' for each of the individual reactions is as…
A: Change in Gibbs free energy is a state function, meaning it does not depend on the path taken by the…
Q: SDS-PAGE 28) Reading DNA, RNA and protein sequences 29) Cloning vectors
A: SDS-PAGE is a powerful tool for studying protein structure and function, as well as for purifying…
Q: Classify the lipoproteins according to the content of each of the lipid fractions it contains.…
A: Lipoproteins are complexes of lipids (chemically diverse group) and proteins (polymers made up of…
Q: 8. The human diet includes "non-digestible" polysaccharides, or fiber, that can actually be digested…
A: Hydrolases are a class of enzymes that catalyze the hydrolysis of chemical bonds through the…
Q: Write the names and structure of basic amino acids.
A: Introduction: In this response, I provided the names and structures of basic amino acids. Amino…
Q: You are studying COVID-19 which infects bat cells through an interaction between a lysine residue on…
A: Proteins are large biomolecules made up of amino acid residues linked via a peptide bond. Amino…
Q: How sucrose and maltose differ with respect to a. Structure b. Source c. Function
A: Introduction: Sucrose and maltose are two common disaccharides that are important sources of energy…
Q: Between your evening meal and breakfast, your blood glucose drops and your liver becomes a net…
A: Glucose is the primary source of energy for the body. Glucose is metabolized through the glycolytic…
Q: Polyprotic acids such as H3PO4, can act as acid-base buffers ℗ only in combination with polyprotic…
A: D. at pH values around any of their pKa's. Polyprotic acids are acids that can donate more than one…
Q: Recombinant DNA construction involves, a) Cleaving DNA with a restriction endonuclease and joining…
A: Recombinant DNA technology involves the manipulation of DNA to create new combinations of genetic…
Q: Which of the following molecules is least likely to be a fatty acid? O H₂C-(CH₂)14-COOH
A: Fatty acids are the building blocks of the fats (like triglycerides, phospholipids, etc.) in our…
Q: Asp residue (both of which are essential for catalysis) with pK, values of 5.9 and 4.5,…
A: In simple terms, pH indicates whether a substance is acid or base. pKa determine how strong is an…
Q: 25. Which lipoprotein prevents the formation of plaque in the artery? a. HDL b. VLDL c. LDL d.…
A: Lipoproteins are made up of fats and proteins. The main function of the lipoproteins is to circulate…
Q: 5 Applications of Enzymes in Household Products
A: Enzymes are biological molecules that catalyze (speed up) chemical reactions in living organisms.…
Q: 2ul St.1 St.2 St.3 St.4 Absorbance 595nm 0.926 0.444 0.421 0.462 0.932 0.412 0.407 0.393 Average Abs…
A:
Q: Can you please help me to make this table profesional, I need two table the other which will be…
A: Proteins are polymers of amino acids linked by peptide/amide bonds by the condensation reaction…
Q: How does Stoichiometry in Chemical Reactions relate to our daily life activities? How also relevant…
A: Stoichiometry is the field of chemistry that uses the relationships between reactants and/or…
Q: How does the structure of chymotrypsin enable it to cleave only peptide bonds that are adjacent to…
A: Introduction: Chymotrypsin is a type of enzyme that specifically cleaves peptide bonds adjacent to…
Q: When performing a separation by ion-exchange chromatography, the rate of elution of an amino acid is…
A: Ion-exchange chromatography is a technique that uses charge to separate and purify molecules. The…
Q: Classify proteins on the basis of solubility giving examples for each class.
A: Introduction: Proteins are complex molecules that play essential roles in many biological processes.…
Q: A 16-year-old female is recently diagnosed with a deficiency of muscle glycogen phosphorylase. She…
A: Type V glycogen storage disease (also McArdle disease) is due to the deficiency of the enzyme muscle…
Q: Identify the mutation(s) that lead to the most loss in transcriptional activity, and discuss whether…
A: Consensus sequence are regions in genome that are conserved i.e. throughout the course of millions…
Q: Name the sulphur containing amino acids. List two disorders of their metabolism.
A: Introduction:Sulfur-containing amino acids are crucial building blocks of proteins in the human…
Q: the peptide sequence in single letter code is T N C H P, please hand draw a peptide diagram…
A: The primary structure of proteins tells us the sequence of arrangement of amino acids in a…
Q: Which of the following enzyme is used in PCR? a) EcoRII b) EcoRI c) Tag DNA polymerase d) HRP
A: PCR (Polymerase Chain Reaction) is a widely used technique in molecular biology that amplifies a…
Q: 33. In bacterial cells, a) glucose-6-phosphate; ATP b) ADP; phosphoenolpyruvate c)…
A: Since you have posted multiple questions, we will provide the solutiononly to the first three…
Q: You have discovered a new amino acid. You find that its side has a pKa-8.00 what percent of this…
A: The pKa of amino acid refers to the equilibrium between the deprotonated neutral nitrogen and the…
Q: Give one example each of di-, tri- and tetra-saccharides.
A: Introduction: Disaccharides, trisaccharides, and tetrasaccharides are complex carbohydrates made up…
Q: Calculate the number of ATPATP generated from one saturated 1212‑carbon fatty acid. Assume that each…
A: Oxidation of fatty acids begins with beta-oxidation of the acyl-CoA derivative of the fatty acid.…
Q: [10'] For a bacteriophage T7, the following data at zero concentration have been obtained:…
A: In order to find the molecular mass, first we need to convert the Sedimentation coefficient value…
Q: Find the sequence for protamine and determine how many arginines it has. Based on the predicted…
A: Protamine is a peptide of usually 49 or 50 amino acids long which is used for packaging of DNA in…
Q: How do you identify the N vs C terminals? For the first part of the question, do you then list the…
A: Amino acids are the monomers of a polypeptide or protein. Amino acids contain an amino (-NH2) group…
Q: Which of the following membrane lipids have a direct glycosidic linkage between the head-group sugar…
A: Glycolipids are a type of membrane lipid that have a direct glycosidic linkage between the…
Q: Invertase works on a _________ molecule, specfically ___________ and it does a _________ reaction…
A: Carbohydrate monomers or monosaccharides are built of carbon, hydrogen and oxygen where the ratio of…
Step by step
Solved in 3 steps with 1 images

- The correct systematic name for the fatty acid below is: a) 3 octadecatrienoic acid b) 9 octadecatrienoic acid c) 3,6,9 octadecatrienoic acid d) 9,12,15 octadecatrienoic acid e) None of the abovewhat is the name for chemical formula Xr3(DG2)4 Xr- xraium DG- dogiteGiven the organic compounds function group below, provide their general formula. Answer 1,2,3,4,5,6,7,8,9 and 10. Thank you
- What is the empirical formula for C3H6O3? C3H6O3 C6H12O6 CH2O None of theseWhat is the structure of the general class of molecules that Ibuprofen belongs to? What is a specific structure of the compound (drawn in line bond format). A sepertate structure included the compound (Ibuprofen) with highlighted functional groups. A context and relevance of the class of molecules and the specific compound for Ibuprofen.Draw the structures of the following acids:(a) 2-Ethyl-3-hydroxyhexanoic acid (b) m-Nitrobenzoic acid
- Give the molecular formula of the functional group that is missing. a. NH+ b. CH3 c. COOH d. C6H12O6What is the product of intramolecular dehydration of pentanol-2 CH3CH(OH)CH2CH2CH3? Select one:a. Pentene-2b. Pentene-3c. Pentene-1d. PentaneThe following chemical reactants produce the ester ethyl ethanoate (C4H8O2): C2H6O + CH3COOH What type of reaction occurs to make ethyl ethanoate? condensation hydrolysis combustion acid-base reaction
- Decide which N atom in each molecule is most basic, and draw the product formed when each compound is treated with CH3CO2H. Zolpidem (trade name Ambien) is used to treat insomnia, whereas aripiprazole (trade name Abilify) is used to treat depression, schizophrenia, and bipolar disorders.Relate the molecular properties to physicochemical properties of the following lauric acid stearic acid oleic acid methyl laurate methyl oleate a-tocopherol hexadecane cholesterol estrogenIdentify the acid on the left and its conjugate base on the right in the following equations:(a) HOCl + H2O ↔ H3O+ + OCl-(b) HONH2 + H2O ↔ HONH3+ + OH-(c) NH4+ + H2O ↔ NH3 + H3O+(d) 2HCO3-2 ↔ H2CO3 + CO3-2 (e) PO4-3 + H2PO4- ↔ 2HPO4-2

