Q: The acid dissociation K of acetic acid (HCH3CO2) is 1.8 × 105. -4 Calculate the pH of a 4.1 × 10 M…
A: The objective of this question is to calculate the pH of an acetic acid solution given its molarity…
Q: Write the mechanism for the following reaction step using curvedarrows, and draw the result of those…
A: Step 1: DecarboxylationDecarboxylation goes through a concerned six membered transition state.Normal…
Q: How would you use Friedel Crafts acylation, Clemmensen reduction, and Gatterman-Koch synthesis to…
A: 1.2.
Q: 1. What would happen to the spinach TLC plate in the lecture if the plate was run with 100% acetone?…
A: 1. If a TLC (Thin Layer Chromatography) plate containing spinach extract was run with 100% acetone,…
Q: Each row of the table below describes an aqueous solution at 25 °C. The second column of the table…
A: Step 1: A. The initial solution contains only water (H2O), making it neutral. When KClO4 is added,…
Q: Refer to the following physical model to answer the questions. White Sulfide ion Blue Zinc ion…
A: The background was white that's why used grey color for Sulfide Ion.
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Out of the potential unknowns in this experiment, which one would be the best choice to create a…
A: The objective of this question is to identify the best choice among the given options to create a…
Q: Predict the product CI. Base
A: Alkyl halide reacts with the base and undergoes an elimination reaction, terminal proton is most…
Q: Modify the given structure of the starting material to draw the major product. Edit Drawing
A: Step 1. the reaction of propylene oxide involves the reaction of propane with oxygen to produce…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Step 1: DecarboxylationDecarboxylation goes through a concerned six membered transition state.Normal…
Q: Question 33 Predict the FINAL (?) product for each of the following reaction or synthetic chain: A B…
A:
Q: OCN-(aq) + OCI-(aq) → CO₂2-(aq) + N2(g) + Cl(aq) (basic media)
A: The objective of the question is to balance the given chemical equation in a basic medium.
Q: Can you explain the problem?
A: Step 1: Step 2: Step 3: Step 4:
Q: What are the formal charges on terminal N, central N, and O atoms in the following molecule? OA-2,…
A: Step 1: Formula for calculating formal charge on an atom : Formal charge = (Numbers of valence…
Q: 3. What changes occur to the atomic number and mass of a nucleus during each of the following decay…
A: The symbol of an element is expressed as, A XZ Where, X = the atomic symbol of the element (like…
Q: Question 31 Predict the FINAL (?) product for each of the following reaction or synthetic chain:…
A: Step 1: Step 2:Mechanism Step 3: Step 4:
Q: Right answer and please fast expert solutions
A: Step 1:
Q: A student artificially pumped 1 mole of each substance seen below in a piston with a moveable lid…
A: The reaction:2 C3H7OH(g) + 9 O2(g) ←→ 6 CO2(g) +8 H2O(g) The number of molecules in reactant side =…
Q: (20 pts) Alcohol A undergoes a remarkable acid-catalyzed conversion to B. Me Me Me HO Me I Me S Me…
A: Step 1: Step 2: Step 3: Step 4:
Q: Course: College Chemistry Course code: CHM 141 Explain three points 1, 2 and 3(red color) based on…
A: Step 1: Step 2: Step 3: Step 4:
Q: When the Ag* concentration is 1.21 M, the observed cell potential at 298K for an electrochemical…
A: The objective of the question is to find the concentration of Cr3+ in an electrochemical cell…
Q: Use cell potential to calculate an equilibrium constant. Calculate the cell potential and the…
A: E0 Cell = E0 Cathode - E0 Anode = E0 reduction - E0 oxidation = 0.16 - 0.77…
Q: A chemistry graduate student is given 500. mL of a 0.50M chlorous acid (HCIO) solution. Chlorous…
A:
Q: The pro great expert Hand written solution is not allowed. No need to upload any image just give me…
A: Step 1:Rules for the IUPAC name of organic compounds:1. Identify the longest carbon chain.2.…
Q: 30. The following data were collected for a certain reaction of the type: A → products Time, min…
A:
Q: Synthesis. Supply the missing reagents required to accomplish each of the following syntheses. Be…
A:
Q: Living chain polymerization of monomer X follows the first-order kinetics. The corresponding…
A:
Q: A chemist fills a reaction vessel with 6.88 atm hydrogen (H2) gas, 7.31 atm oxygen (O2) gas, and…
A: Given: ΔfG°,kJ/molP,atm2H2(g)0.006.88+O2(g)0.007.31⇌2H2O(g)−228.61.19Step 1: Write the…
Q: Mangrove bark is extracted by treating the finely ground wood with hot water. The original bark…
A: Original bark:Moisture content = 7%Tannin content = 33%Soluble non-tannin material = 20%Residue…
Q: Please draw all steps for number 4 3. Show how using the tosyl chloride reagent from the alcohol…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Submit Problem 14 of 21 Draw the products of the reaction sequence shown below. Ignore inorganic…
A:
Q: None
A: The reaction involves the addition of HBr to safrole in the presence of water at 10°C. This reaction…
Q: Help solve 1,2,3,4,5.
A: 1.To find the standard Gibbs free energy change (ΔGrxn∘) for the ionization of acetic acid…
Q: From benzene, synthesize the following molecule. Show all steps of the forward reaction with…
A: Step 1: Step 2: Step 3: Step 4:
Q: Below is the resolved absorption spectrum of HBr for the vibrational transition v = 0 → V = 1;…
A: Our usual method of determining the effective spring constant and bond length from the resolved…
Q: Question 34 Predict the FINAL (?) product for each of the following reaction or synthetic chain:…
A: Step 1: Step 2:Mechanism Step 3: Step 4:
Q: Predict the major product of the following transformation: CO₂Et H₂O+ Heat C10H100 Modify the given…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: None
A: The reaction given is a hydroboration-oxidation reaction.In this type of reaction, an -OH group is…
Q: Predict the product(s) for this reaction. H-SCH2CH3 THF SCH2CH3 I SCH2CH3 || III = ?
A: Identify the Nucleophile and Electrophile: Within the molecule, recognize the functional group…
Q: Part A Account for the difference in the products of the following reactions: ф-оф-о Check all that…
A:
Q: Select the nonprotein nitrogen compounds from the listed analytes. Question 57 options:…
A: The objective of the question is to identify the nonprotein nitrogen compounds from the given list…
Q: In an attempt to synthesize compound C through a two-step process, a chemist discovered after…
A: Step 1:• In this reaction, Firstly base abstract the acidic proton to forms enolate ion which…
Q: Give the major product of the following reaction. O HCB3 Br Br Br Br مية Br Br Br Br OH منه Br Br2 ?…
A: Step 1:Introduction to question : Determine the major product of the reaction : Step 2: Introduction…
Q: What is the major organic product of the following reaction? 01 NH NH AICI
A: In electrophilic aromatic substitution, the position that an electrophile attacks is based on the…
Q: Predict the major product formed in the reaction below: Bu Me 1. BH3.THF 2. H2O2, NaOH A A B B © C D…
A: In hydroboration-oxidation reaction, the addition of an alcohol group proceeds in an…
Q: 4) Propose a synthesis 6 pts OH ? -ОН 5) Propose multistep syntheses for the following compounds…
A: The objective of the question is to propose a multistep synthesis for a given compound starting from…
Q: None
A:
Q: Consider three hypothetical solids:AX,AX2 and AX3 (each X forms X). Each of these solids has the…
A: Solution : Solubility is defined as the maximum amount of solute dissolved in an aqueous…
Q: 1 1/1 point Mercury (I) chloride has a Ksp of 1.3 x 10-18. Find its solubility in units of mole/L…
A:
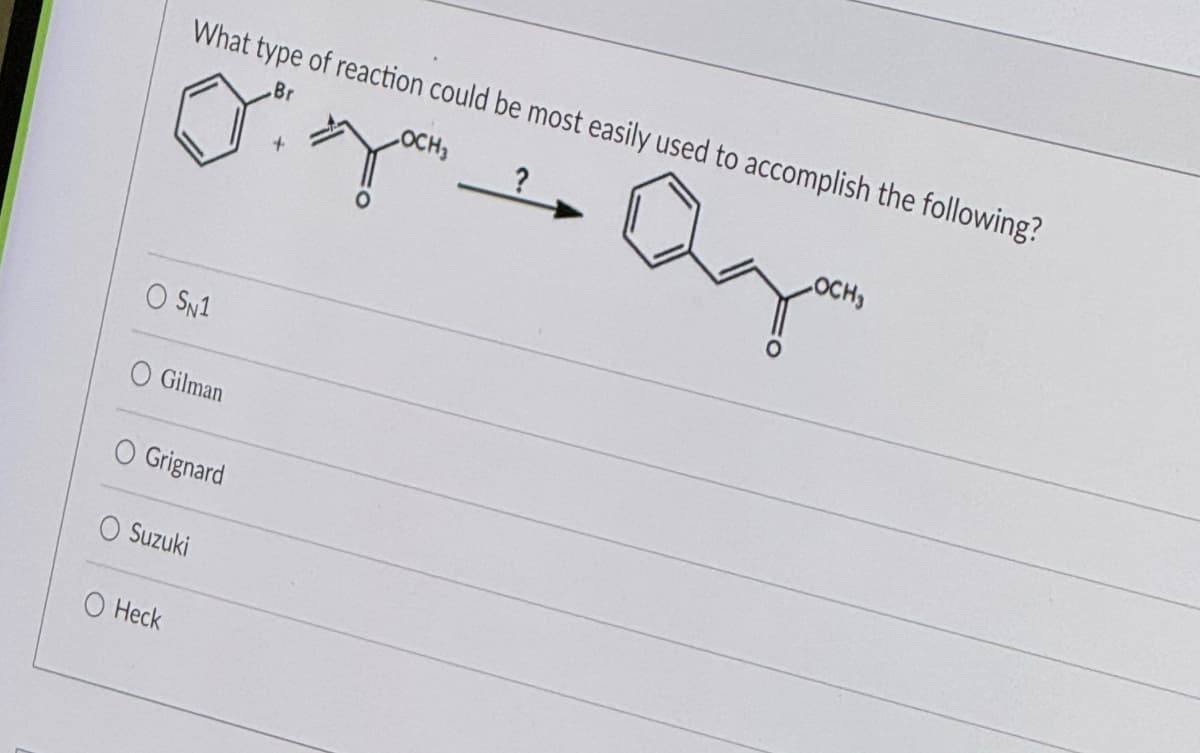
Step by step
Solved in 2 steps with 1 images

- Compute the amount of reagents which will be used for the preparation of the following reagents. e. 1.5 L of 0.04 M KMnO4. A colorless liquid, C4H6O, having a boiling point of 97-98°C was found to be soluble in water and also in ether. It gave a negative test for the presence of halogens, sulfur, and nitrogen. It did however, give a positive test to 2,4-dinitrophenylhydrazine reagent. It gave a negative result when treated with ceric nitrate solution and also Tollen’s reagent. Treatment with ozone followed by hydrolysis in the presence of zinc gave formaldehyde as one of the products. What is the structure and name of the colorless liquid?Pick the correct starting material and explain why the other ones are wrong and why the one picked is correct. Explain in detail with drawn out reactions.
- Write the reactions involved in the following reactions:(i) Clemmensen reduction(ii) Cannizzaro reactionNa2SO4 + BaCl2 → BaSO4 + 2NaCl Reaction Involved between Na2SO4 & BaCl2 ?Supply the missing reagent in the following reaction: (see attachment) Question 13.docx 13 KB A. Lindlar, H2, mCPBA B. O3 or KMnO4 C. fused KOH D. NH3 E. NaNH2
- what is the limiting reagent for a reaction between 4-nitrobenzic acid and 2 thionyl chloride MW 4-nitrobenzic acid:167.12 g/mol MW thionyl chloride: 118.97 g/molComplete the steps to these problemsAn unknown has a chemical formula of C4H10O. When chromic acid was added to the unknown, it turned green. When the Lucas reagent was added to a separate portion of the unknown, it turned cloudy in 5 minutes. When NaOH and KI/iodine was added to the unknown, a yellow precipitate formed. What is the structure of the unknown compound?



