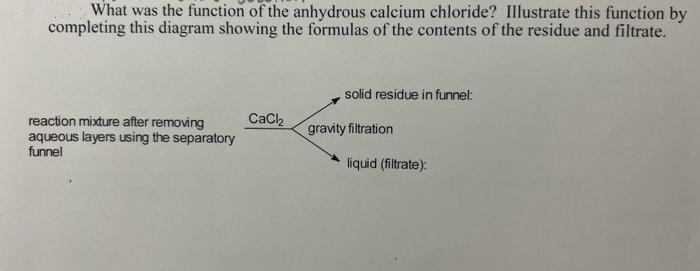
What was the function of the anhydrous calcium chloride? Illustrate this function by completing this diagram showing the formulas of the contents of the residue and filtrate. solid residue in funnel: CaCl reaction mixture after removing aqueous layers using the separatory funnel gravity filtration liquid (filtrate):
Q: Mg|Mg(NO3)2(aq)||AGNO3(aa) |Ag6)
A:
Q: 2. Write down the chemical reaction for the heating of the hydrated form of copper II sulfate. The t...
A: Mass of hydrated copper sulphate = 4.1g Mass of anhydrated copper sulphate = 2.6g Mass of water = ...
Q: Consider the reaction 2NO(g)+Br2(g)⇌2NOBr(g),Kp=28.4, at 298 KK In a reaction mixture at equilibri...
A: The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical eq...
Q: Find the equilibrium concentrations of A and B for a=1 and b=2 . Assume that the initial concentrati...
A:
Q: Q: What are the solutions, explain it?
A: The solution is the homogeneous mixture of two or more substances.
Q: Suppose that in the experiment the student failed to heat the crucible and hydate sample long enough...
A: Question : Suppose that in the experiment the student failed to heat the crucible and hydate sampl...
Q: 2. a. Identify and describe the type of bonding present in C,H. b. What is the electron configuratio...
A:
Q: You have the following polypeptides at pH 7.25 and are attempting to separate them using an anion ex...
A: For an amino acid residue, If pKa > pH, the ionizable group remains in its protonated state. ...
Q: What organic product would you obtain from reaction of 1-pentanol with CrO3, H2O, H2SO4?
A: Structure of 1-pentanol is CH3-CH2-CH2-CH2-CH2-OH which is a primary alcohol.
Q: Use the References to access important values if needed for this question. a. Use strain energy incr...
A: One 1,3-diaxialinteraction creates 0.9 kcal/mol of energy. And gives instability to the compound.
Q: *00 0.0 A 0.370-mol sample of a metal oxide (M2O3) weighs 55.4 g. How many moles of O are in the sam...
A:
Q: A beaker contains water up to the 130 mL mark. We introduce an object 38 g of dough and we observe t...
A:
Q: = is attached to The stereochemistry of that carbon is The sulfide group The stereochemistry if that...
A: We will give the priopity to the group according to CIP rule. Priority is given on the basis of at...
Q: 1. Provide two advantages and two disadvantage of flow cytometry analysis. Briefly explain each.
A:
Q: Suppose a weak acid has Ka = 4.0 x 10^-9. which is equivalent to Kb? and why??
A:
Q: a. How many protons does this element have? b. How many neutrons does this element have? c. How many...
A: The atomic number of an atom is the number of protons present in the atom. For a neutral atom, the n...
Q: What amount of 0.38 M solution can be made from a296mL 4.65 M solution? Answer should be in L Ro...
A: Given, The amount of 0.38 M solution can be made from a296mL 4.65 M solution is:
Q: Kr has 36 protons and 36 electrons. Give the symbol of two anions and two cations that are isoelectr...
A: Krypton(Kr) belongs to the noble gas family, i.e. group 18. It's atomic number is 36 so it has 36 pr...
Q: 38.The density of carbon dioxide is 1.8 kg/m^3. So, the volume occupied by 7.2 kg of carbon dioxide ...
A: The mass per unit volume of a substance is known as the density of the substance. The density of a s...
Q: Please help quick organic chemistry. and dont use PCC reagent AN OH
A: Given,
Q: 7) putting the least acidic first. Arrange the following compounds in order of increasing acidity, O...
A:
Q: 2. Provide an explanation (using resonance structures) for why the barrier for rotation about the ma...
A:
Q: At 1200 K, the following reaction has a Ko = 7.6 x 108. %3D H2 (g) + Cl2 (g) 2 HCI (g) What is Ko fo...
A:
Q: Drag and drop the product of the following fatty acid reaction. + CH;NH/H
A:
Q: how you wo accomplish the ing syntheSIS. Indicate all necessary reagents, and show any intermediates...
A: Organic reaction mechanisms:
Q: Explain in a more clinical detail the mode of actions of the following on the human skin cell or tis...
A:
Q: Hi! I need this question really quickly! Thank you!
A: Mass of magnesium chloride = 1.3443 g Volume of HCl = 54.90 mL Concentration of HCl = 0.656 M
Q: What is the mass of a 98 Liter sample of water vapor (H,0)?
A: Given :- volume of water vapor = 98 L To calculate :- mass of water vapor in sample
Q: HO, H CH;0,
A: Chiral carbon: Carbon with 4 different atoms or groups Chiral nitrogen: Nitrogen with 3 different at...
Q: Choose which waste disposal container you should dispose of the unused 0.1 M FeClz solution. (A) org...
A:
Q: 3.) Express in ppt.ppm, and ppb fra fion the conce n of 250 g salt solution with 0.0045 9 salt disso...
A: Given, Mass of salt dissolved = 0.0045 g Mass of salt solution = 250 g Required, ...
Q: Provide the correct IUPAC name for S2Cl8
A:
Q: (van’t hoff) use van’t hoff to calculate freezing and boiling points; don’t know what went wrong
A: Vant Hoff factor,i = 1.83 Molality of solution = 7.75 × 10-2 M Kf = 1.86 °Ckg/mol Kb = 0.512 °Ckg/m...
Q: Draw all possible stereoisomers of the structures below. Indicate the isomeric relationships between...
A: The molecules with the same molecular formula but different structures are known as isomers. If the ...
Q: How many grams of iron are needed to produce 3 g of iron(III) chloride? Balance the equation: ____ ...
A: Introduction: From the given equation, Fe + Cl2 ---> FeCl3...
Q: Indicate the number of: protons neutrons and electrons for 204pb nuclide.
A: Here we have to find the number of protons, neutrons and elctrons present in 204Pb nuclide isotope. ...
Q: Solve both parts otherwise I will downvote
A: We are given two reaction and in both we have to write the mechanism of reduction of alcohol to alde...
Q: Hydrogen and iodine react to give hydrogen monoiodide as follows: H2 (g) + I2 (9) –→ 2HI(g) At 300 K...
A: Equilibrium constant Kc depends on concentration of reactant and product which are in gaseous state....
Q: 00 %24 3. Balance the following equation: Ca 3(PO 4) 2( S) + SiO 2( s) + C( s) → CaSiO 3( s) + CO( g...
A: In a balanced chemical equation, All the atoms will be the same in both the side.
Q: For an unknown acid lab, titrating with 0.1 M NaOH, what is an acid that is slightly brown solid and...
A: The acids that are solids are mostly organic acids (other than formic, acetic , propanoic and butano...
Q: 6. How would you synthesize the enone below from a crossed Aldol reaction?
A: We have to synthesis the given enone by cross aldol reaction. Cross aldol reaction involve of two di...
Q: For the reaction N2(g)+3H2(g)⇌2NH3(g) Kp = 2.60×10−3 at 290. ∘C . What is Kc for the reaction at t...
A:
Q: The indigo dye used to color blue jeans has the formula C6H1ON2O2. How many molecules of indigo are ...
A: Given :- amount of C6H10N2O2 = 50.0 g To calculate :- total number of C6H10N2O2 molecules
Q: The gas constant R has the value 287 J/Kg * K for dry air. Using the ideal gas law (P = PdryRT): a.)...
A:
Q: Give the condensed structure and IUPAC Name of the following
A: Given structures are : Give the condensed structure and IUPAC Name of the following = ?
Q: Select 'True' or 'False' for each of the following items: When salt is sprinkled on an icy road, t...
A:
Q: How many moles of glucose are in 4.76 L of a 3.4 M glucose solution?
A: Volume of solution = 4.76 L Molarity of solution = 3.4 M Molarity = (Moles) / (Volume of solution in...
Q: The change in internal energy of the combustion of solid naphthalene (C10H8) was determined to be eq...
A:
Q: What volume of hydrogen gas at STP is produced from the reaction of 46.51 g of Mg? Balance the equa...
A: Here we have to determine the volume of hydrogen gas produced at STP by the reaction of 46. 51 gram ...
Q: time (s) [Ser] at T=335 0.150 0.143 20 0.120 40 0.090 90 0.015 75 0.038 What is the time necessary f...
A: It is a zero order reaction. Due to change in concentration is directly proportional to time.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Using a spectrophotometric technique, the phosphorus content in a mineral will be prolonged. The procedure used is described below: 8.35 g of sample are dissolved and treated so that all phosphorus is converted to the blue orthophosphate complex and made up to volume with distilled water end of 100 ml. The transmission of the resulting solution is 30.6%. If the orthophosphate standard solution of sodium, which is treated in the same way, and which has a final concentration of 2.467x10-4 M, gave a transmittance of 20.5%, calculate the phosphorus content in the driven sample in ppm Na3PO4.Calculate the percent error in Avogadro's number calculated by using the following experimental data. Pipette calibrated as 30 drops/mlDiameter of the watch glas:15.5 cmNumber of drops to complete monolayer:23Concentration of stearic acid solution: 1.2x 10^-4g/ml.Which of the following could DECREASE the amount of the recovered crystals in the purification by recrystallization experiment? Collecting the recrystallized benzoic acid by filtration using a pre-weighed filter paper. Using a long-stemmed funnel in the hot filtration step. Cooling the filtered solution rapidly. Washing the recovered crystals using hot solvent. a. II, III b. II, IV c. II, III, IV d. I, II, III, IV
- How was the precipitate obtained after filtration? a. By drying the precipitate on the filter paper using a desiccator. b. By charring the filter paper and igniting the product in a crucible. c. By redissolving in hydrochloric acidA sample of fried potatoes weighing 200 g was extracted using a volatile organicsolvent. The recovered cooking oil weighed 15 g. What methods from thisexperiment could be used to separate the cooking oil after the extraction? (evaporation, gravity filtration or vacuum filtration?) why so?Calculate the mass percent of the ascorbic acid in the same show full solution Situation: The pounded Vitamin C tablet was half into two below are the calculated data in the experiment. Each of the half are transfered in a 250ml erlen meyer flask and added 75ml of deionized water for the vitamin C to dissolve. Then it was prepared for titration where the burette was filled with 0.09794M standardize NaOH solution then a total of with 3 drops 2% phenolphthalein indicator was added to each erlen meyer flask l, swrill it and begin the titration. below are the following data gathered: Vitamins used: Nature Made vitamin C 500mg tablet Trial 1 Mass: 0.3108g of Vitamin C tablet Initial volume: 1.6mL Final volume: 15.4mL Trial 2 Mass: 0.3349g Initial volume: 15.4 mL Final volume: 30.2 mL
- A 5.000-g soil sample was analyzed for potassium content by extracting the potassium using 10.00 mL aqueous ammonium acetate solution. Following the extraction, the soil was filtered and rinsed. The filtrate with rinsings was diluted to exactly 50.00 mL. Then, 1.00 mL of this solution was diluted to 25.00 mL, and this dilution was tested with an instrument. The concentration in this 25.00 mL was found to be3.18 ppm. What is the concentration of the potassium in the soil in ppm?Correlate the structures of the standard food dyes to their relative position in the paper chromatogram. Refer to the structures shown below. What are the identities of the unknowns in paper chromatography of food dyes? How did you arrive at this conclusion?3. Assume that in extraction from water into toulene, analyte A has distribution ratio of 10. A 20 mL portion of an aqueous solution of A is extracted with toulene. Which of thw following procedures will result in most efficient removal of A from the aqueous phase into toulene? (Please show the solution of the correct answer) A. 1 extraction with 20 mL of touleneB. 1 extraction with 40 mL of touleneC. 2 extractions with 20 mL of toulene eachD. 4 extractions with 10 mL of toulene each
- A sample of an iron ore was prepared for Fe3+ analysis as following: 3.4g of the sample was added anddissolved in acid environment then diluted to 250 mL using volumetric flask. After that, 10 mL of the resultingsolution was transferred by pipet to a 50-mL volumetric flask and continue to be diluted. The scientists foundout that this solution gives the concentration of Fe3+ as 2.3 mg/L. Find the weight percentage of Fe3+ in theoriginal sample.5.00 mL of stock solution is diluted to 25.00 mL, producing solution ALPHA. 10.00 mL of solution ALPHA is diluted to 25.00 mL, resulting in solution BETA. 10.00 mL of solution BETA is then diluted to 25.00 mL, producing solution GAMMA. dilution factor for ALPHA from stock solution = 0.167 dilution factor for BETA from ALPHA solution = 0.0476 part c and d?Solution Calculations Using unit analysis, clearly show the set-ups for your calculations. You wish to prepare 50.0 mL of 2.30 x 10–2 M solution of Levothyroxine (hint-how many grams will you need of Levothyroxine?) 1. Describe how to prepare this solution from the pure drug and deionized water. Analytical balances and volumetric flasks are available to you. (Assume that Levothyroxine dissolves in water, at least to the extent necessary for this problem.) Use complete sentences in your description of the solution preparation process. a. A patient is given 2.0mL of a 3.00% (m/v) solution of Levothyroxine, how many milligrams of the drug did they receive? b. What is the molarity of a 3.00% (m/v) solution of Levothyroxine?

