Q: A polypeptide is shown below. Please answer the following questions. OH В A C H;N E H D H a. Match…
A: A polypeptide is a long, unbranched chain of amino acids joined by peptide/amide bonds. The peptide…
Q: Which of the following is a purine that forms 3 hydrogen bonds with its complementary base? (a) A…
A: Nucleic acids were first discovered by Miescher while studying the pus cells discarded from the…
Q: Which of the following are nitrogenous bases with the general structure shown in the picture?…
A: The nitrogenous base shown in the picture contains a bi cyclic ring which indicates that it is a…
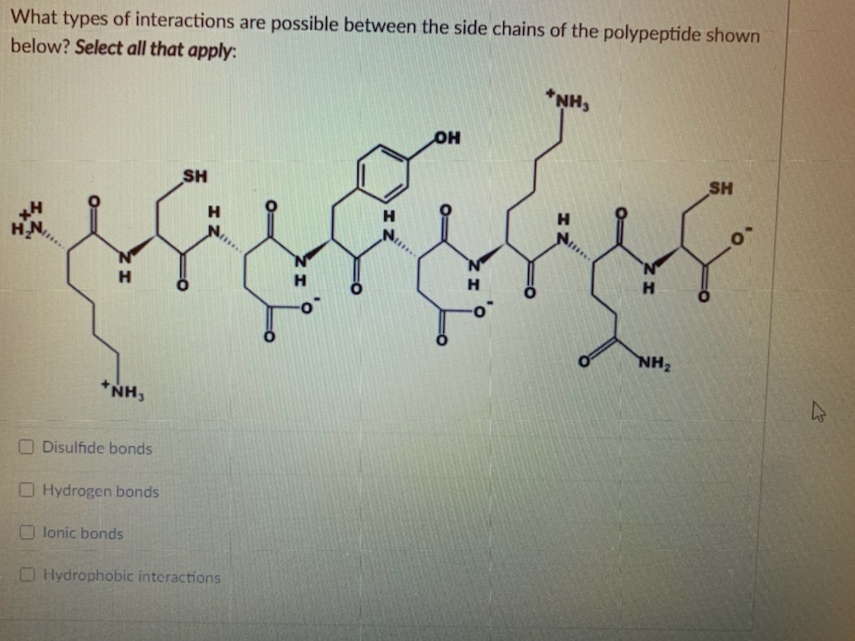
Q: Do any of the amino acid side groups shown below have the potential to form an ionic bond with any…
A: Ionic bonds are formed between the exchange of electrons between the donor and acceptor groups.
Q: Draw the following amino acids interactions: 1. H-bond - between -0H of Serine amino acid and C=0…
A: Between an electronegative atom with a lone pair of electrons and a hydrogen atom that is covalently…
Q: O H NH; CH3 CH; CH3 CH2 CH: CH2 OH H CH: CH2 H. H H. CH2 CH: NH- -C -N - -C- -A-C-N --C00 H. HO HO H…
A: When many amino acids joined with the help of peptide bonds forms the polypeptide. Amino acids that…
Q: Identify the following types of biomolecules and label and identify the functional groups
A: A molecule synthesized by cells or living organisms is referred to as a biomolecule. The major type…
Q: Which of the following amino acid structures would neither bear a positive nor a negative charge…
A: Amino acids are the molecules that contain amino as well as carboxyl group. Alpha-amino acids have…
Q: What form exists at the isoelectric point of each of the following amino acids: (a) valine; (b)…
A: The isoelectric point of amino acid is the pH at which amino acid has no net charge and shows no…
Q: What is the formation of 2 tripeptides of glycine? Where is the location of the peptide bonds and…
A: Proteins are polymers of amino acids. Amino acids sequence in a protein is guided by the nitrogen…
Q: Write the structure of the following peptides: a) glycyl alanine b) tyrosyl alanyl glycine c)…
A: The first peptide is composed of two amino acids that mean it is a dipeptide. The second peptide is…
Q: What type of intermolecular forces exist between the side chains of each of the following pairs of…
A: Amino acids are organic compounds that combine to form proteins. Amino acids and proteins are the…
Q: Creating tertiary structures of proteins involves the linking of amino acid functional groups with…
A: Proteins are the building blocks of the body. All the life forms consist of proteins in their cell…
Q: For each of the following, name an amino acid in which the R group contains it: a hydroxyl group, a…
A: Amino acid is the organic acid which is attached to all four different functional groups including…
Q: You have a solution with the mixture of the following peptides: 1) Wing-Cys-Gly 2)phe-tyr-trp-phe…
A: Proteins or polypeptides are made up of chain of amino acids. The name itself indicates that an…
Q: What is the two factors determining the primary structure of a polypeptide chain
A: The factors that determine the primary structure of the polypeptide chain include peptide bonds and…
Q: If the amino acids alanine and glycine react to form a peptide bond, is there more than one reaction…
A: Amino acids are made up of a central carbon atom, a carboxyl group, an amino group, and an R group…
Q: Which of the following are saponifiable lipids? (Recall that ester bonds are broken by base…
A: A saponifiable lipid is part of the ester functional group. They are made up of long chain…
Q: What parts of a polypeptide participate in the bonds thathold together secondary structure? Tertiary…
A: Proteins are biologically functional molecules that are comprised of one or more than one…
Q: Which of the following group(s) are present in natural amino acids: a. Imidazole group c. Indole…
A: Natural amino acids and the group present in them are:-
Q: Which of the following creates peptide bonds?
A: Peptide bonds are covalent bonds and is responsible for linking amino acids and forming polypeptide…
Q: Why is the formation of a peptide bond called a condensation reaction? Explain briefly
A:
Q: In the structure shown, which arrow is pointing to a peptide bond? OH LOH NH2 H OH HO-P=0 H. `A ÓH…
A: Peptide bond: It is a covalent bond used to join two amino acids to form a peptide chain. Generally,…
Q: Show a tertiary structure of ACGGC after a disulfide bond forms.
A: Protein folding is the physical process by which a primary sequence of amino acids or a linear…
Q: Which of the following shows a structure of an a-amino acid? OH H2N, NH2 NH2 H2N, он
A: Amino acids contain an amino group and a carboxylic group and due to that, they are called amino…
Q: List down which amino acids are capable of the following interactions in the tertiary structures of…
A: There are 20 standard amino acids are there which make up all the proteins inside the cell. Amino…
Q: The pKa of the side-chain imidazole group of histidine is 6.0. What is the ratio of uncharged to…
A: A molecule is fully protonated at low pH, as the pH increases more than pKa of an ionizable…
Q: The pH vs charge graph for a triprotic amino acid is shown below. Please answer the following…
A: A. This is histidine triprotic amino aciD. It's the triprotic, with a third pKa value associated…
Q: Draw the titrimetric profile of tyrosine and calculate its isoelectric pH
A: Tyrosine is a amino acid containing a aromatic hydroxyl side chain. Titration of tyrosine with a…
Q: Refer to the accompanying figure to answer the following question(s). R R EN-C-C-N-C-C-O-H H H А. В.…
A: Q. At which bond would water need to be added to achieve hydrolysis of the peptide, back to its…
Q: what are the r groups in proteins
A: Proteins are the basic building blocks that are formed by repeated linkage of amino acids through…
Q: Which of the following is the C-terminal amino acid in the polypeptide…
A: In a polypeptide chain, the amino acid consist of an amino group on the alpha carbon atom at one end…
Q: In the side chain of Lys, which of the following group is attached to the ε-carbon atom?…
A: Lysine, which is also denoted by K is one of the 20 essential amino acids. Lysine contains one…
Q: What kind of bond would you expect between the side chains of the following amino acids?(a) Cysteine…
A: Amino acids are molecules which when combined together forms a macromolecule known as protein. It…
Q: Which of the followings is not a structural polysaccharide?
A: Polysaccharides consist of hundreds or thousands of monosaccharide units. Polysaccharides are also…
Q: Refer to the structure of an oligopeptide below: Taking note of the component residues, would such a…
A: Amino acids are organic molecules. They are monomers of proteins. The alpha-amino acids take part in…
Q: What is the percent ionization of these amino acids? 1. Aspartame at pH 5.4 2. Glutamate at pH 11.8…
A: Since we are entitled to answer up to 3 sub-parts, we’ll answer the first 3 as you have not…
Q: With the following amino acid side chain can the side chain of threonine form hydrogen bonds? Q.)…
A: Hydrogen bonds are electrostatic interactions formed among the macromolecules. In a biological…
Q: dentify the following types of biomolecules and label and identify all of the functional groups
A: A structural segment of nucleic acid that possesses a sugar molecule like ribose sugar attached to a…
Q: The structure of the dipeptide Gly-Asn is given by The structures of the amino acids Gly and Asn are…
A: Glycine (Gly - 3 letter code ) and Asparagine (Asn) are amino acids. Amino acids are the building…
Q: What is the pH value where the protein molecule carries no electrical charge? a. isoelectric b.…
A: The pH refers to the indication of whether a substance is an acid or base. Acidic substances lie at…
Q: Which of these statements about the tertiary structure of a polypeptide is(are) untrue? You may…
A: Protein play wide variety of essential function in our body. They provide strength and structural…
Q: The structure of D-arabinose is shown below. How many stereoisomers are possible for this molecule…
A: Arabinose is an aldopentose sugar. Arabinose found in the nature as part of the polymers such as…
Q: The ring structure of the carbohydrate glucose is provided below. Map any chiral centers with a 1
A: Chiral centers are carbon atoms with 4 different substituents bonded to it. Glucose can exist in…
Q: What is meant by “polarity” of a polypeptide chain and by “polarity” of a chemical bond? How do the…
A: Anything which has distinct two or more ends is said to be polar. Amino acids have an alpha carboxyl…
Q: How do the following noncovalent interactions help to stabilize the tertiary and quaternary…
A: The protein undergoes several conformations to attain the mature functional protein.
Q: Where is hydrogen bonding observed in proteins? Between one part of the protein backbone and another…
A: Proteins are the biomolecules that are formed from amino acids joined by peptide bonds. There are…
Q: In a portion of polypeptide shown below, which bond is described by g rotation? a b NH,--- ---CO
A: A dihedral angle or torsional angle is the angle that exists between two intersecting planes. In…
Q: What type of tertiary structure interactions are most likely between the following amino acids? (Use…
A: Amino acids are building blocks of proteins; they include a carboxylic acid group on the alpha…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- Which of the following levels of protein structure may be affected by hydrogen bonding? (a) primary and secondary (b) primary and tertiary (c) secondary, tertiary, and quaternary (d) primary, secondary, and tertiary (e) primary, secondary, tertiary, and quaternaryWhat type of interaction would you expect between the following R groups in the tertiary structure of a protein? -CH2COO- and -CH2Ch2CH2Ch2NH3+ a. salt bridgesb. disulfide bondsc. hydrophobic interactionsd. hydrogen bondsLabel the N- & C termini Label ALL alpha -carbons (with an alpha) Label ALL peptide bonds(highlight or draw an X through these) Circle all AAresidues label which ones are nonpolar, polar,-acidic, & basic For each side chain, label/identifywhat types of R - group interactions it canparticipate in (H - bond, disulfide bond, ionic/electrostatic interactions, or hydrophobicinteractions.) Peptide Structure
- Which two types of bonding are in the 1ubq (Ubiquitin)? Explain your choices. a) Hydrogen bonding b) Ionic bonding c) Covalent bonding d) Disulfide bondingThis type of noncovalent bond is responsible for clustering fatty acid tails of phospholipids in a micelle. a. Disulfide bonds b. Hydrogen bonds c. Hydrophobic interactions d. Ionic interactions e. van der Waals interactionsPeptide bond formation occurs between which functional groups? SELECT ALL THAT APPLY A. Amino B. Carboxyl C. Alpha-Carbon D. Nitrogen
- What maintains the secondary structure of protein: A) peptide bond. B) Hydrogen bonds between the amino group of one peptide bond and the carboxyl group of another peptide bond. C) Hydrogen bonds between the R group of one amino acid and the carboxyl group og another peptide bond. D) disulfide bonds. E) Hydrophobic interactions.How do the following noncovalent interactions help to stabilize the tertiary and quaternary structure of a protein? Give an example of a pair of amino acids that could giverise to each interaction.(a) Hydrophobic interactions(b) Salt bridges (ionic interactions)A structure of tyrosine is given and I need to draw the structure for neutral solution, pH 2 and pH 12 3 drawing 1. neutral solution 2. pH 2 3. pH 12
- Which type of interaction stabilizes the alpha (α) helix and the beta (β) pleated sheet structures of proteins? Group of answer choices A. hydrophobic interactions B. peptide bonds C. hydrogen bonds D. nonpolar covalent bonds E. ionic bondsWhich of the following best represents the backbone arrangement of two peptide bonds? A) Ca—N—Ca—C—Ca—N—Ca—C B) Ca—N—C—C—N—Ca C) C—N—Ca—Ca—C—N D) Ca—C—N—Ca—C—N E) Ca—Ca—C—N—Ca—Ca—CWhich level(s) of protein structure result(s) from non-covalent interactions involving both backbone groups and side chains? (Select all that apply!) a) Primary b) Secondary c) Tertiary d) Quaternary

