Q: Give a clear handwritten answer with explanation...give the mechanism of all sub parts reactions…
A: 1) SN2 mechanism: Concerted mechanism with inversion of configuration 2) E2 mechanism: Concerted…
Q: Using the information below explain why CN- is a stronger Bronsted base even though in the reaction…
A: Bronsted base is the species that accept proton.
Q: Please correct them and use the structural form the way it's written. Whether a mechanism…
A: E2 mechanism transition state requires an antiperiplanar orientation of the leaving group. Hence,…
Q: Rank the following species from best nucleophile to poorest nucleophile in an aqueous solution:
A: Nucleophilicity depends upon the tendency to donate electron pair. This tendency depends upon…
Q: ustify why do electrophilic substitution reaction in naphthalene take place at Alpha position. and…
A: Electrophilic substitution reactions are those in which displacement of a functional group by an…
Q: Solvent of reaction is methylene chloride. Part a) Draw arrow-pushing mechanim. Show ALL resonance…
A: The deprotection of a BOC-protected amine is a simple carbamate hydrolysis in acidic conditions. The…
Q: Compounds containing triple bonds are also Diels–Alder dienophiles.With this in mind, draw the…
A: Diels-Alder reaction is type of cycloaddition pericyclic reaction . It is [4+2] type cycloaddition…
Q: Increasing alkyl substitution stabilizes an alkene by an electrondonatinginductive effect. Explain ?
A: Alkene are hydrocarbon having carbon - carbon double bond . Increasing alkyl substituents ,…
Q: In the following pairs of nucleophilic substitution reactions, which one has a higher reaction rate?…
A: Given reactions are nucleophilic substitution reactions.
Q: (c) Nitrobenzene will undergo electrophilic aromatic substitution at meta position, but not at the…
A:
Q: following in the order of the Arrange reactivity to electrophilec aromatic Substitetrion reaction…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: (a) Delineate the fate of reactions with the support of detailed the following mechanism:
A: i)This reaction proceeds with a mechanism like a benzyne as shown below. (As specified by…
Q: Which of the following is compatible with a Friedel-Crafts reaction?
A: Friedel-Craft alkylation reaction: When the benzene reacts with an alkyl halide in presence of Lewis…
Q: Which of the following species cannot be considered an electrophile?
A: Electrophile is neutral or positively charged species. Nucleophile is neutral or negatively charged…
Q: Predict the product of photochemical ring closure of the three given, show the MO involved and…
A: Photochamical products findings
Q: mechanism for the of hydrolysis (1-bromo of Propose a reaction methylpropyl)cyclohexane. Including…
A: Stereochemistry is branch of chemistry in which we deal with arrangement of atoms in three…
Q: which
A: Nucleophilic substitution reaction in which an nucleophile substitute any group from reactant.
Q: or product(s) of
A: Addition of KMnO4/H+ results in oxidation of ketones. The reaction involves carbon–carbon bond…
Q: (a) For which aromatic compound do you expect nitration to take place faster: furan or thiophene?…
A:
Q: In the problem-solving feature above, methylcyclohexanone was seen to react at itsunsubstituted a…
A: The regiochemistry is not observed has to be explained.
Q: Which will react slower in polar protic solvents
A: Polar protic solvent is polar solvent.
Q: Which of the dompounds is most basic and why?
A: We have to select the most basic compound
Q: Why witting reaction is more suitable for making alkene as compared to other methods of preparation…
A:
Q: The following carbocation undergoes a rearrangement by a hydride shift as shown below. The reason…
A: Given, a carbocation which undergoes rearrangement by hydride shift We need to explain the reason…
Q: For each of the followung reaction, determine if diasteromer A or diasreromer B forms in greater…
A:
Q: Rank the following in order of increasingly exothermic heat of hydrogenation. Explain. A B
A: Enthalpy of hydrogenation is the amount of energy released when one mole of an unsaturated compound…
Q: Give a clear handwritten answer with explanation..give the mechanism with explanation of given bleow…
A: In this question, we will see the reaction mechanism for the given transformation from the reactant…
Q: How many different products obtained upon ozonolysis of are ?compound A
A: unsaturated compounds (alkynes,alkenes etc) reacts with ozone to form carbonyl compounds ,.…
Q: H.C.,
A: -> First of all in presence of methyl iodide methylation occur and quaternary ammonium salt…
Q: ome me
A: 1. Step 1 : Birch reduction. Step 2 : The starting material is treated with acid results…
Q: Give a clear handwritten answer with explanation..give the mechanism of given bleow reactions...
A: Butyl lithium will acts as strong base, it abstract acidic proton resulting a nucleophile is…
Q: Predict the main product of each of the following reactions with their
A: a. alpha hydrogen containing aldehydes or ketone in presence of base to give aldol first, further…
Q: CH₂OH H
A: Here we have explain major product formed in the following given reaction.
Q: Give a clear handwritten answer with explanation..give the mechanism of given bleow reaction with…
A:
Q: the reduction of α-methylvaleraldehyde, results to?
A: Reduction of aldehyde give alcohol
Q: Two of the following will give the same dimethylcyclooctatriene prod (A) while the third one will…
A:
Q: Draw the frontier orbitals for the thermal and photochemical cycloreversion of this reaction
A: Depending on the thermal and photochemical condition the reaction mechanism changes .
Q: The following reaction is Friedel-Crafts alkylation. Considering the mechanism for Friedel-Crafts…
A:
Q: What is the correct order of the words in the blanks in the following sentences? If there are…
A: The different properties of the substituents on the benzene ring depends upon the type of group…
Q: what is the plausible synthesis?
A:
Q: Fill in the blanks: The rate of the SN1 reaction depends on the and therefore involves kinetics.
A:
Q: Explain why the + charge of the carbocation always appears at the carbon of the (former) double…
A: CH2=CH2-CH2+ in this case of carbocation is stabilised by conjugation of double bond that means +ve…
Q: Provide the product and mechanism and justify regiochemistry .CI Но- of
A:
Q: Provide the mechamism for the given reaction.
A: In this question, we will draw the mechanism. First we need to redraw the structure of starting…
Q: Provide the complete mechanism using curved arrow formalism for the reaction of isopropylbenzoate…
A: Sodium benzoate can be separated from isopropyl alcohol by acidic hydrolysis. Benzoic acid is…
Q: Question The presence of which of the folowing conbutors to he intermedate for chionation of…
A: Electrophilic aromatic substitution is an organic reaction in which an atom that is attached to an…
Q: Consider the below molecule and its hydrogen atoms labeled a through e. Rank the named hydrogen…
A: acidity - increase order - Hd<Hc<He<Ha<Hb
Q: Explain clearly Giving and example and mechanisms, explain the aromatic Substitution reactions.
A: The electrophilic aromatic substitution reaction is the type of reaction where the pi-electron…
Q: among the following molecules, which one(s) would not be able to undergo a friedel crafts acylation…
A: The answer is as follows:
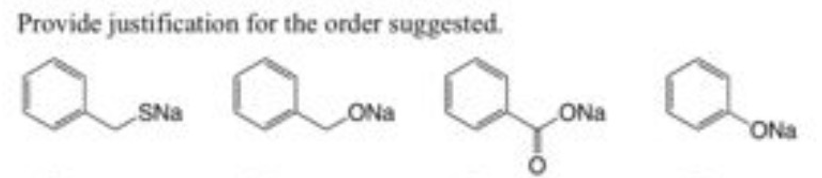
which is the most and least nucleophilicc, Arrange in the order of increasing nucleophlicity and provide reasons why
Step by step
Solved in 3 steps with 1 images

- Describe with a suitable example why the characterisation of chiral compounds is important to the pharmaceutical industry and indicate why separation of the enantiomers can prove challenging. Explain how capillary electophoresis can be used in the analysis of these compounds.. Discuss the truth of the following statement. Explain why it is true or false Every SN1 reaction produces racemic mixtures in the productsWhich of the statements below is FALSE regarding SN2 reactions?
- Explain the Mechanism - Isomerization of Geranyl Diphosphate to NerylDiphosphate ?During purification of 2,2’-bipyridine, the compound was accidentally exposed to a mineral acid. Elemental analysis gave the followingresults: C 62.35%, H 4.71%, N 14.54%. Suggest theidentity of the isolated compoundgive explanation in detail
- Represent a diastereoisomer of theproposed molecule, indicate theabsolute settings of your carbonschirals and discuss whether this is optically activeor not - justify:An organic chemist was subjected to sodium fusion and the resulting lassaigne's extract boiled with FeSO4 and acidified with concentrated H2SO4, yielding a prussian blue color. Give the structure of one of the organic compounds which fits the description.Calculate the index of hydrogen deficiency of this compound Q.) Ascorbic acid (vitamin C), C6H8O6

