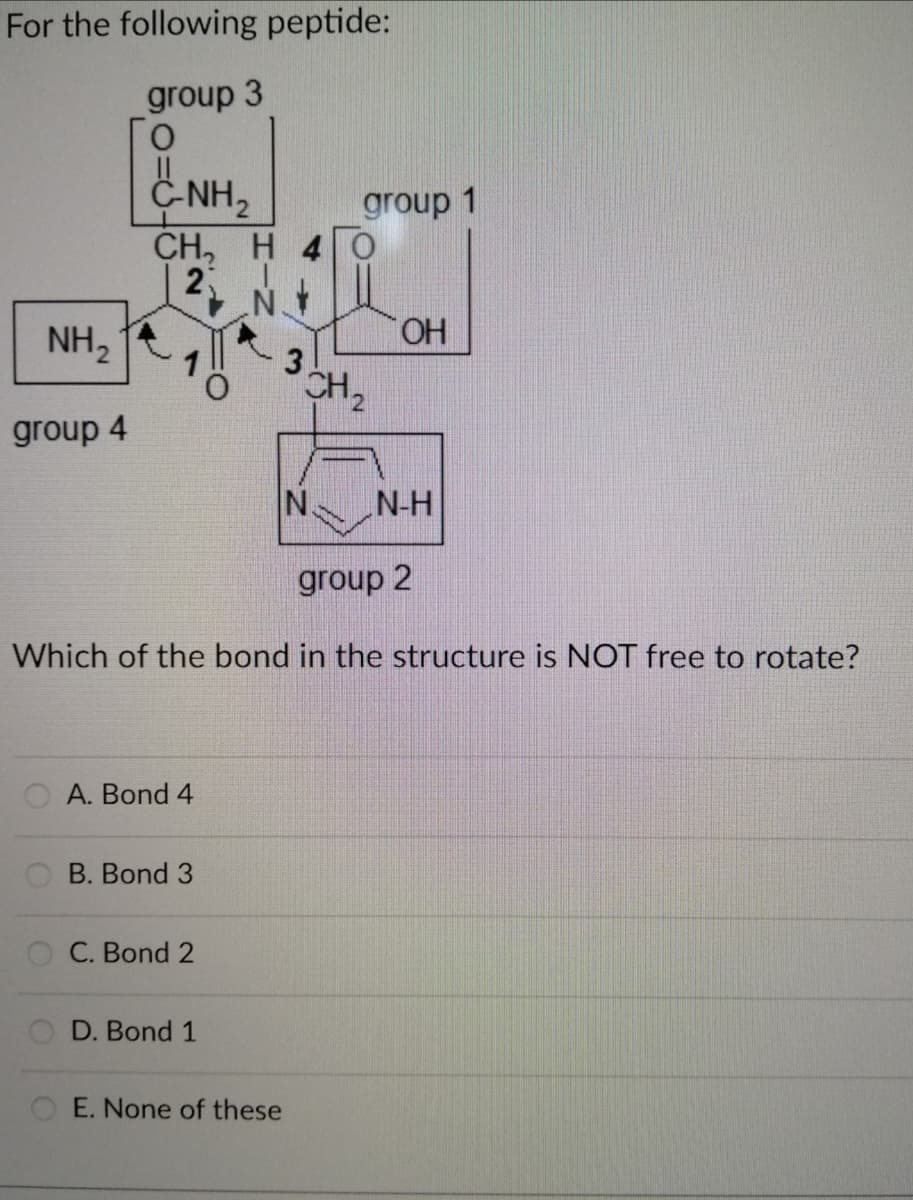
Which of the bond in the structure is NOT free to rotate?
Q: What are the disulfide bonds.
A: Protein is known to be a naturally occurring complex molecule substance. This is present in all…
Q: A polypeptide is shown below. Please answer the following questions. OH В A C H;N E H D H a. Match…
A: A polypeptide is a long, unbranched chain of amino acids joined by peptide/amide bonds. The peptide…
Q: Give the molecular formula of the functional group that is missing. a. NH+ b. CH3 c. COOH d.…
A: Q. Give the molecular formula of the functional group that is missing. a. NH+ b. CH3 c. COOH d.…
Q: Do any of the amino acid side groups shown below have the potential to form an ionic bond with any…
A: Ionic bonds are formed between the exchange of electrons between the donor and acceptor groups.
Q: Which of the following structures exhibit geometric isomerism? Draw and name the two in each case.
A: Introduction Isomers are compounds having the same molecular formula but have a different…
Q: Which of the structures below DOES NOT correspond to a "correct" (i.e. probable or possible)…
A: Carbohydrates are the compound of carbon, hydrogen and oxygen. It can be broadly classified as the…
Q: Identify the chirality center (sometimes called chiral atom) in each molecule. If the molecule does…
A: A chiral center is defined as an atom in a molecule that is bonded to four different chemical…
Q: Draw 2,2‑dimethylbutane. Show all hydrogen atoms. 2.)Draw 3‑ethylhexanoic acid. Draw the structure…
A: Organic compounds are the compounds that contain carbon-hydrogen bonds. There are many different…
Q: Which of the following best represents the backbone arrangement of two peptide bonds? A)…
A: Peptide bonds present between two amino acids. Two amino acids are joined together with the help of…
Q: An iconic bond is a bond in which ?
A: Bond is formed as a result of electrostatic attraction between two atoms which participate in the…
Q: ZI A IZ НО. B OH IZ N H НО, E ZI .N.
A:
Q: Which of the following creates peptide bonds?
A: Peptide bonds are covalent bonds and is responsible for linking amino acids and forming polypeptide…
Q: In the structure shown, which arrow is pointing to a peptide bond? OH LOH NH2 H OH HO-P=0 H. `A ÓH…
A: Peptide bond: It is a covalent bond used to join two amino acids to form a peptide chain. Generally,…
Q: Define the following under the topic of carbohydrates:
A: Carbohydrates are organic molecules that are polyhydroxy aldehydes or polyhydroxy ketones. They are…
Q: Identify the following parts of the polypeptide molecule shown below: A. Number each amino acid B.…
A: Proteins are the ultimate products of the genes. DNA is transcribed into m RNA and this is…
Q: H H |H I peptide chains H Type of bond Choose ペーUーエ アー O=U 1.
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Match the molecular geometry to its name. 1. Tetrahedral 2. Linear 3. Octahedral SUSMIT
A: Molecular geometry is the 3-D plan of the atoms that comprise a molecule. It incorporates the…
Q: holesterol has only one pi bond. What else can you say about its structure?
A:
Q: The configuration of the given structure below is: * CH,OH он ÓH O alpha-L O alpha-D O beta -L O…
A: The carbohydrates can be represented by two types of configurations: L-isomer and D-isomer. When the…
Q: Define the following terms:(a) Chiral (b) Achiral(c) Chiral carbon (d) Enantiomer
A: Stereoisomerism is a form of isomerism in which molecules have the same molecular formula and…
Q: Give the name of the structure below NH, CH 0-P-0-P-0-P-0- CH H он он
A: since you have posted 2 structures, we will help you out with the 2nd stricture only. firstly lets…
Q: The peptide bond is a stronger bond than the ester bond. What structural feature of the peptide bond…
A: The bond holding together two amino acids is a peptide bond. It forms when the carboxylic group of…
Q: List the 3 types of of macromolecules that form polymers, and next to each one list the monomers…
A: Macromolecules are large molecules or polymers which are formed from the simple monomers which are…
Q: Which of the following bonds is/are likely to be polar (choose all that apply)?
A: A type of covalent bond in which is formed between two atoms in which one atom is highly…
Q: What is the importance of having a strong chemical bond? Cite one practical use.
A: In human physiology, three types of chemical bonds are important because they keep together…
Q: Which of the following types of bonds are present in the primary structures of proteins? O Hydrogen…
A: Proteins are composed of twenty standard amino acids attached together through amide bonds. These…
Q: IUPAC name for the structure given. (DO NOT write "cis or trans") * ннннн ннн ннннн…
A: Systematic names given to a chemical structure is known as the IUPAC name. IUPAC stands for…
Q: In the structure shown, which arrow is pointing to a phosphoester bond? OH HO NH2 H ОН HO-P=0 `A ÓH…
A: DNA is the hereditary material in almost all organisms except some viruses. The basic unit of DNA is…
Q: why do the sodium ion and the chloride ion form a bond?
A: The two types of bonds which usually forms between atoms are 1) Ionic bond 2) Covalent bond
Q: Which among the bonds indicated here in a polypeptide structure is rotation impossible? (kindly show…
A: Polypeptide structure consists of amino acids which are linked to one another where the alpha…
Q: pairings that maximize the number of hydrogen bonds within pairs, what else do you notice about the…
A: Base pairing The DNA is composed of four nitrogenous bases which are Adenine represented as A…
Q: Which of the following applies to quaternary structure? It has two alpha and two beta subunits O a.…
A: Quaternary structure can be spotted in proteins consisting of two or more identical or different…
Q: what determines if a bond is polar?
A: A polar molecule is defined as in which the distribution of electrons are not even between the…
Q: Which of the following is not a comple lipid?
A: Note: There is an error in spelling error in the question stem but it seems that the question is…
Q: Which of the following best describes the glycosidic bond in the disaccharide shown? CH,OH CHOH CH…
A: A bond formed between the anomeric carbon atom of a monosaccharide and the oxygen atom of the…
Q: What are the names of the seven functional groups found in organic molecules? Underline the two that…
A: Organic molecules are the molecules with vast or large and complex chemical structures. These are…
Q: Indicate which orbitals overlap to form the s bonds in each molecule.a. BeBr2 b. HgCl2 c. ICN
A: a)There is an overlapping of SP-hybridized orbital of beryllium molecule with the P orbital of the…
Q: Structure Name Classification NH2 A) B) HN C) D) H. NH2 E) F) G) H) N. 1) J) NH N. ZI ZI ZI
A: In nucleic acids , attached to each of these sugars is a nitrogenous base that is composed of carbon…
Q: The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by…
A: At pH=1 (pH<7), there is an excess of H+ ions in the aqueous solutions. So, the COO-n group…
Q: The chemical structure of ethane (C2H6) is provided below. Which of the following isomers is ethane…
A: Option (5) none of the above is correct answer. (1) Ethane only have one form. It has too few…
Q: The peptide bond is a stronger bond than that of esters. Whatstructural feature of the peptide bond…
A: The strength of the peptide bond is largely attributable to the resonance between nitrogen and the…
Q: HH H H H C-C- Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η H H H-C-O C- онн C-C- H H H H H H H CH, H H…
A: There are many macromolecules present in the living organism. They include carbohydrates, lipids,…
Q: When you linked together with the structure of the triglycerides and the dipeptide bond, what will…
A: Introduction Triglycerides are a kind of fat. they're the foremost common sort of fat in your body.…
Q: Identify all of the chirality centers in the structure. `NH OH a h
A: The chiral center or atom (in this case carbon) has four different groups or atom attached to it.…
Q: What is the hydrolyzed component of the structure below
A: Phosphatidylserine is a phospholipid and is an intrinsic component of the cell membrane.
Q: Which of the following is a correct monomer/polymer pairing? O a. monosaccharide + monosaccharide +…
A: Carbohydrate is a biological macromolecules that are formed by three main element : carbon, hydrogen…
Q: Which of the following Fischer structure is repres structure? CHOH
A: The Fischer and Haworth projection are the two different ways to represent the organic molecules. As…
Q: In a portion of polypeptide shown below, which bond is described by g rotation? a b NH,--- ---CO
A: A dihedral angle or torsional angle is the angle that exists between two intersecting planes. In…
Step by step
Solved in 3 steps

- Which of the following is not true about the properties of a peptide (amide) bond? a.The bond order of the peptide bond is 2 b.The bond is in resonance which makes it (and the 6 atoms around it and included within it) planar c.Delocalization of electrons through the peptide bond contributes to a net dipole for the bond d.Peptide bonds are very, very strongly favored to be in the trans conformation, but proline is an exception because it is more isoenergetic due to its sidechain ring, and can be cis.The peptide bond is a stronger bond than that of esters. Whatstructural feature of the peptide bond gives it additional bondstrength?Peptide bond formation occurs between which functional groups? SELECT ALL THAT APPLY A. Amino B. Carboxyl C. Alpha-Carbon D. Nitrogen
- Which of the following correctly describes peptide bonds? * A. They are a special type of amide bond. B. They are very stable bonds. C. They form when water separates from an amino group and a carboxylic acid. D. They are a bond that shows resonance. E. All of these Which group consists only of amino acids with polar side chains? * A. serine, threonine, and leucine B. serine, threonine, and cysteine C. serine, threonine, and valine D. serine, threonine, and isoleucineWhich of the structures below DOES NOT correspond to a "correct" (i.e. probable or possible) structure for a monosaccharide? (remember that unlabeled vertices are carbon atoms) a) A b) B c) C d) DThe peptide bond is a stronger bond than the ester bond. What structural feature of the peptide bond gives it additional bond strength?
- Tertiary structure of protein is maintained by Select one: a. Hydrogen bond b. All of these c. Di-sulphide bond d. Peptide bondBased on the structure below: 1. Based on the chemistry, is the drug phototoxic? (Yes or No) 2. Is the drug is acid-stable? (Yes or No) 3. Which of the following is TRUE about the structure shown? A. The drug is doxycycline. B. The drug is resistant against dehydration. C. The drug forms into a lactone when dehydrated. The drug is bactericidal. D. The drug is phototoxic.For the tripeptide: methionylisoleucylcysteine (i) draw the structure of each tripeptide (ii) label the amide bonds in your structures (iii) identify the N-terminal and C-terminal amino acids
- Which of the following bonds is/are likely to be polar (choose all that apply)? a. H-H b. O-H c. C-H d. O-OWhich two types of bonding are in the 1ubq (Ubiquitin)? Explain your choices. a) Hydrogen bonding b) Ionic bonding c) Covalent bonding d) Disulfide bondingGiven the choices on the imageA. what is the structure of a phospolipid?B. structure of a 1,2-diacylglyrol (DAG)?C. structure of a monoacylglycerol (MAG)D. structure of ceramide

