Which biological solution in Data Table II is the most acidic and what information helped you come to that decision? What can you explain about the chemical composition of the solution based on its pH?
Which biological solution in Data Table II is the most acidic and what information helped you come to that decision? What can you explain about the chemical composition of the solution based on its pH?
Chapter10: Reconstitution Of Powdered Drugs
Section: Chapter Questions
Problem 8SST
Related questions
Question
Which biological solution in Data Table II is the most acidic and what information helped you come to that decision? What can you explain about the chemical composition of the solution based on its pH?
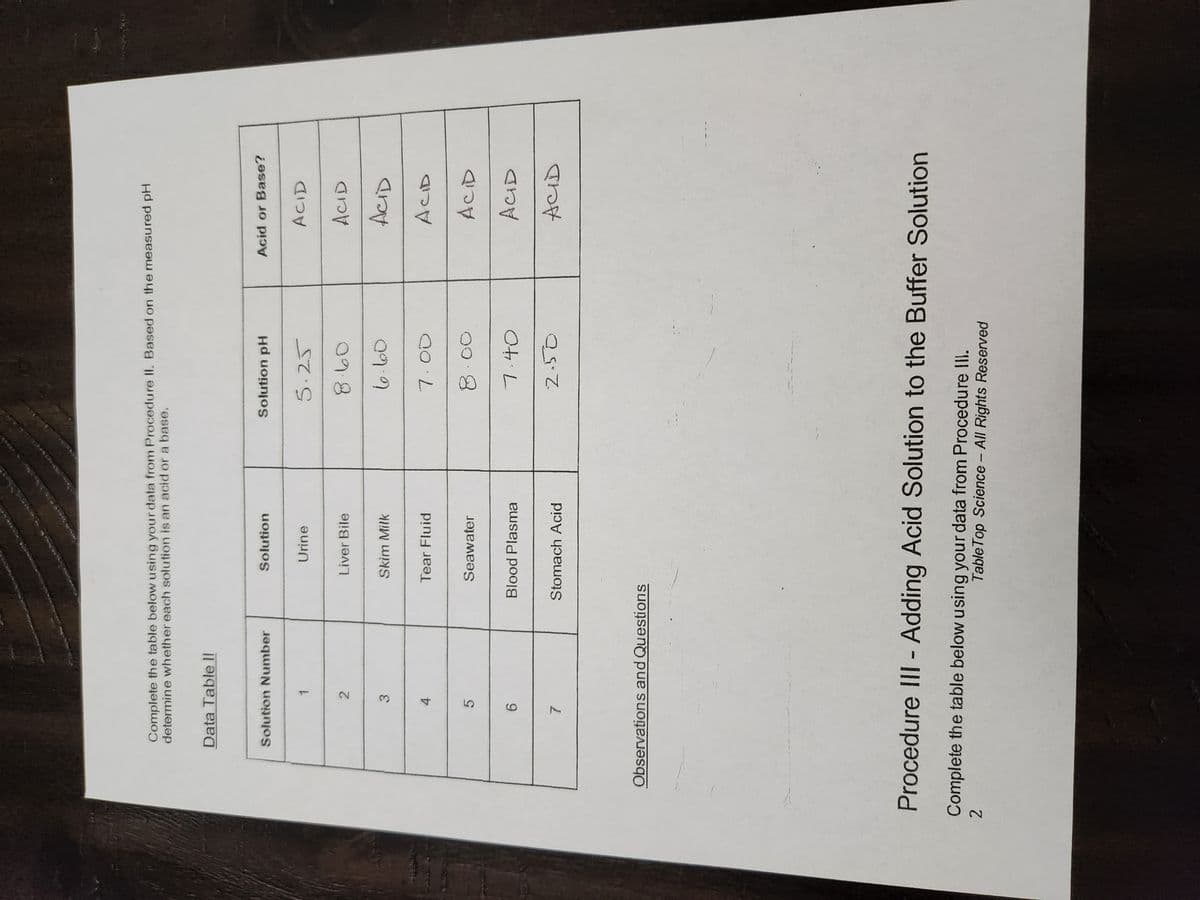
Transcribed Image Text:Complete the table below using your data from Procedure II. Based on the measured pH
determine whether each solution is an acid or a base.
2
Data Table Il
Solution Number
4
5
3
6
7
1
2
Solution
Urine
Observations and Questions
Liver Bile
Skim Milk
Tear Fluid
Seawater
Blood Plasma
Stomach Acid
Solution pH
5.25
8.60
6-60
7.00
7:40
2.50
Acid or Base?
ACID
ACID
ACID
ACID
ACID
ACID
ACID
Procedure III - Adding Acid Solution to the Buffer Solution
Complete the table below using your data from Procedure III.
TableTop Science- All Rights Reserved
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

