Q: 2. а) Predict possible product for each transformation. i. EtO,C. NaOEt ii. OCH3 OCH3 ii. СНО MENH2,…
A:
Q: Which undergoes faster SN1 substitution reactions with hydrogen halides, 2° or 3° alcohols? Explain…
A: Since, you have asked to solved multiple questions, so I will solved first question for you. You…
Q: i. H. Hl.. Rh 'CI insertion А Ph3P" H2C=CH2 ii. CO PPH3 -PPH3 Ph3P-Ru 'PPH3 +2 Ph-C=C-Ph В…
A:
Q: In which of these solvents would an SN1 reaction be the fastest? a.100% ethanol b.80% ethanol,…
A: e.100% water
Q: In each case below select the synthetic procedure that could be used to carry out the transform The…
A: A question based on addition to alkene, which is to be accomplished.
Q: 1. For each of the following pairs of SN2 reactions, indicate which reaction occurs faster: a)…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Assume one equivalent of reagents and appropriate workup after each step, unless otherwise…
A: Substitution reactions are the chemical reactions that involve replacement of one group by other.…
Q: -Br -Br Br II III Order of increasing reactivity
A: The compounds given are,
Q: Which conditions will cause the interconversion shown? how? H2 / Lindar catalyst a. LI/ NH3lig) H2 /…
A: Answer :- b) Li/NH3(liq)…
Q: Arrange these compounds from fastest SN2 reaction rate to slowest SN2 reaction rate. Fastest rate…
A: We have the 4 compound , we have to predict the rate of SN2 reaction of these compounds.
Q: n each case below select the synthetic procedure that could be used to carry out the transformation…
A: The product of the following reaction method are shown as follow.
Q: Identify reagents that can be used to complete the following transformation. OH O BH3-THF O 1)…
A:
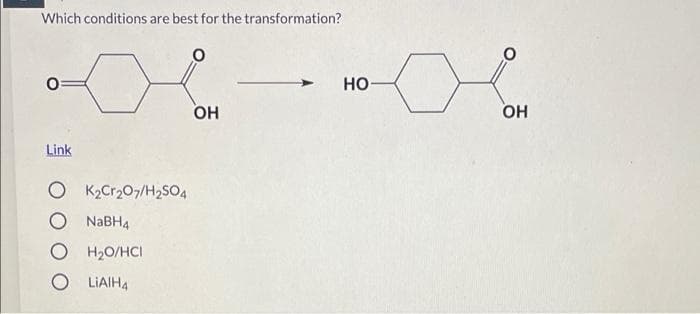
Q: Which is the best method for the following transformation? OH A. 1. BH3, THF 2. H₂O₂, OH- B. H¹, H₂O…
A:
Q: - he transformations above can be performed with some combination of the reagents listed below ne…
A: ->First of all Grignard reagent reacts with carbonyl and form alcohol. -> Then alcohol oxidize…
Q: CH2 ? H3C H3C COOH Reagents HBr e. PB13 f. NaCN then H3O* g. NBS, CCI4 h. CrO3, H2SO4 a. b. Mg,…
A: In this question, we have to find out the correct answer of given problem by the help of reagent…
Q: ycits – Where'd they go?! o your synthetic toolbelt to fill in reagents (a) - (g) in the spaces…
A: Given : We have to write the reagents and products for the following transformation.
Q: он The transformation above can be performed with some reagent or combination of the reagents listed…
A:
Q: reaction or conditions in each pair that you expect will react FASTER for the mechanism provided.…
A: Since you have asked a question with multiple sub-parts, we will solve first three sub-parts for…
Q: 2. The reaction below proceeds through two different pathways. 60% of reaction proceeds through Sn1…
A: Solution - According to the question - Given - The given reaction is a nucleophilic substitution in…
Q: Which molecule reacts fastest with ethanol by SN1 mechanism? 1-bromopentane 3-bromopentane…
A: Given molecules: 1-bromopentane 3-bromopentane 2-bromo-2-methylbutane 1-bromo-2,2-dimethylpropane To…
Q: i. Hl.. insertion Rh: Ph3P Rh PPH3 A H2C=CH2 CO ii. -PPH3 \PPH3 В Ph3P-Ru: 'PPH3 +2 Ph-C=C–Ph…
A:
Q: Venlafaxine, an antidepressant, was found to be metabolically unstable. The Medicinal Chemistry team…
A:
Q: CH3(CH2 )sBr NaOH CH3(CH2)5OH + Br + A) The rate would increase because SN2 reactions favor a polar…
A: Nucleophilic substitution biomolecular reaction (SN2): The bimolecular nucleophilic substitution…
Q: Which of the following series of reagents could be used to carry out the conversion shown? NO2 Br O…
A: Conversion of benzene into m-nitrobromobenzene.
Q: 7.4 Nucleophilic Strength in SN2 Reactions The reaction below will occur most rapidly in which of…
A:
Q: 4. Provide reasonable mechanisms for the conversions below. но- H3C, ICHS H,O* H3C CH3 он
A: The given mechanistic pathway demonstrates a ring contraction mechanism by means of…
Q: Provide the reagents necessary to carry out the conversion shown. + HO OH HO
A: 1. O3 2. DMS 3. Na2Cr2O7/H2SO4/H2O
Q: Arrange the compounds below in order of increasing reactivity in a SN2 reaction. 1. (CH3)2СНCI 2.…
A:
Q: For the transformation shown, select the most appropriate reagent(s) to effect the change, ?? H. 1)…
A: The reaction is the Ozonolysis Reagent : (1) O3 , (2) DMS (dimethyl sulphide) This is called…
Q: Q1 Radical Halogenation Show the complete (initiation, propagation, and termination) stepwise…
A: The given reaction follows the free radical mechanism that happens in three steps namely called:…
Q: Which molecule reacts fastest with ethanol by SNl mechanism? 1-bromopentane b. 3-bromopentane…
A: Answer : option c. 2-bromo-2-methybutane react fastest with ethanol by SN1 mechanism.
Q: In both examples below the reactants shown are combined to bring about a nucleophilic substitution…
A:
Q: Which of the following reaction conditions is NOT expected to result in the following…
A:
Q: Complete the reactions given below, write down the type of mechanism (SN1, SN2, E1, E2
A: SN1 and SN2 reaction require weak nucleophile and strong nucleophile.
Q: Which mechanism is going to predominate under these conditions? A SN1 SN2 Br NaOC(CH3) or t-buOH E1…
A: The reaction given is,
Q: 10. (8pts each) Rank the reactivity toward the SN1 mechanism. Place answers in the box provided. CH₂…
A: Concept: SN1 reaction: SN1 reactions are nucleophilic substitutions, involving a nucleophile…
Q: Which reagents will give the transformation shown? LOH (1) Вн3: (2) Н2О2 a. (1) Hg(OAc)2; (2) NaBH4…
A:
Q: NaOEt CI DMF
A:
Q: What type of mechanism is exhibited in the reaction? SN1, SN2, E2, E1?
A: In the given reaction, alkyl halide is primary and Nucleophile(KCN) is a strong nucleophile.
Q: Which Sn2 reaction would be expected to occur most slowly? O A. CH3CH2B + CN1-i B. CH3CH21 + CN1-i…
A: More good leaving groups more will be the reactivity of alkyl halide in SN2 since order of leaving…
Q: What is the expected product of the reaction sequence shown? Assume only the expected major product…
A: the reaction can be shown as:
Q: Which synthetic sequence will give the following overall transformation? how? (1) LDA / THF; (2)…
A: In given reaction alkyne is converted into alkane with addition of 1 carbon.
Q: Which of the following statements correctly describe SN2 and SN1 reactions? 1) SN2 reactions proceed…
A: To solve this problem we have to know about SN1 and SN2 reaction.
Q: .CI .CI aq. HBr 65 °C HO. Br
A: The reaction taking place is given as,
Q: In each case below select the synthetic procedure that could be used to carry out the transformation…
A:
Q: Which undergoes SN1 reaction fastest? BAU2000 54 Br Br Br Br
A:
Q: a) Senarai order berikut mengikut kereaktifan meningkat tindak balas SN1 List the following order of…
A: A question based on SN1 reaction, which is to be accomplished.
Q: In each case below select the synthetic procedure that could be used to carry out the transformation…
A:
Q: For SN1 Explain the order in which 1o (primary) alkyl halides reacted (fastest to slowest) and…
A: The rate of the SN1 reactions depends upon the concentration of alkyl halide only. This kind of…
Q: The oxymercuration/demercuration of the compound (methylcyclohexene) below will form what product?
A: Oxymercuration-demercuration of 1-methylcyclohexene with water and mercuric acetate is followed by…
Step by step
Solved in 2 steps with 2 images

- i need help filling out the following SN2 reactions with appopiate reactants, products, or reagents,In light of your answer to Problem 11-74, explain why one of the following isomers undergoes E2 reaction approximately 100 times as fast as the other. Which isomer is more reactive, and why?Order each of the following sets of compounds with respect to SN1 reactivity.
- Complete the following reaction equations :(i) C6H5Cl + CH3COCl →(ii) C2H5NH2 + C6H5SO2Cl →(iii) C2H5NH2 + HNO2 →Give 3 examples of a reaction mechanism of E1 that follows Zaitsev's rule.The first step of three different SN1 reactions are shown below. Which reaction proceeds the fastest and why? Rank the three steps of these reactions in order of increasing reactivity.
- Identify which substitution mechanism best fits the following statement: The reaction proceeds through a concerted mechanism. A) SN1 B) SN2Dialkyl peroxides are a family of compounds which can be explosive when heated. These explosions begin with the following bond fragmentation (which is then followed by other reaction steps). Which process best describes this fragmentation? E2 Elimination Heterolytic dissociation Homolytic dissociation RacemizationThe E1 mechanism (unimolecular elimination) of Elimination ?

