Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question
100%
Transcribed Image Text:Zoom
Bb Genesis for Pare x
e Parents at Fran x
* No Fear Shakes x
O Playing a Game x
A
Schittone Chem x
Launch Meeting X
quizizz.com/join/game/U2FSDGVKX18%252FxiAfP9ituPBRQWmX%252BRVJDQHSDEZLCK%252FZ%252FLEC5EAWCLLP32XSY9).
Bb my work and answ.
M HBO Max
A Achieve3000: Level.
E Laisha Brito -Writin..
8 9th
E 2210
9/10
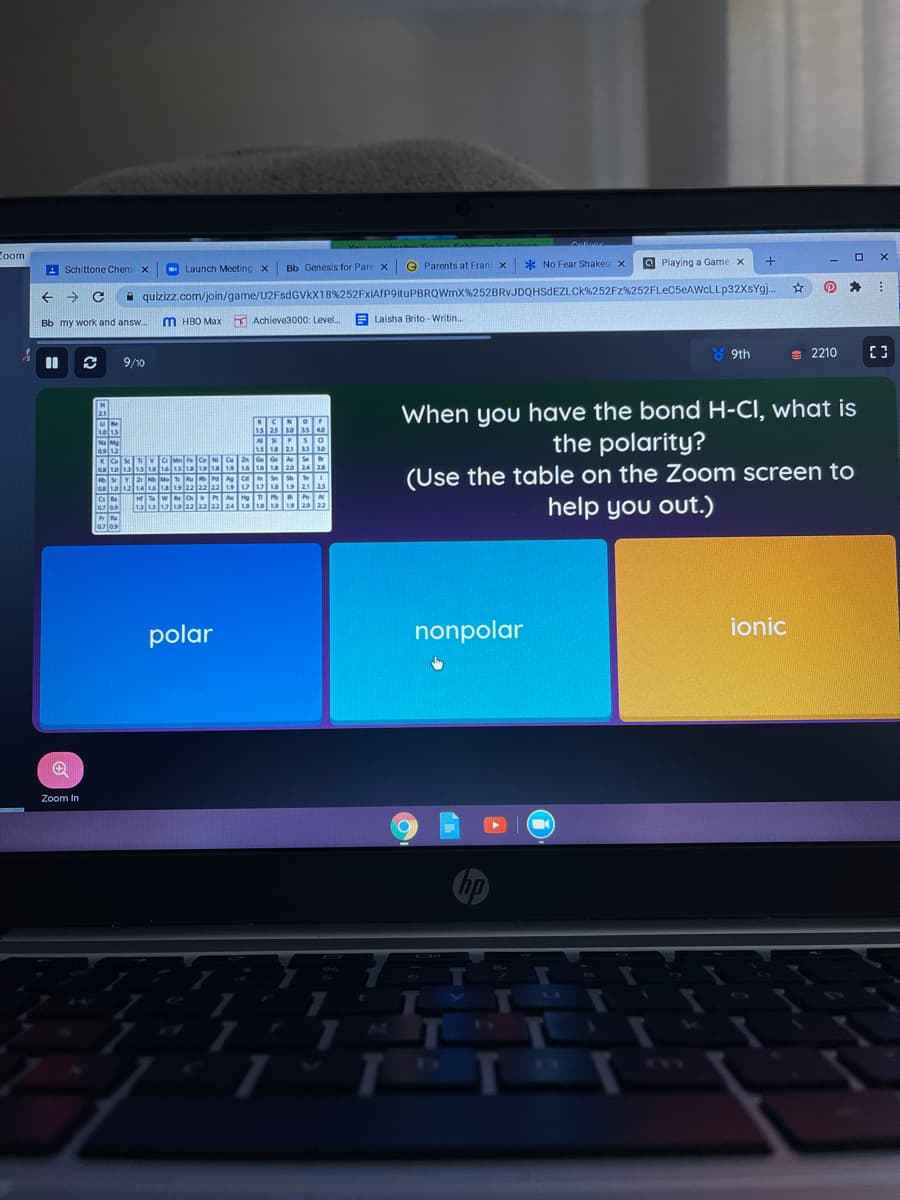
When you have the bond H-CI, what is
the polarity?
(Use the table on the Zoom screen to
help you out.)
2.1
1s 25 10as40
les 12
KCa
NSP Sa
15 21 as s0
ZnG Ge A Se
SY2Nh Mo u A Ce n s el
as 01214 a 22 22 22 1 uua1s 21 25
HT w o RA Hn A
1315 139 22 aa a2 2A 19 a 19 20 22
Tazlos
polar
nonpolar
ionic
Zoom In
wm
Transcribed Image Text:E 9th
2210
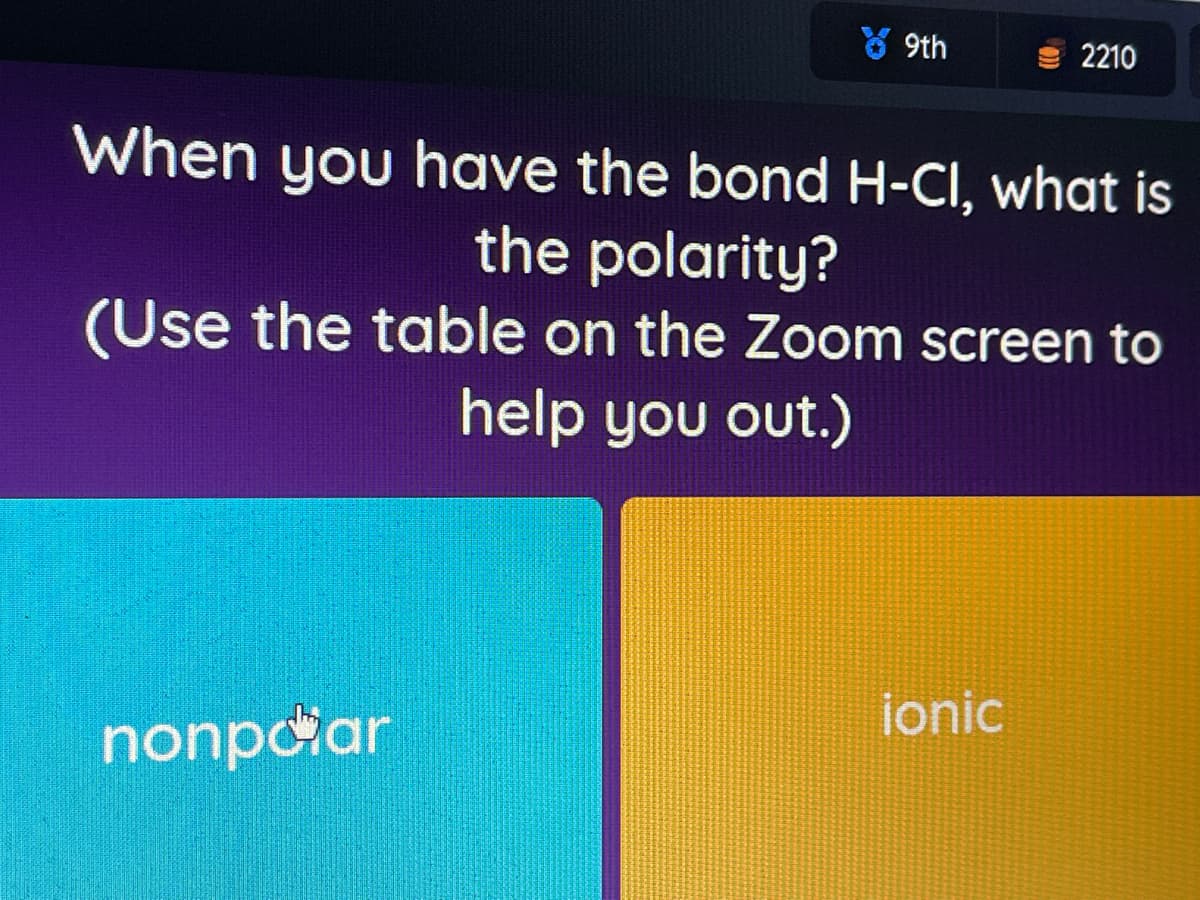
When you have the bond H-CI, what is
the polarity?
(Use the table on the Zoom screen to
help you out.)
ionic
nonpciar
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning