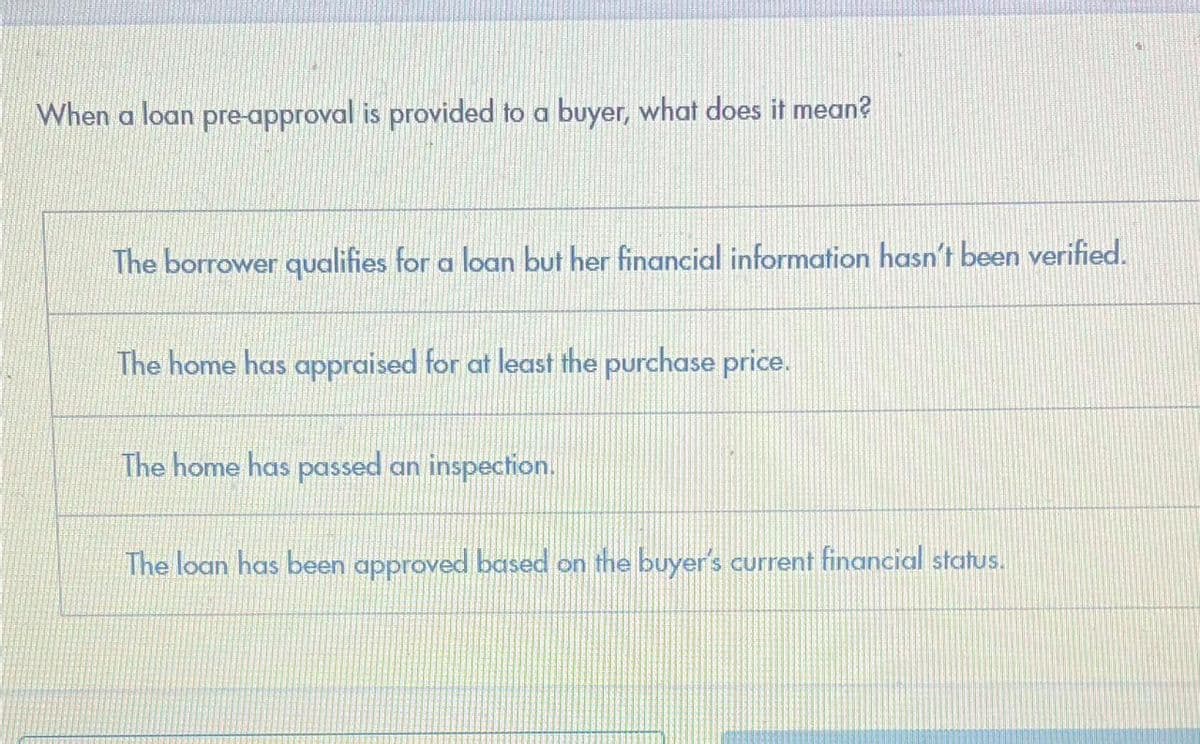
When a loan pre-approval is provided to a buyer, what does it mean? The borrower qualifies for a loan but her financial information hasn't been verified. The home has appraised for at least the purchase price. The home has passed an inspection. The loan has been approved based on the buyer's current financial status.
Q: Retrosynthesis, include reagents and intermediate, thank you.
A: Given is retrosynthesis reaction. The given starting compound is diethyl malonate. So, we can…
Q: Draw products and the mechanisms for each of the following:
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: Please provide explanations
A: Answer shown in attachment provide in explanation box Explanation:
Q: Sodium phosphate is added to a solution that contains 0.0051 M aluminum nitrate and 0.014 M calcium…
A: Solubility product: It is defined as the product of its dissolved ion concentrations raised to the…
Q: Using the priority rule, identify the configuration at each chiral center and the configuration of…
A: Note: Since you have posted a question with multiple sub parts, we will provide the solution only…
Q: There was a titration between 0.15 M NaOH with a monobasic acid HCI (10.0 mL). Phenolphthalein was…
A: Volume of NaOH(V1) = 18.5 mLMolarity of NaOH(M1) = 0.15 MVolume of HCl(V2) = 10.0 mL
Q: What is the IUPAC name of the following compound? CI
A: Given molecule:We have to write the IUPAC name of the given moleculeGiven Answer Options:…
Q: While ethanol (CH3CH2OH) is produced naturally by fermentation, e.g. in beer- and wine-making,…
A: Given that,volume of flask = 5.0 LPressure of ethylene gas,PC2H4= 1.0 atmPressure of water vapour,…
Q: What volume (in mL) of 0.586 M Ba(OH)2Ba(OH)2 (aq) must be added to a 5.00 mL sample of vinegar to…
A: The relationship between the molarity and volume of acid and base during a neutralization reaction…
Q: Ag(s) Pb(s) salt bridge True True Determine which of the following statements about the cell shown…
A: The galvanic cell is cell which drives electrical energy from spontaneous redox reaction happening…
Q: Please help with E, F, G:
A: E) For the reaction starting with the alkyne, the reagents used in step a) and step b) and the…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Incorrect, 2 attempts…
A: The objective of the question is to predict the product formed in the following reaction given.
Q: Provide mechanism with arrows and charges H3CO NaOH H₂O مسم
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: A chemical reaction is carried out in a se of win J. 0-464J ○ 458J ○ 230 J 0-230 J
A: Initial volume= 1.78 L Final volume= 3.19 L Pressure= 3.25 atm
Q: Name the structure using IUPAC
A:
Q: Functional Group Type of Compound Name Hydroxyl group Alcohol Ethan___________…
A: The objective of the question is to identify the names of the compounds based on their functional…
Q: Payalben
A: The objective of the question is to find the equilibrium concentration of B in the given reaction.…
Q: Please provide explanations
A: answer attached below Explanation:Step 1: Step 2: Step 3: Step 4:
Q: Draw the major product o
A: Given is organic reaction. The name of given reaction is hydrobromination reaction. The given…
Q: Show how you would accomplish the following synthetic conversions. (a) 1-butene 1-bromobutane (b)…
A: The reactant can be converted into products by selecting the suitable reagents and reaction…
Q: 2. Determine absolute configuration (R or S) for the following compounds. Cl OH R S Br HO OH R S…
A: Absolute configuration is the spatial arrangement of atoms in a molecule.The absolute configuration…
Q: 3. a. E,R is? b. E,S c. S,Z 4. What is the π-energy diagram of the compound in question 3? a. b. d.…
A: The answer is 3- b4-cExplanation:Approach to solving the question: Detailed explanation: Examples:…
Q: What quantity of heat (in kJ) will be absorbed by a 82.3 g piece of aluminum (specific heat = 0.930…
A: The objective of this question is to calculate the amount of heat absorbed by a piece of aluminum as…
Q: Type in your numeric answer showing two decimal places to identify the pH of a solution with the…
A: Given that,The concentration of hydronium ion,(Assume unit in M)pH of the solution= ?
Q: An acetal can be hydrolyzed by treatment with aqueous acid. For the reaction below: H3CO OCH 3 H*…
A: Acetals are those class of organic compounds where two oxygen atoms are bonded to a single carbon…
Q: If 20.0 g of LiOH is added to 0.750 L of 1.00 M Cd(NO₃)₂, how many grams of precipitate will be…
A: The balanced equation for this precipitation reaction is: Cd(NO3)2 + 2LiOH → Cd(OH)2 + 2LiNO3 The…
Q: What are the products of an aldol cyclization for the following compound?
A: We have to predict the cyclization products.
Q: Acetic acid reacts with NaOH. At equilibrium the [CH3COOH] = 0.539 and the [CH3COO-] = 0.207 Ka =…
A: The objective of this question is to calculate the pH of a solution of acetic acid (CH3COOH) and…
Q: What volume (in mL) of 0.586 M Ba(OH)2Ba(OH)2 (aq) must be added to a 5.00 mL sample of vinegar to…
A: Given data:At equivalence point all the amount of acid gets neutralized by a specific amount of base…
Q: Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid…
A:
Q: A solution of HBr with a volume of 25.00 mL is titrated to the endpoint, with 0.550 M Ba(OH)2. If it…
A: 2HBr(aq) + Ba(OH)2(aq) --> 2H2O(l) + BaCl2(aq)For HBr Solution,Concentration of HBr solution =…
Q: Consider the following chemical equilibrium for the weak acid HF in water: 1 HF(g) + H2O(l) F(aq) +…
A: According to Le Chatelier's principle states that when a system present at equilibrium is subjected…
Q: Give an acceptable IUPAC name for each of the compounds
A: Basic rules for IUPAC nomenclature:1) Select longest chain or block of carbon atoms as parent. 2)…
Q: Using the priority rule, identify the configuration at each chiral center and the configuration of…
A: compound 1 the red centre is (s) -configuration. And the green double bond is with (E) In 2nd…
Q: Determine the mass in grams of 8.14 × 10²¹ atoms of copper. (The mass of one mole of copper is 63.55…
A: The objective of the question is to find the mass in grams of a given number of copper atoms. We…
Q: 3. Draw the structure of 3,4-diiodohexane that will be superimposable with its mirror image
A: Molecules can have different spatial arrangements of atoms even if they have the same chemical…
Q: A student used 500mg of tabletop sweetener to make a total of 5ml of solution with biuret reagent.…
A: The student prepared a solution containing 500 mg of tabletop sweetener in a total volume of 5 mL.…
Q: When the following bridged bicyclic alkene undergoes hydroboration-oxidation, how many products…
A: Hydroboration is the addition of borane or diboranes to the olefins to form the alkyl borane. The…
Q: 7. Reaction Scheme.
A: Given is organic reaction scheme. The given starting compound is ketone.
Q: of the following pairs, indicate which solution has the higher concentration of Iion. me words in…
A: The molarity of a solution is the number of moles of solute dissolved per 1 L of the solution.
Q: H3CO OH 2 HCONH,
A: The reaction of ketone with formamide results in the formation of an imine. An aldehyde and ammonium…
Q: You have 50.0mL of 0.116M HCl. You add 10.0mL of 0.25 M NaOH What is the pH of the solution now?
A: The objective of this question is to calculate the pH of a solution after the addition of a base…
Q: provide mechanism
A: We have to predict the mechanism.
Q: A solution of HBr with a volume of 25.00 mL is titrated to the endpoint, with 0.550 M Ba(OH)2. If it…
A: 2HBr(aq) + Ba(OH)2(aq) --> 2H2O(l) + BaCl2(aq)For HBr Solution,Concentration of HBr solution =…
Q: What is the expected final product for the shown reaction? Assume the amount of CH3OH used is in…
A: We have to choose the correct option.
Q: 66.667 ml of 3.000 M H2SO4 (aq) solution was neutralized by the stoichiometric amount of 4.000 M…
A: The objective of the question is to calculate the enthalpy change (ΔH) of the reaction in J/mol…
Q: A solution of HCl with a volume of 25.00 mL is titrated to the endpoint, with 0.250 M NaOH. If it…
A: Given:HCl (aq) + NaOH (aq) H2O (l) + NaCl (aq)Concentration (M1) of NaOH = 0.250 MVolume (V1) of…
Q: Draw all of the steroisomers for 2, 3 dibromo 1-butanol
A: Stereisomers are compounds with the same molecular formula and structural arrangements but differ in…
Q: 2. Complete the reaction mechanisms started above by drawing the curved arrow for the second step of…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 37. Complete the following reaction: CH₂CH H + H₂O/H*
A: The objective of the question is to predict the product formed in the following reaction given.
Unlock instant AI solutions
Tap the button
to generate a solution
Click the button to generate
a solution
- In each case, describe the change as a chemical or physical change. Give a reason for your choice. Salt dissolves when you add it to water. Food is digested and metabolized in your body. Crystalline sugar is ground into a fine powder. When potassium is added to water there is a purplishpink flame and the water becomes basic (alkaline).how many eggs are needed to make 1 dozen waffles. assuming you have enough of all the others given: 2 cups flour + 3 eggs + 1 tbs oil -> 4 wafflesPart F In volcano vents, sulfur is converted directly from the emitted hot gases to form mounds of solid sulfur. This is an example of Deposition. Boiling. Sublimation. freezing.
- Choose the one correct or one incorrect statement regarding the technique of weighing by difference: the weighing beaker should be moved with a strip of paper used as a handle or strap preparing 4 samples requires only 4 weighings the balance should be tared after each sample is removed from the weighing beaker the weighing beaker must be clean but does not need to be dry the receiving flasks must be both clean and dry before weighing beginsYou can calculate cost effectiveness using the following formula: volume of HCI neutralized per dose /cost per dose (formula) If you calculated that your volume of HCl neutralized per dose was 10.22 mL, calculate the cost effectiveness for Brand X. You can directly use the cost per dose you calculated in the previous problem. Do not include a unit in your answer. Report your answer to two decimal places. Brand X antacid: 2 tablets / 500 mg dose 40 tablets / container Cost: $3.75 / containerGiven the following values, determine the percentage of copper recovered. Show all work. Mass of weigh boat 0.214 g Mass of weigh boat + copper wire 0.501 g Mass of copper wire Mass of weigh boat (different from above) 0.204 g Mass of weigh boat + copper powder 0.418 g Mass of copper powder Percent of copper recovered
- Balance each chemical equation Na2S (aq) + Zn(NO3)2 (aq) ----> NaNO3 (aq) + ZnS(s)You can calculate cost effectiveness using the following formula: volume of HCI neutralized per dose / cost per dose If you calculated that your volume of HCl neutralized per dose was 23.73 mL, calculate the cost effectiveness for TUMS. You can directly use the cost per dose you calculated in the previous problem. Do not include a unit in your answer. Report your answer to two decimal places. TUMS antacid: 2 tablets / 500 mg dose 50 tablets / container Cost: $4.98 / container1. Iron metal rusts in an atmosphere of moist air. a. Physical Propertyb. Chemical Property 2. Mercury metal is a liquid at room temperature.a. Physical Propertyb. Chemical Property3. Silver salts discolor the skin by reacting with skin protein.a. Physical Propertyb. Chemical Property4. Nickel metal dissolves in acid to produce a light green solutiona. Physical Propertyb. Chemical Property 5. Potassium metal has a melting point of 63°C.a. Physical Propertyb. Chemical Property6. Titanium metal can be drawn into thin wires.a. Physical Propertyb. Chemical Property7. Silver metal shows no sign of reaction when placed in hydrochloric acid.a. Physical Propertyb. Chemical Property8. Copper metal possesses a reddish-brown color.a. Physical Propertyb. Chemical Property9. Beryllium metal, when inhaled in a finely divided form, can produce serious lung disease.a. Physical Propertyb. Chemical Property
- Use the algebraic equation to determine the mass if the volume was 622 mL. mass (g) = (3.60 g/mL)(volume (mL)) + 11.4 g y= mass (g) m = 3.60 g/mLx= 622 mL b = 11.4 gTo keep homamade ice cream frozen, it is placed in a bucket of ice-water and salt mixture. A mixture is commonly composed of 1.70 kg of salt (NaCl) and 5.25 kg of ice.Density of an aqueous solution of nitric acid is 1.43 g/mL. If this solutioin contained 36.0% nitric acid by mass, what volume of solution would be needed to supply 1.50 mmol of nitric acid?

