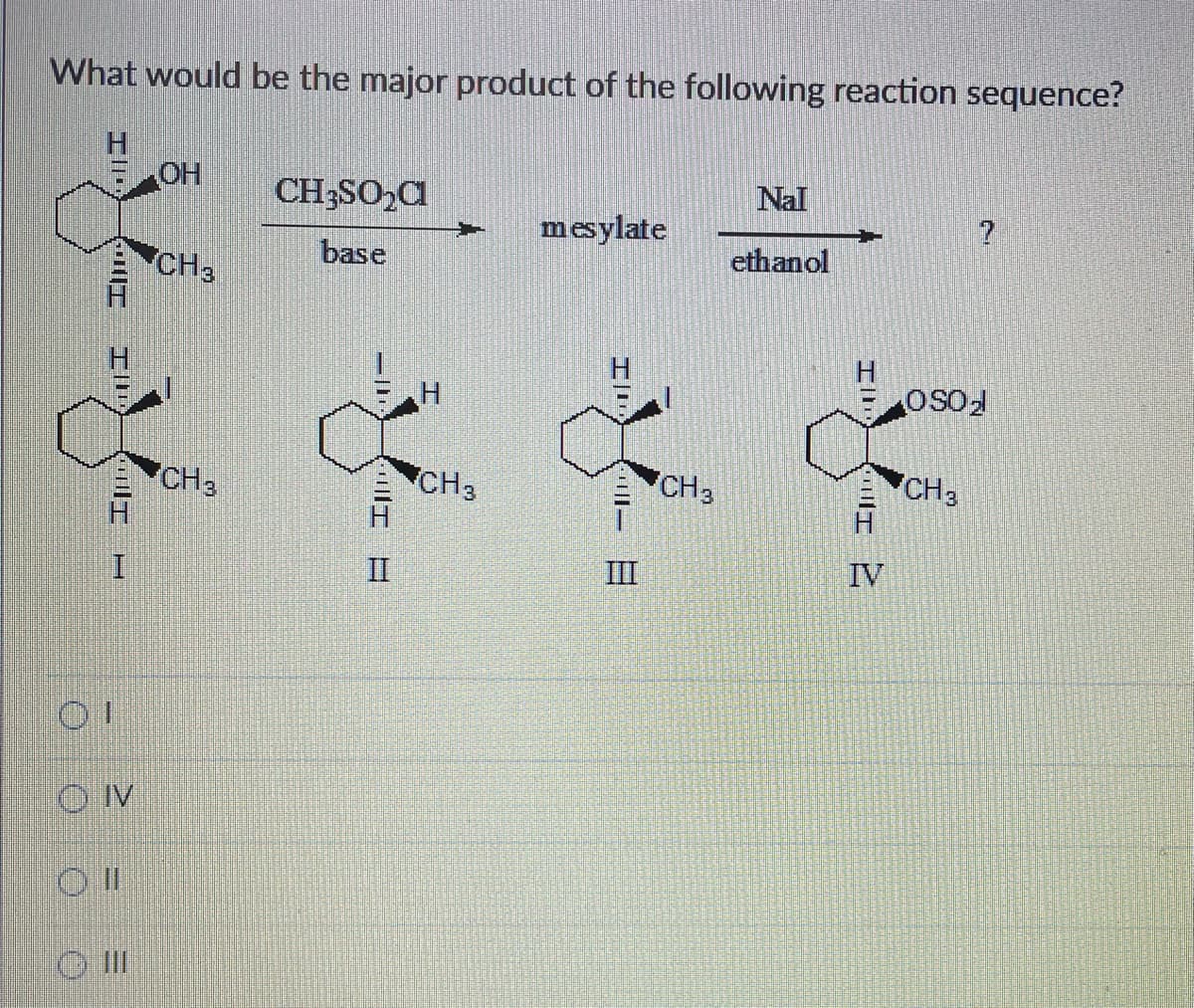
What would be the major product of the following reaction sequence? O( DIT III HT P OH CH3 CH3 CH3SO₂C base E Ill CH3 mesylate IL - III CH3 Nal ethanol 7 I 2 ? OSO₂ CH3
Q: How to use C NMR and H NMR? And what are some other important information should I know when…
A: CNMR i.e. 13C NMR is used to detect number of non-equivalent carbon atom present in the given…
Q: Consider the molecule of furan. Its Lewis structure is shown. H रु. How many sigma (0) and how many…
A: -> Sigma bond and pi bonds are type of covalent bond that differ in the overlapping of atomic…
Q: A patient is prescribed 5mg/kg of a medication. The patient weighs 8.1Kg. The medication is supplied…
A: According to the question we have, The dosage for the patient is given by = 5mg/kg The weight of the…
Q: Predict the ideal bond angles around each central atom in this molecule. H H oxygen: carbon: .. -C
A: we have to determine the ideal bond angle of carbon and oxygen in the given structure
Q: What is the molar mass of an unknown gas with a density of 10.1 g/L at 4.00 atm and 35.0 °C?
A:
Q: Maleic acid (H₂C₂H₂O) is a diprotic acid with Kal = 1.20 × 10-³ and Ka2 maleic acid (H₂C₂H₂O4)…
A:
Q: Consider the molecule of cyanogen. Its Lewis structure is shown. : N EC- Which carbon orbitals are…
A:
Q: Data Table: Analysis: 1. 2. 3. 4. 5. 6. 7. 8. 12. 13. 9. 10. ches of Hg. Convert this measurement to…
A:
Q: 0.144 mol of a weak acid, HA, is placed in enough water to make 1.00 L of solution. The final pH of…
A:
Q: Consider the molecule of Cyanic acid, HOCN. Its Lewis structure is: What is the electron-domain…
A:
Q: Dr. Evil prescribed Austin 2.0 mL of 1:40 evil truth serum. However, Scotty ordered 1:100 evil truth…
A:
Q: CHg+O, → CO, +H,O If 80 grams of ethane (C₂Hg) is burned in excess oxygen gas (O₂), calculate the…
A:
Q: Use the orbital approximation to estimate the energy of an Li atom. How does this value compare to…
A: The atomic number of Li atom is 3. According to Aufbau Principle, the electrons are filled in an…
Q: Based on the experimental results shown in the table, Identify whether the inhibitor acting on this…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 2NH₂(g) → N₂(g) + 3H₂(g) ΔΗ = 92. kJ AS = J K AG 25. kJ Which is spontaneous? this reaction the…
A: Given : For first reaction : ∆H = 92. kJ ∆G = 25. kJ Temperature = 70 °C = 343 K For second…
Q: Me Me Me Me Me 1. H-B 2. NaOH, H₂O₂
A:
Q: Determine the mass of KOH (molar mass 56.108 g/mol) required provide enough KOH to combine with…
A:
Q: In the neutralization reaction below17.3 mL of H2SO4 solution are required to neutralize 32.2 mL of…
A:
Q: The data below was collected using the procedure described in your lab notebook: Mass (before…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: QUESTION 2 If I write 3 Na2CO3, how many atoms of sodium do I have? 3 02 O None of these 6
A:
Q: What is the molar mass of an unknown gas with a density of 1.90 g/L at 1.00 atm and 25.0 °C?
A:
Q: When copper metal is added to nitric acid, the following reaction takes place Cu (s) + 4 HNO3 (aq) →…
A: For the first question, we have to find the volume of NO2 gas that will be formed when 3.09 g of…
Q: The 1HNMR spectra and IR spectra for anisaldehyde are shown below. Complete the spectral data:…
A: The structure of Anisaldehyde is represented as follows:
Q: Consider the NCO anion. the following three Lewis structures can be drawn for it: Structure 1…
A:
Q: Data Table Use the following table to record your data for each trial. Atmospheric pressure Mass of…
A: Given, Reaction of magnesium metal with HCI .
Q: When the following skeletal equation is balanced under acidic conditions, what are the coefficients…
A:
Q: An internal standard is incorporated into an analysis method when systematic sources of variation…
A: High-performance liquid chromatography, formerly known as high-pressure liquid chromatography, is a…
Q: 1. How many sigma and pi bonds are in aspartame → sigma pi
A: In a compound first formed bond is Sigma bond and if second bond is formed then the formed bond is…
Q: 1.65 g of an unknown gas at STP fills a 500. mL flask. What is the molar mass of the gas?
A: At STP, Volume of one Mole of gas is 22400 ml.
Q: What is the structure based on the HNMR?
A: Nuclear Magnetic Resonance (NMR) spectroscopy is an analytical chemistry technique used to deduce…
Q: Draw by hand a Newman projection formula for the stable chair conformation of terpin hydrate. View…
A:
Q: II. Give an acceptable name for each of the following structures. 1. 2. H₂C-CH-C=C-CH-CH₂-CH3 CH3…
A:
Q: Which of the following is FALSE with respect to ketone bodies? The production of ketone bodies is…
A: Ketone bodies refer to molecules with a ketone group that are also soluble in water. The production…
Q: -5 A 5.23 X 10 mole protein is dissolved in 31.2 mL of water at 28°C. What is the osmotic pressure…
A:
Q: What is the product that results from the following synthetic sequence? xs Br₂ H₂O O A OB O OD (A)…
A:
Q: A rock contains 0.688 mg of 206 Pbfor every 1.000 mg of 238 Upresent. Assuming that no lead was…
A: The question follows first order kinetics and the age of the rock is calculated using number of…
Q: e) BCl3 Polar bonds? [Select] Polar molecule? [Select] Intermolecular Forces? [Select] H Polar…
A:
Q: What is the skeleton equations from word equation? Zinc metal reacts with iron (II) oxide to…
A:
Q: Calculate the pH of a titration at the point when 15.0 mL of 0.15 M NaOH is added to 30.0 mL of 0.20…
A: Given -> Volume of NaOH = 15.0 ml Molarity of NaOH= 0.15 M Volume of HNO3 = 30.0 ml Molarity of…
Q: Using the answers to the questions attached answer the question: Why are compounds composed of…
A: From Dalton's law of multiple proportions in atomic structure, when two elements react to form more…
Q: he charges and sizes of the ions in an ionic compound affect the strength of the electrostatic…
A: Lattice energy mainly depends upon the size and charge of its constituent ions. so to compare the…
Q: 1. A chemical reaction produces 210 J/g. How much energy is this in units of cal/lb? 1 pound = 16…
A: According to the question we have The energy of the reaction is given by = 210 J/g we have to…
Q: Arsenic acid (HAs04) is a triprotic acid with Ka1 = 5.5x10-3, Ka2 = 1.7×10-7, and Ka3 = 5.1x10-12.…
A:
Q: 4. Draw Lewis structures for the following (include formal charges): (a) BH4; (b) OPCI3; (c)…
A: Note-Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: e) BCI3 Polar bonds? [Select] Polar molecule? [Select] Intermolecular Forces? [Select] X Polar…
A: According to the answering guidelines, I'm posting the solution for the first 3 subparts. Kindly…
Q: Given the reaction: CH4 + 2 02 yields CO2 + 2 H₂O How many oxygen atoms go INTO the reaction? O 16…
A: Two oxygen atoms combine to form a oxygen molecule (O2).
Q: It is also known that like dissolves like". Polar molecules tend to hang out with polar molecules,…
A: Structure of the dyes: Blue No.1 dye…
Q: What do they mean by accepted molar mass of butane in question #7? I've calculated the molar mass of…
A: Introduction: The accepted molar mass is nothing but the actual molar mass calculated from the…
Q: O There is no subshell fitting this description 3p O 3s O 3d
A: n = Represent principle quantum number l = Azimuthal quantum number
Q: 3. The solubility product constant for calcium fluoride, CaF2, is 3.40 x 10-11. a. Find the molar…
A:
Step by step
Solved in 2 steps with 1 images

- Propose a mechanism to show how 3,3-dimethylbut-1-ene reacts with dilute aqueousH2SO4 to give 2,3-dimethylbutan-2-ol and a small amount of 2,3-dimethylbut-2-ene.The trimethylsilyl (TMS) protecting group is one of several silicon protecting groups for alcohols. For each reaction, draw the mechanism for the protection of (R)-3-bromo-1-butanol with the following silyl chlorides, using triethylamine as the base: (a) terf-butyldimethylsilyl chloride (TBS-C1] (b) triisopropylsilyl chloride (TEPS-C1) (c) triethylsilyl chloride (TES-C1)Show the products from reaction of p-bromoaniline with the following reagents: (a) CH3I (excess) (b) HCl (c) HNO2, H2SO4 (d) CH3COCl (e) CH3MgBr (f) CH3CH2Cl, AlCl3 (g) Product of (c) with CuCl, HCl (h) Product of (d) with CH3CH2Cl, AlCl3
- The following reactivity order has been found for the saponification of alkyl acetates by aqueous NaOH. Explain. CH3CO2CH3 > CH3CO2CH2CH3 > CH3CO2CH(CH3)2 > CH3CO2C(CH3)3Please describe the difference in the outcome of treating a terminal alkyne with HgSO4, H2SO4, H2O and with 9-BBN, followed by H2O2 and NaOH.Which of the following will produce enantiomeric pair on treatment with HOH? I II III IV
- What steps are needed to prepare phenylacetylene, C6H5C = CH, from each compound: (a) C6H5CH2CHBr2; (b) C6H5CHBrCH3; (c) C6H5CH2CH2OH?The base-promoted rearrangement of an a-haloketone to a carboxylic acid, known as the Favorskii rearrangement, is illustrated by the conversion of 2-chlorocyclohexanone to cyclopentanecarboxylic acid. Jo Han Sow La NaOH, THF O Nat H3O+ OH NaOH, THF The mechanism involves the following 5 steps: -ō cyclopropanone intermediate 1. Abstraction of a proton to form enolate anion 1; 2. Formation of a cyclopropanone intermediate 2 with expulsion of chloride ion; 3. Addition of hydroxide ion to form tetrahedral intermediate 3; 4. Collapse of the tetrahedral intermediate and breakage of the three-membered ring to form carbanion intermediate 4; 5. Proton transfer to form the rearranged carboxylic acid. For the following reaction, draw the reaction out on paper, and then draw the structure of cyclopropanone intermediate 2 in the window. 1. NaOH, THF 2. H3O* OHWhat is the major product of the following reaction sequence?Tek seçenek.
- select the most appropriate reagent(s) to effect the change. K2Cr2O7, H+ H2, Pd 1. Disiamylborane, 2. HO–, H2O, H2O2 NaOCl H2SO4, HgSO4Can you draw out the following reaction mechanism: 1. Benzyl alcohol + H2SO4 → Benzyl oxonium ion + HSO4- 2. Benzyl oxonium ion + CrO3 → Chromate ester 3. Chromate ester → Benzylic carbocation 4. Benzylic carbocation + H2O → Benzaldehyde + H3O+ 5. Benzyl alcohol + H2CrO4 → Benzaldehyde + Cr3+ + 3H2SO4 Byproduct: CrO3 + 3 H2O --> Cr3+ + 3 H2SO4Predict the major products of the following reactions.(a) (R)-2-hexyl tosylate + NaCN(b) the tosylate of cyclohexylmethanol + excess NH3

