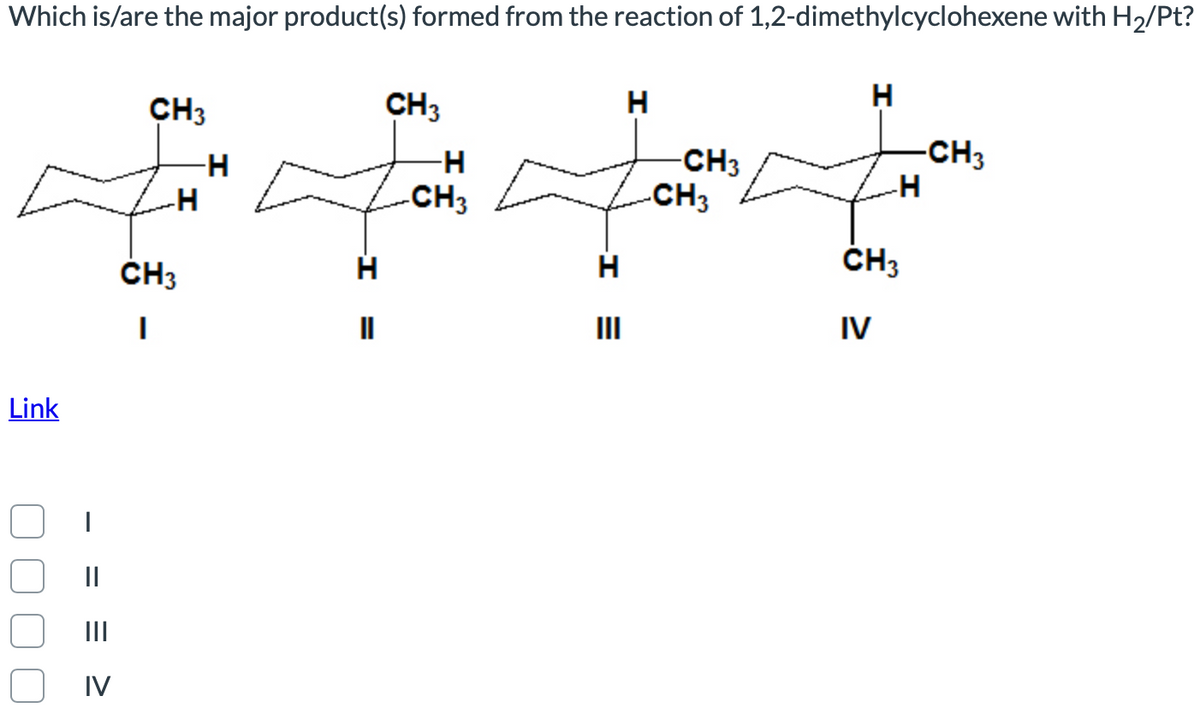
Which is/are the major product(s) formed from the reaction of 1,2-dimethylcyclohexene with CH3 CH3 H -CH3 -CH3 CH3 -CH3 ČH3 H ČH3 II II IV Link || II
Q: alkene. CH2 H2 H3C CH3 name:
A:
Q: What reagent/reagents is/are necessary to transform the starting molecule into the desired product?…
A: The above given reaction for the formation of Benzyl bromide using Toluene. It is based on free…
Q: The reaction CH3-CH=CH-CH3 CH3CH(I)CH(I)CH3 can be achieved with
A:
Q: What reagent/reagents is/are necessary to transform the starting molecule into the desired product?…
A: The conversion of starting molecule to desired product takes place in two steps. In step 1 the given…
Q: Match each alkene to its heat of hydrogenation.Alkenes: 3-methylbut-1-ene, 2-methylbut-1-ene,…
A: The structures of given compound are shown below:
Q: LEAST stable
A:
Q: Any proper mechanism of hydroboration- oxidation must explain the observed____[ex anti] addition of…
A:
Q: Oxidize the below molecule as many times as possible. Then name each product. ОН CH3 `CH3 H3C.
A: Given compound 2-methylbutan-2-ol
Q: KOCH,
A:
Q: Fed and select to confomen of the same trisubstituted crdohesane derivative from the brary of…
A: The question is based on the concept of conformers. conformers are the pair of species having same…
Q: What alkene is the major product formed from attached alkyl halide in an E1reaction?
A: E1 reactions are two step reactions in which the first step is the cleavage of carbon halogen bond…
Q: n of pure HBr with the alkene shown.
A: Alkene react with hydrogen bromide (HBr) to undergo addition reaction and forms alkyl bromide
Q: Draw the major organic product(s) of the following reaction. CH3 H,C-c-CEC-H CH3 NaNH2 / NH3()…
A: The given reaction is an example of elimination reaction of terminal alkyne using strong base sodium…
Q: a convenient method For making the bollowing Give conversions cyclopropyne a dibromocyclopropane
A: Given: cyclopropyne and 1,1-dibromocyclopropane To find: convert cyclopropyne and…
Q: Rank the following alkenes from most to least stable. А. В. С. D.
A: In this question we have to tell the stability of the alkene from most stable to least stable.
Q: i) CH2 = CH2 Reagent: Conditions: to CH2OHCH2OH CH3CHCH2 ii) CH;CH2CH,Br Reagent: Conditions: to…
A:
Q: What reagent/reagents is/are necessary to transform the starting molecule into the desired product?…
A:
Q: Draw the structure of the principal organic product in the blank box. Indicate stereochemistry where…
A: a). HBr addition to an unsymmetrical alkene in the presence of a peroxide goes via the generation…
Q: How many distinct monobromination products are chlorination? possible when the alkane below…
A:
Q: Label the following alkenes from most stable, moderate and least stable.
A: Various alkene is given Stability Order = ?
Q: Which of the following statement/s is/are false about the reaction of 3-methylhex-3-ene with H3O+?
A: The acid catalyzed reaction of water with alkenes results in the formation of alcohol. The product…
Q: The preparation of CH3-CH2-CH2- NH2 from CH3-CH2-Br could be achieved by using: O NACN then LİAIH4…
A: The product to be produced is a primary amine CH3-CH2-CH2-NH2 forming from CH3-CH2-Br.
Q: Give the major product(s) of the following reaction. AICI, ? heat There is no reaction under these…
A: Given:
Q: Upon hydrogenation, which of the following alkenes
A:
Q: What Is/are all af the possible productis) of the dehydrohalogenation of…
A:
Q: Use the Hammond postulate to explain why (CH3)2C = CH2 reacts faster than CH3CH = CH2 in…
A: The addition of HX to the alkene is the type of the electrophilic addition reaction. In this…
Q: IV II ||
A:
Q: How many alkene products are possible in the following reaction? NaOEt - ? CH3 "CH3 H
A: The above reaction is E2 elimination. H and Chlorine are anti to each other prefers E2 elimination.
Q: What is/are the possible product(s) of dehydrohalogenation of cis-1-bromo-2-ethylcyclohexane? Et…
A: Given that : We have to identify the product(s) of dehydrogenation of cisd-1-bromo-ethylcyclohexane.
Q: The reaction shown below is a typical reaction of alkenes and is generally known as what? + A-B H A…
A: •Alkene has double bond, which contains π-electron,so alkene act as electron rich species. •Due to…
Q: 1,3-dichloro 2-mctuyl cycionexanc 6-Chloro -s-ernyi decane mcloheptane Br 1-flu
A:
Q: Which compound is second least stable, giving off the second most heat of hydrogenation or…
A: Heat (enthalpy) of hydrogenation is defined as the change in enthalpy which occurs when one mole of…
Q: Draw the structure of the product of the reaction between the compound shown below and H,SO.…
A:
Q: Recall from Section 7.9 that most ketones and aldehydes exist primarily in their keto form, as shown…
A: Tautomerism refers to a phenomenon of isomeric forms, differentiated by proton transfer, existing in…
Q: Arvange the folloming alfkenes n the ader of stability A) B) c) D)
A:
Q: By hydrohalogenation of CHF2-CH=CH2 halogen atom is added to:
A: The Hydrohalogenation occurs via following processes : At first the alkene abstracts the proton…
Q: Name the alkene below. Use only E/Z designators to indicate stereochemistry.
A: Naming of alkene use the suffix -ene. E notation means two bulky group are present on opposite side.…
Q: reactions and products for C9H8O4 C9H8O4 + O2 --> C9H8O4 + H2O --> C9H8O4 + HCl --> C9H8O4 + OH-…
A: A chemical equation can be represented by using symbols and formulas of the participating species.…
Q: What other alkene is also formed along with Y in Sample Problem 9.3? What alkenes would form from X…
A:
Q: The first reaction can lead to over substitution. In the second reaction, no over substitution is…
A: Benzene gives an aromatic electrophilic substitution reaction. The reaction completes in two steps.…
Q: Which of the reactions favor formation of the products?
A: The high value of a compound implies that it is a weak acid. Since weak acid is more stable,…
Q: फोण्ड मैकट वकडुण्सर नoीन्ण्माल वड क्रचe नr विंडल false amd Explaim your gmswer ?ņ (i Alglic…
A:
Q: Dtermine the Product A from the rxn Et H2O, H2SO4 A heat Co2Et CO2H Et а. b. С. d. Et `Et
A: The first step is the attack of water molecules to the ester carbonyl groups to give the carboxylic…
Q: For each reaction, decide whether substitution or elimination (or both) is possible, andpredict the…
A: The reaction of haloalkanes with an electron rich species may either be a nucleophilic substitution…
Q: Match each alkene to its heat of hydrogenation. Alkenes: 3-methylbut-1-ene, 2-methylbut-1-ene,…
A: The structures of given compound are shown below:
Q: The image below shows a permitted cycloaddition reaction.?
A: For the permitted cycloaddition there should be supra-supra overlap between the terminal lobes of…
Q: n of cyclohexene with KMNO4 and NAOH at
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- reactions and products for C9H8O4 C9H8O4 + O2 --> C9H8O4 + H2O --> C9H8O4 + HCl --> C9H8O4 + OH- --> C9H8O4 + Na --> C9H8O4 + F -->What alkene is the major product formed from attached alkyl halide in an E1reaction?1. When you add H-Br in a terninal alkene, the product has the rule od Markovnikov? 2. The type pf intermetary thag forms when we add HgSO4/H2SO4/H2O in an alkyl is? Please solve my both questions.thank you.
- Which structure will not yield cis- or trans- isomers after a reaction with H2/Lindlar catalyst or Na/NH3?Elimination occurs when (Z)-3-bromohex-3-ene is treated with NaNH2. Under the same conditions, 1-bromocyclohexeneundergoes elimination much more sluggishly. Explain whyQ7 Consider the reaction, where the alkane shown is subjected to radical bromination at 25 °C. Br₂ light Describe the major monobromination product. product
- What is the expected major product of reacting cyclohexane carbaldehyde jwith CH3NH2?Assuming a 56.8% yield, how kany ml of alkene is required to produce 22.5 mL of 2,3-dimethyl-2-butanol? MW of alkene: 84.16, d: 0.653 MW of 2,3-dimethy-2-butanol: 102.17, d:0.823The reaction CH3-CH=CH-CH3 CH3CH(I)CH(I)CH3 can be achieved with
- For each reaction, decide whether substitution or elimination (or both) is possible, andpredict the products you expect. Label the major products. chlorocyclohexane + NaOC(CH3)3 in (CH3)3COHQuestão 10A certain hydrocarbon had the molecular formula C16H26 and contained two triple bonds. Ozonolysis resulted in CH3(CH2)4CO2H and HO2CCH2CH2CO2H as the only products. What is the reasonable structure for this hydrocarbon? Hexadec-6,10-dino undec-1,5-dino Hept-1,5-dino hex-1,5-dino nahDraw all products of the reaction of (1S,2R)-1-bromo-1,2-dimethylcyclohexane in 80%H2O/20%CH3CH2OHat room temperature..

